INTRODUCTION
The rapa whelk, Rapana venosa (Gastropoda: Muricidae), one of the most important edible gastropods in Asia (Yoo, 1976; Kwon et al., 1993), it is found along the coasts of Korea, China and Japan, especially, in Korea, it is mainly found in silty sand of the intertidal and subtidal zones. However, the standing stock of this species has gradually been decreasing due to extensive loss of habitats from reclamation projects and reckless over-harvesting. Therefore, it has been noted as a target natural resources organism that should be managed by a reasonable fishing regime. To clarify the effect of female shell height on reproductive potential of a marine gastropod according to some differences in salinity concentrations, deposition of the egg capsules in female Rapana venosa (Valenciennes) inhabiting three different regions (the coastal zone of the Gwangyang Bay, the upper- and the lower reaches of the Seomjin River) should be investigated by dissecting microscopy and visual observations. Recently, the environmental, ecological surveys on environmental conditions of benthic fauna have been carried out around Gwangyang Bay and the upper-and the lower reaches of the Seomjin River. In this survey especially, a number of specimens of the rapa whelk R. venosa were collected by the fishing net in this area. In general, however, it is well-known that R. venosa inhabits in higher salinities in the seawater or brackish water. Exceptionally, some characteristics were found that especially, external morphological features of the egg capsules attached to the shells of the living rapa whelks in the special environmental area (low salinity) remarkably varied with those in the open seawater area with normal salinity. Therefore, comparative studies on morphological, reproductive characteristics of this species under open or relatively closed environmental conditions, such as high or low salinities and sufficient food organisms or resource management are very important.
So far, there have been some previous studies on R. venosa on reproductive aspects, including reproductive cycle (Chung et al., 1993, 2002; Chung & Kim, 1997), spawning and eggs capsules (Habe, 1960; Amio, 1963; Harding & Mann, 1999), on classification (Habe, 1969; Wu, 1988), on morphology (Lee & Kim, 1988). However, there is still uncertainty in many aspects of reproductive biology by brackish water with low salinity. Little information is available on correlations between shell height and egg capsule height, significance of egg capsule height, fecundity in the egg capsules per individual, and morphological variations of egg capsules by environmental factors such as normal sea water, brackish water with low salinity management and reproductive potential of R. venosa. Therefore, the purpose of the present study is to clarify correlations between female shell height and egg capsule height, Shell height and number of egg capsule per egg mass, and shell height and number of eggs and embryos in the egg capsule using calculation of Pearson's correlation coefficient and linear regression.
MATERIAL AND METHODS
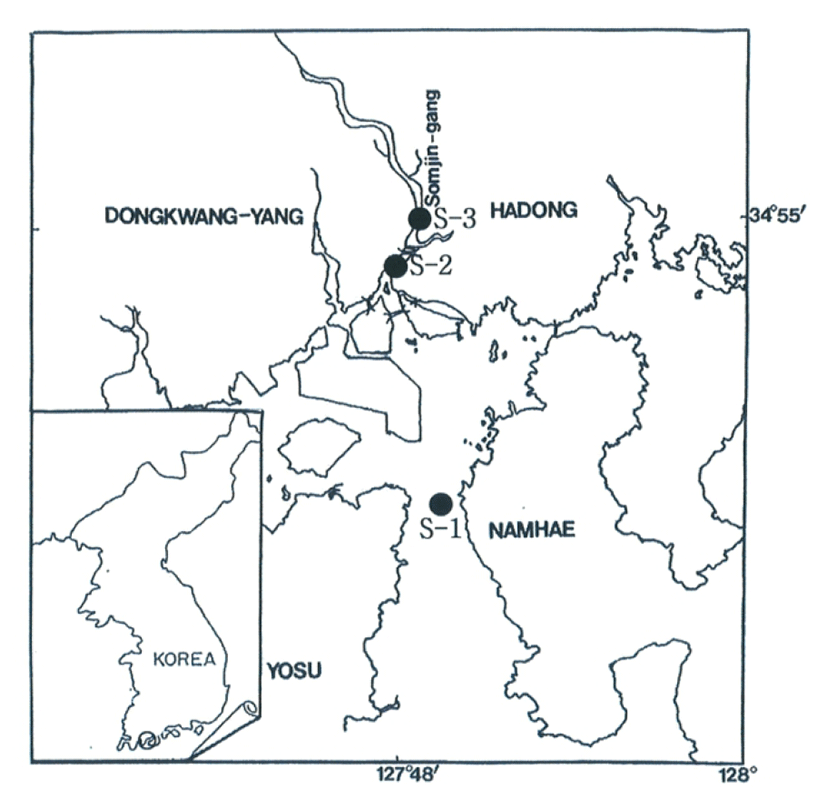
Seawater temperatures and salinity concentrations were quoted seasonally in the coastal zone of the Gwangyang Bay (S-1), the lower reaches of the Seomjin River (S-2), and the upper reaches of the Seomjin River (S-3), Hadong, Gyeongsangnam-do, Korea, from February to August, 2005.
To get some important sea water temperatures and salinities in the Gwangyang Bay and the brackish water temperatures in the upper and lower reaches of the Seomjin River, unpublished data of seawater temperatures and salinity concentrations in Gwangyang Bay measured daily at 10:00 am at the Gwangyang Iron Company, Gwangyang, Jeollanam-do, Korea were used for this study. And also unpublished data of brackish water temperatures and salinity concentrations in the upper and lower reaches of Seomjin River which is provided by Water Environment Information System of Yeongsan River Basin Environmental Office, Jeollanam-do, Korea were used for this study.
The specimens of the rapa whelk, R. venosa were collected during the spawning period (June to late July) by the fishing net in the coastal zone in Gwangyang Bay (S-1), the lower reaches of the Seomjin River (S-2), and the upper reaches of the Seomjin River (S-3) in Hadong, Gyeongsangnam-do, Korea (Fig. 1). The rapa whelks ranging from 4.90 cm to 10.74 cm in shell height in Gwangyang Bay (S-1), from 5.01 cm to 10.09 cm in the lower reaches of the Seomjin River (S-2), and from 4.90 cm to 9.64 cm in the upper reaches of the Seomjin River (S-3) were used for the present study. After transporting to the laboratory, shell height of female individuals collected at three regions (S-1, 2, 3) were immediately measured, and female rapa whelks used for this experiment were fixed with 10% neutral formalin.
To investigate the number of egg capsules per individual and the number of eggs and embryos in an egg capsule (collected at S-1, S-2, S-3 during June to July) were fixed in the formalin Boxes. The specimens were washed to remove the formalin in the tap water in an FRP indoor aquarium (80 cm × 60 cm × 60 cm) for one hour. The egg capsule heights of the adult rapa whelks (> 5.22 cm in shell hight) were measured and their egg developmental processes were checked under a dissecting microscope.
The specimens fixed with neutral formalin were collected at three different regions of salinity concentrations (S-1: the coastal zone of the Gwangyang Bay; S-3: the upper reaches of the Seomjin River; S-2: the lower reaches of the Seomjin River). To confirm the correlations between characteristics of rapa whelk at each zone, we calculated Pearson's correlation coefficient, and calculated the slope (a) and intercept (b) of the linear regression equation by carrying out the linear regression analysis between each variable.
RESULTS
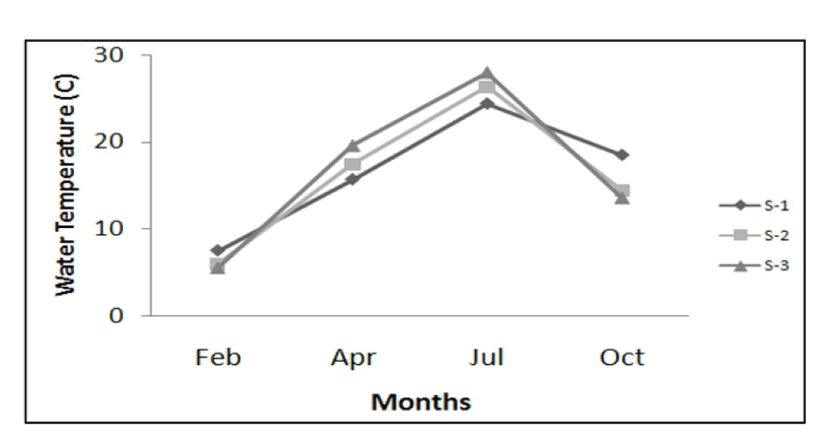
Environment seawater temperatures and salinity concentrations were measured in the habitats of rapa whelks at three different regions (S-1, S-2, S-3) from February to October, 2005. As shown in Fig. 2, during the study period, the range of seawater temperatures at S-1 were 7.5 – 18.5°C (average 16.5°C), those of S-2 were 6.0 – 14.4°C (average 16.1°C). And the range of seawater temperatures at S-3 were 5.5 – 13.6°C (average 16.7°C).
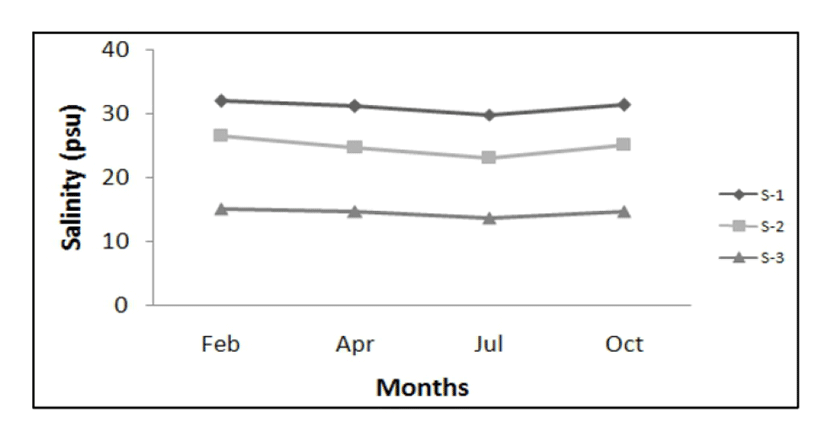
Environment salinity concentrations measured in the habitats of rapa whelk at three different regions (S-1, S-2, S-3) from February to October, 2005. As shown in Fig. 3, the range of salinity in the seawater at S-1 were 29.7 – 31.9 psu (average 31.0 psu), those at S-2 were 23.0 – 26.5 psu (average 24.8 psu). And the range of salinity at S-3 was 15.1 – 14.6 psu. (average 14.5 psu). In particular, salinity concentration in July temporarily decreased because of heavy rain.
As shown in Table 1, the range of female shell heights in three regions were 5.22 – 10.74 cm (average 8.49 ± 1.73 cm) at S-1, 5.01 – 10.09 cm (average 7.96 ± 1.59 cm) at S-2, and 4.90 – 9.64 cm (average 7.47 ± 1.50 cm), respectively. Over all, the size of female shell height at S-1 (coastal zone) was the largest, that of female shell height at S-2 (the lower reaches) was middle size, and that at S-3 was the smallest.
In three sampling areas during the spawning period of this species (between June and July), the smallest rapa whelk (4.90 cm SH) laid egg capsules with lower egg capsule heights of 1.24 cm (Fig. 4B) at S-3 (lower salinity average 14.5 psu) where is located in the upper reaches of the Seomjin River, while the largest rapa whelk (10.74 cm SH) laid egg capsules with high egg capsule heights of 2.82 cm (Fig. 4A) at S-1 (high salinity average 31.1 psu) where is located in Gwangyang Bay (Table 1).

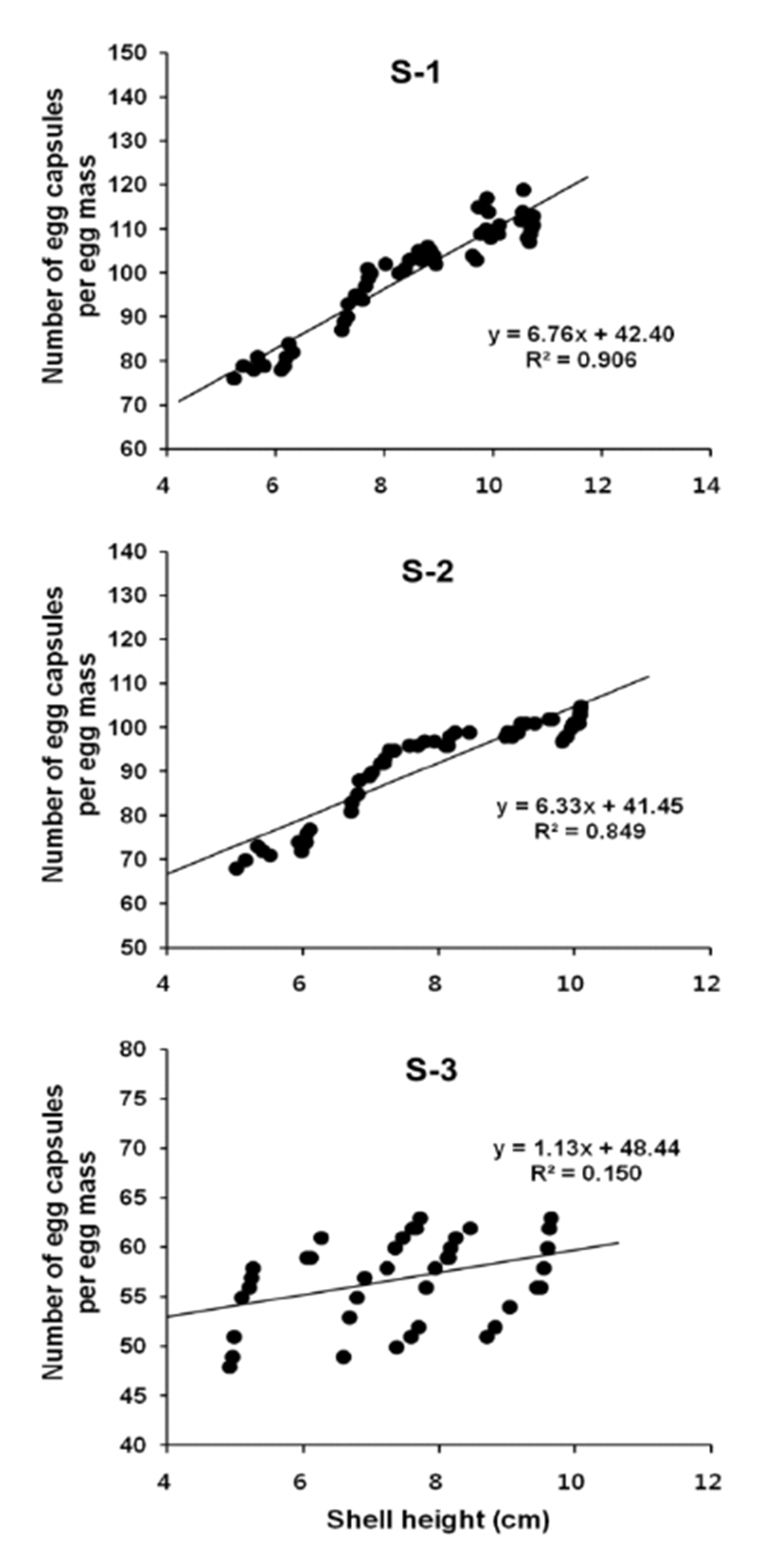
Of three sampling areas, during the spawning period the largest number of the egg capsules in an egg mass laid from an individual ranged from 76 – 119 (100 ± 12) at S-1 (coastal zone of Gwangyang Bay), were 68 – 105 (92 ± 11) at S-2 (the lower reaches of Seomjin River) and ranged from 48 – 63 (57 ± 4) at S-3. Therefore, the number of egg capsules laid from a female individual at S-1 showed larger than S-2 and S-3 habitats (Table 1).
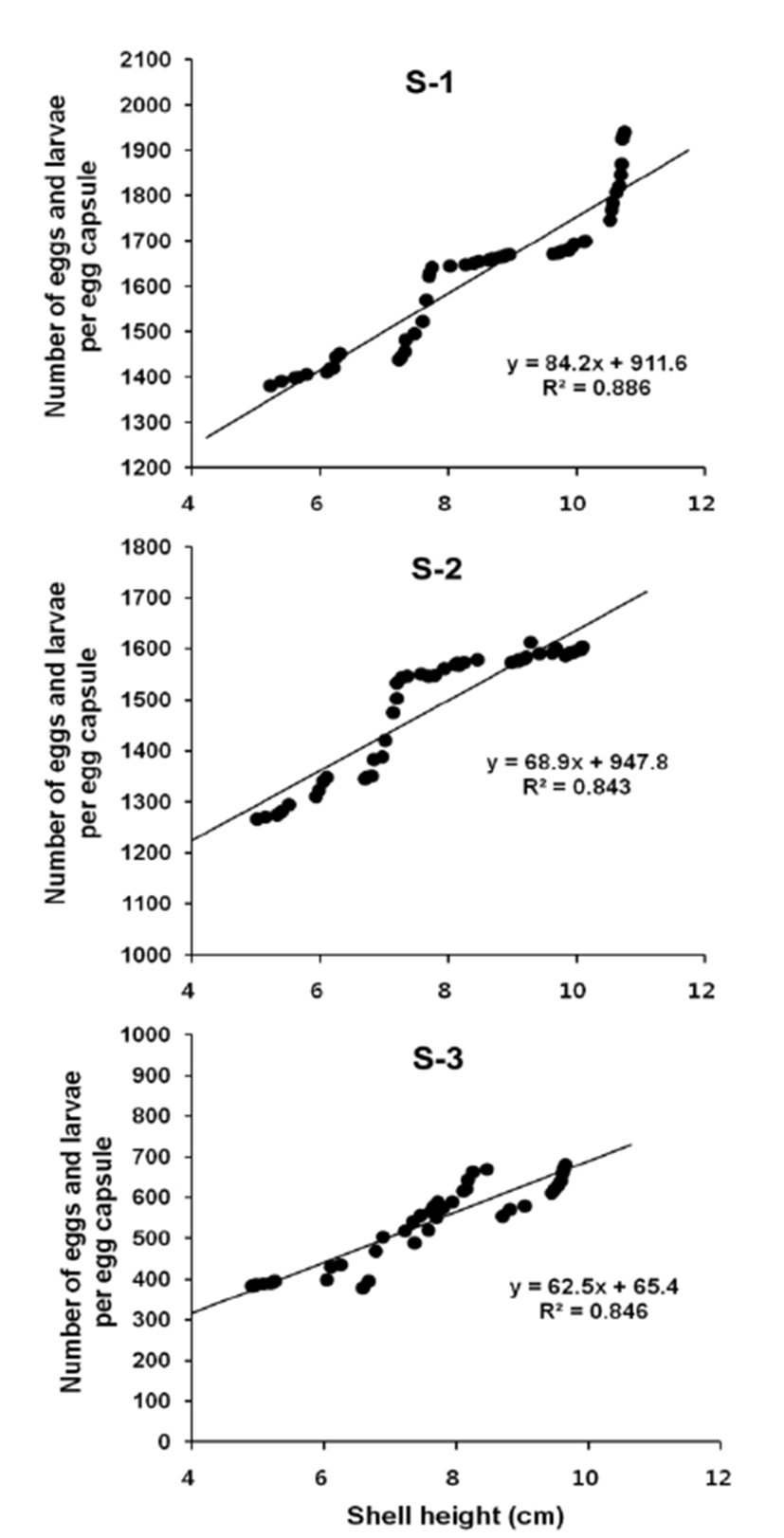
Fecundity (number of eggs and larvae) of the rapa whelks ranged from 532 ± 102 eggs (Fig. 4C) and larvae (Figs. 4D, E) in a low egg capsule height of 1.33 ± 0.06 cm at S-3 to 1,627 ± 155 eggs (Fig. 4C) and larvae (Figs. 4D, E, F) in high egg capsule height of 2.56 ± 0.15 cm at S-1. Over all, the fecundity of the rapa whelk increased with the increase of salinity concentrations and female shell height (Table 1).
To confirm correlation between female shell height and egg capsule height, female shell height and the number of egg capsules, female shell height and number of larvae, egg capsule height and number of the egg capsules, and number of egg capsules and number of larvae of rapa whelks Rapana venosa, we calculated Pearson's correlation coefficient and also calculated the slope (a) and intercept (b) of linear regression to conduct linear regression analysis between the variables.
As shown in Table 2, 3, 4. in S-1, S-2, S-3, the range of Pearson's correlation coefficients (r) between most variables showed r = 0.8 – 0.9 (p < 0.01) in S-1 and S-2. Therefore, in S-1 and S-2, there is a significant correlation between all variables. However, in S-3 (the upper reaches of Seomjin River), in particular, in case of correlations between female shell height and egg capsule height, Pearson's correlation coefficient (r) = 0.239, in this case, there is no significance between female shell height and egg capsule height. According to the results of correlation between female shell height and number of egg capsules, Pearson's correlation coefficient (r) was 0.387 (p < 0.05), In addition, in case of correlation between egg capsule height and number of larvae, Pearson's correlation coefficient (r) was 0.324 (p < 0.05). Although Pearson's correlation coefficients were relatively low value, these showed significant correlations between all variables.
| Variable | Shell height | Egg capsule height | Number of egg capsules | Number of larvae |
|---|---|---|---|---|
| Shell height | 1.000 | 0.982** | 0.952** | 0.941** |
| Egg capsule height | 1.000 | 0.920** | 0.941** | |
| Number of egg capsules | 1.000 | 0.899** | ||
| Number of larvae | 1.000 |
| Variable | Shell height | Egg capsule height | Number of egg capsules | Number of larvae |
|---|---|---|---|---|
| Shell height | 1.000 | 0.878** | 0.922** | 0.918** |
| Egg capsule height | 1.000 | 0.941** | 0.893** | |
| Number of egg capsules | 1.000 | 0.973** | ||
| Number of larvae | 1.000 |
| Variable | Shell height | Egg capsule height | Number of egg capsules | Number of larvae |
|---|---|---|---|---|
| Shell height | 1.000 | 0.239ns | 0.387* | 0.920** |
| Egg capsule height | 1.000 | 0.776** | 0.324* | |
| Number of egg capsules | 1.000 | 0.563** | ||
| Number of larvae | 1.000 |
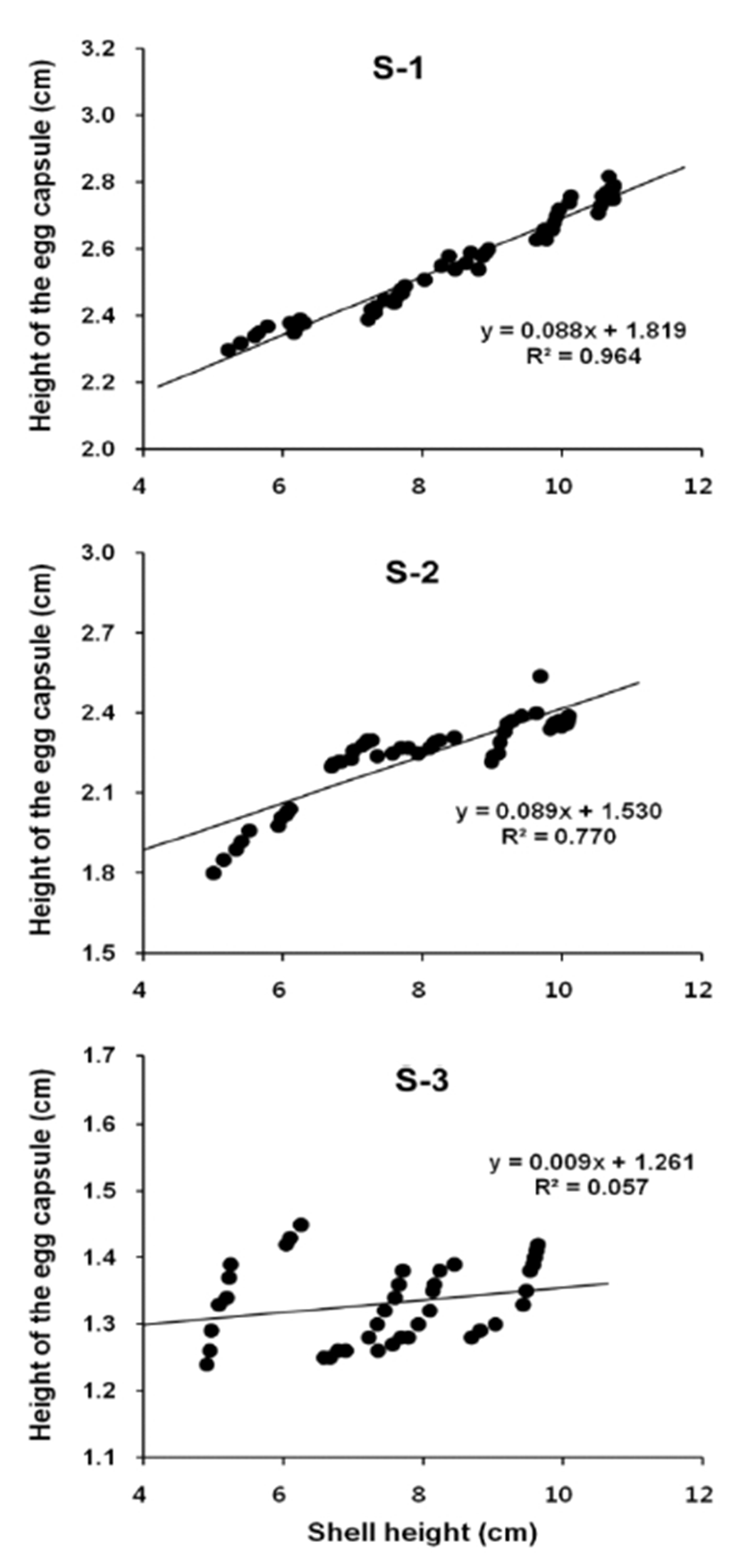
Egg capsule height in relation to female shell height was described using a linear regression equation. And correlations between variables were confirmed by the calculation of Pearson's correlation coefficient (r). In S-1, the egg capsule height = 0.088 SH + 1.819, R2 = 0.964 (Fig. 5, S-1). Pearson's correlation coefficient r = 0.982 (Table 2). Therefore, there is a significant positive correlation between Egg capsule height and female shell height. In S-2, the egg capsule height = 0.089 SH + 1.530, R2 = 0.770 (Fig. 5, S-2). Pearson's correlation coefficient r = 0.878 (Table 3). Therefore, there is a significant positive correlation between Egg capsule height and female shell height. In S-3, the egg capsule height = 0.009 SH + 1.261, R2 = 0.057 (Fig. 5, S-3). Pearson's correlation coefficient r = 0239 (Table 4). Therefore, there is no significant correlation between egg capsule height and female shell height.
Number of egg capsules per egg mass in relation to female shell height was described using a linear regression equation. And correlations between variables were confirmed by the calculation of Pearson's correlation coefficient (r). In S-1, number of the egg capsule per egg mass = 6.76 SH + 42.40, R2 = 0.906 (Fig. 6, S-1). Pearson's correlation coefficient r = 0.952. (p < 0.01). Therefore, there is a significant positive correlation between number of egg capsule height and female shell height (Table 2). In S-2, number of the egg capsule per egg mass = 6.33 SH + 41.45, R2 = 0.849 (Fig. 6, S-2). Pearson's correlation coefficient r = 0.922 (p < 0.01). Therefore, there is a significant positive correlation between number of egg capsule height and female shell height (Table 3). In S-3, number of the egg capsule per egg mass = 1.13 SH + 48.44, R2 = 0.150 (Fig. 6, S-3). Pearson's correlation coefficient r = 0.387 (p < 0.05). Therefore, there is a significant positive correlation between number of egg capsule height and female shell height (Table 4).
Number of eggs and larvae per egg capsule in relation to female shell height was described using a linear regression equation. And correlations between variables were confirmed by the calculation of Pearson's correlation coefficient (r). In S-1, number of the eggs and larvae per egg capsule = 84.2 SH + 911.6, R2 = 0.886 (Fig. 7, S-1). Pearson's correlation coefficient r = 0.941 (p < 0.01). Therefore, there is a significant positive correlation between number of eggs and larvae per egg capsule and female shell height (Table 2). In S-2, number of the eggs and larvae per egg capsule = 68.9 SH + 947.8, R2 = 0.849 (Fig. 7, S-2). Pearson's correlation coefficient r = 0.918 (p < 0.01). Therefore, there is a significant positive correlation between number of eggs and larvae per egg capsule and female shell height (Table 3). In S-3, number of the eggs and larvae per egg capsule = 62.5 SH + 65.4, R2 = 0.846 (Fig. 7, S-3). Pearson's correlation coefficient r = 0.920 (p < 0.01). Therefore, there is a significant positive correlation between number of eggs and larvae per egg capsule and female shell height (Table 4).
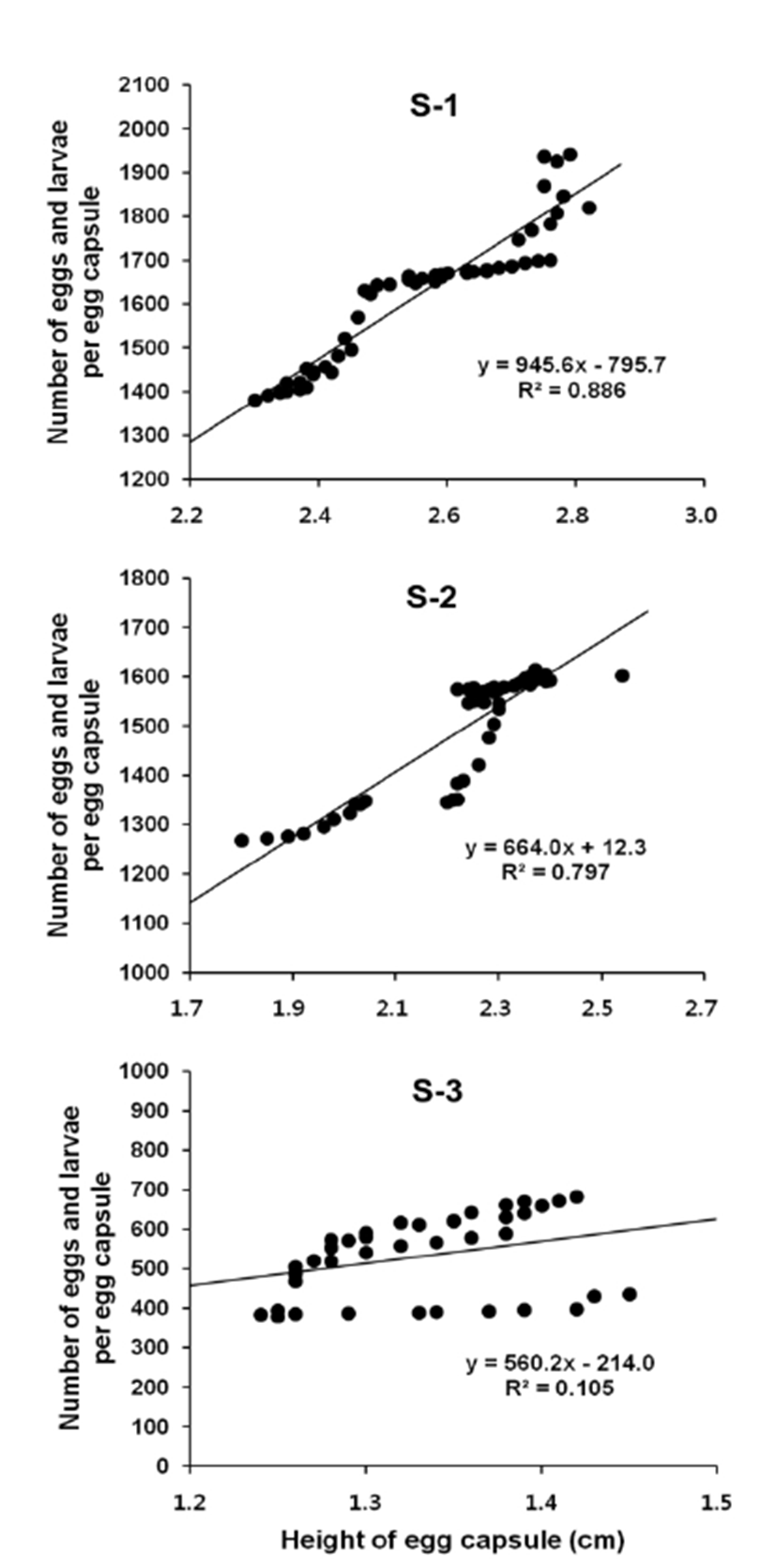
Number of eggs and larvae per egg capsule in relation to egg capsule height was described using a linear regression equation. And correlations between variables were confirmed by the calculation of Pearson's correlation coefficient (r). In S-1, number of the eggs and larvae per egg capsule = 945.6 Egg capsule height + 795.7, R2 = 0.886 (Fig. 8, S-1). Pearson's correlation coefficient r = 0.941 (p < 0.01). Therefore, there is a significant positive correlation between number of eggs and larvae per egg capsule and egg capsule height (Table 2). In S-2, number of the eggs and larvae per egg capsule = 664.0 Egg capsule height + 12.3, R2 = 0.797 (Fig. 8, S-2). Pearson's correlation coefficient r = 0.893 (p < 0.01). Therefore, there is a significant positive correlation between number of eggs and larvae per egg capsule and egg capsule height (Table 3). In S-3, number of the eggs and larvae per egg capsule = 560.2 Egg capsule height + 214.0, R2 = 0.105 (Fig. 8, S-3). Pearson's correlation coefficient r = 0.324 (p < 0.05). Therefore, there is a significant positive correlation between number of eggs and larvae per egg capsule and egg capsule height (Table 4).
DISCUSSION
The surface of the exit part of the egg capsule of R. venosa is flattened and has chitinous capsular form (Figs. 4A, B). The egg capsules in the genus Rapana have a curved sickle shape, with a long cylindrical stem, and an albumenous substance being contained in the egg capsules. In this study, the sizes (13 mm × 20 mm) of the egg capsules of R. venosa in low salinity (average 14.5 psu) and deficient food conditions at S-3 were smaller than those of the rapa whelk (average 26 mm × 25 mm) in higher salinity at S-1, as observed in the Japanese purple shell (30 × 2.5 mm) and those of the Korean shell (30 × 2.6 mm) in the western coastal waters of Korea (higher salinity) (Chung et al., 1993). Rawling (1990, 1994, 1995) and Middelfart (1994, 1996) described that the morphology of egg capsule varies within the marines neogastropods, showing differences in shape, size, surface texture (D'Asaro, 1966, 1970, 1988, 1991, 1993; Bandel, 1973).
Although all individuals belong to the same age class which were collected in the three regions of different salinities, the average value of their shell heights at S-1 were slightly larger than S-2 and S-3 regions because the growth pattern of shell height vary with salinity concentrations. Therefore, commonly female individuals, which female shell heights were larger, showed a tendency to be easily found in higher salinity.
The relationships between rapa whelk female size and egg case height, number of larvae per egg capsule, number of egg capsules produced per female whelk per year and fecundity, and the number of larvae per female per year were quantified and are described for a size range of rapa whelks from Gwangyang Bay and the upper- and the lower reaches of Seomjin River. Morphological differences in egg capsules of R. venosa according to shell heights at three different salinity areas appeared.
The relationships between rapa whelk female size and egg capsule heights showed that egg capsule heights produced by female rapa whelk increased with the increase of rapa whelk female size and salinity concentrations. In particular, of three different area of salinity concentrations, egg capsules of female rapa whelk at the S-1 area were the largest and longest, and those in S-3 area were the smallest.
The relationships between rapa whelk female size and total number of egg capsules per female rapa whelk showed that total number of egg capsules produced by female rapa whelk increased with the female size and salinity. Therefore, of three different area, total number of egg capsules (or egg masses) produced by female rapa whelk at the S-1 area (Gwangyang Bay: the highest salinity area) were the largest, and those in S-3 area (upper reaches of Seomjin River: the lowest salinity area) were the smallest. In this study, correlation between female shell height and number of egg capsules showed that there is a significant positive correlation.
The relationships between rapa whelk female size and the fecundity (number of developing eggs, embryos and larvae) per egg capsule showed that fecundity increased with the female size. Of three different areas, the fecundities of female rapa whelk at the S-1 area (the highest salinity area) were the highest, and those in S-3 area were the lowest. Therefore, the fecundities in S-2 area (the lower reaches of Seomjin River: the lower salinity area) were smaller in number than those in S-1 area. In the same conditions of salinity concentration, larger individuals being composed of rapa whelk female size (or individuals in the older age group) produced a larger number of egg capsules (with longer egg capsule height containing a larger number of fecundity) than those produced by individuals in the young age group. Consequently, in case of the rapa whelks, total number of larvae in the egg capsules per female individual was associated with the individual sizes, food availability and salinity.
Higher egg capsule height produced from the rapa whelks were mainly found at S-1 habitat while lower egg capsule height produced from females were particularly, found at S-3 habitat. Therefore, the rapa whelks in higher salinity habitat produced larger egg capsules, while those in lower salinity habitat produced smaller egg capsules. This fact provides a clear evidence that the level of egg capsule height is controled by salinity. In this study, the rapa whelks produced a number of egg capsules at S-1 where is higher salinity concentration habitat (average 31.1 psu, coastal zone), while the rapa whelks laid a small number of egg capsules at S-3 where is lower salinity concentration habitat (average 14.5 psu, upper reaches). In this case, the number of egg capsules produced by a female rapa whelk at S-2 habitat (average 24.8 psu, lower reaches) was larger than those produced by a female rapa whelk at S-3 habitat. Therefore, this fact provides a clear evidence that the number of the egg capsule produced is controlled by salinity.
In the present study, R. venosa in Korea spawned 192 – 382 egg capsules per individual, and the mean number of eggs per egg capsule was 500. Thus, the fecundity (the total number of eggs) from each individual ranged from 96,000 to 191,000 eggs. Chung et al., (1993) reported that the Korean purple shell spawned 984 to 1,241 eggs (average 1,096 eggs) in an egg capsule, and fecundity of the Korean purple shell ranged approximately from 320,000 to 450,000. Amio (1963) described that the number of eggs in each egg capsule of R. venosa in Japan ranged from 790 to 1,300. From 2002 to 2005, Buan Hatchery, National Fisheries Research and Development Institute has been investigated the spawning times during the spawning season from May to early August. in case of the largest rapa whelks, total spawning times ranged from 7 to 8 times for 3.5 months: average 2.00 – 2.22 times/month, However, in case of smallest rapa whelk, total spawning times ranged from 5 to 6 times for 3.5 months: 1.42 – 1.71 times per month. Therefore, it is assumed that fecundity of the rapa whelk at S-1 might be approximately, 182,000 – 1,302,000 eggs/ind./yr. Accordingly, this result is similar to the results reported by Harding et al. (2007).
Judging from these results, these results indicate a close similarity in reproductive output between the Korean and Japanese purple shells under the optimum salinity and sufficient food conditions with higher salinities, but fecundity of this species under lower salinity and deficient food conditions were smaller than those of Chung et al. (1993) and Amio (1963). Therefore, fecundity of the Korean purple shell may be closely related with external factors such as salinity and deficient food organism, etc.
In this study, the rapa whelks produced a number of egg capsules at S-1 where is higher salinity concentration habitat, while the rapa whelks laid a small number of egg capsules at S-3 where is lower salinity concentration habitat. In this case, the number of egg capsules produced by a female rapa whelk at S-2 habitat was larger than those produced by a female rapa whelk at S-3 habitat. Therefore, this fact provides clear evidence that the number of the egg capsule produced is controlled by the salinity. In this study, according to the results of correlations between female shell height and fecundity per egg capsule, using a Pearson's correlation coefficient and a linear regression equation, there is a significant positive correlation between variables at S-1, S-2, and S-3 regions. Therefore, it is assumed that female fecundity in the egg capsule increased with the increase of shell height.