INTRODUCTION
Equine chorionic gonadotropin (eCG) is a unique member of the gonadotropin family, as it can perform both LH- and FSH-like activity in non-equid species as well (Apparailly and Combarnous, 1994; Chopineau et al., 2001). Members of the glycoprotein family, which includes chorionic gonadotropin (CG), luteinizing hormone (LH), follicle-stimulating hormone (FSH), and thyroid-stimulating hormone (TSH), comprise two non-covalently linked α- and β-subunits (Min et al., 2004; Park et al., 2010). The β-subunits of eCG and eLH have identical primary structures (Min et al., 1994). Thus, eCG could be an ideal model for studying structure/function relationships of gonadotropins, as it exhibits the properties of both its pituitary and placental counterparts (Guillou and Combarnous, 1983; Galet et al., 2009).
In mares, eCG is secreted from binucleate trophoblastic cells, called endometrial cups, in maternal plasma during the first half of equine gestation. These cells detach from the chorionic girdle of the conceptus between days 37–120 of pregnancy (Boeta and Zarco, 2012; Flores-Flores et al., 2014; Conley, 2016). Progesterone has not been detected in the second half of gestation in mares (Conley, 2016). The presence of eCG during pregnancy may have a luteogenic effect, leading to the formation of supplementary CL after 35 days of pregnancy as a result of the ovulation and/or luteinization of dominant follicles at different times (Allen, 2001a). Previous studies on biological activity of rec-eCG, rec-eCGβ/α also had higher in vitro bioactivity than the eLH (Min et al., 1996). Deletion of carboxy-terminal peptides (CTP) from the dimeric eCG induced a 50% decrease in the secretion of the truncated hormone, compared to the wild type (Galet et al., 2000). The protein was truncated downstream of residue 109, which significantly decreased its secretion. Thus, the CTP upstream of the residues of the eCGβ-subunit at the 'seat-belt' region were also involved in the folding and secretion of gonadotropins (Galet et al., 2009).
In our previous study, we had produced rec-eCG from CHO-K1 cells, which were shown to exhibit dual LH- and FSH-like activity in rat Leydig and granulosa cells (Min et al., 1996, 2004), which expressed rLH/CGR and rFSHR (Park et al., 2009, 2010; Jeoung et al., 2010). A deletion downstream of amino acid 87 of the eCGα-subunit resulted in it not being secreted into the supernatant of the medium in both the transiently and stably transfected cells (Jeoung et al., 2010). A mutant with a deletion downstream of amino acid 95 of the eCGα-subunit showed no LH-like or FSH-like activities, indicating that the C-terminal region of the eCGα-subunit plays a pivotal role in the activity of cells expressing the rFSHR and rLH/CGR. We had also reported that the amino acid sequence at 94–96 of eCGβ-subunit appeared to be very important for signal transduction of the FSHR (Park et al., 2010).
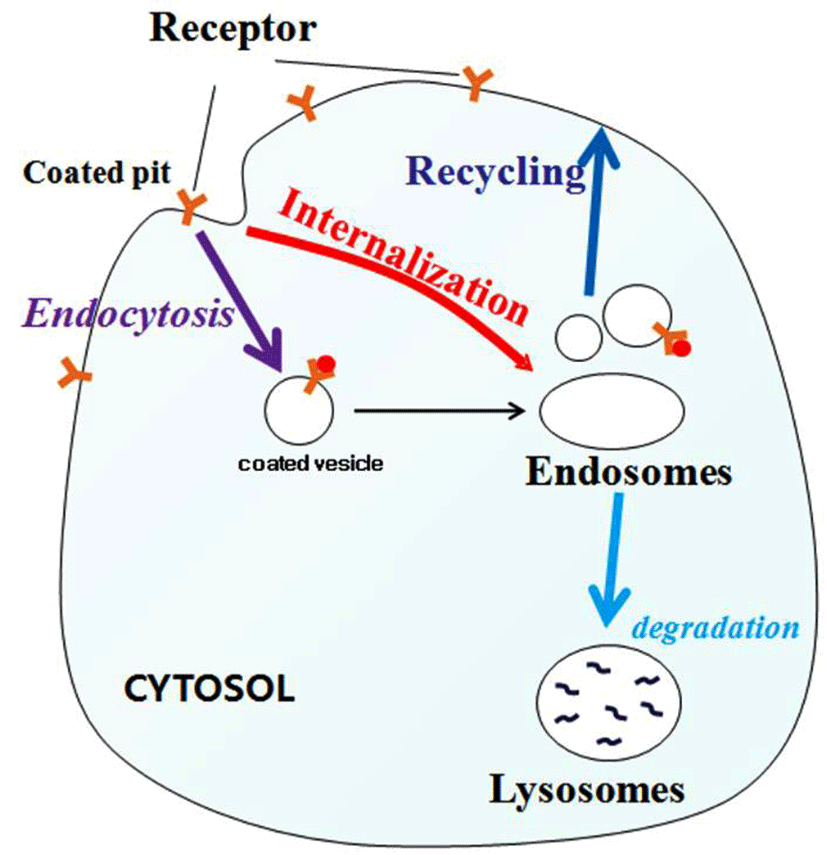
Along with the FSHR, LH/CGR is a member of the rhodopsin-like subfamily of G-protein-coupled receptors (GP CRs), which has been shown to mediate the internalization of its agonist (Segaloff and Ascoli, 1993; Ascoli et al., 2002; Flores-Flores et al., 2014). The internalized receptors are either degraded in lysosomes (Ghinea et al., 1992) or undergo resensitization and reinsertion into the plasma membrane (Ascoli and Segaloff, 1987; Lazari et al., 1998). Thus, the majority of internalized GPCRs are recycled back to the plasma membrane, instead of being routed to the lysosomes. Thus, internalization is thought to play an important role in receptor resensitization, rather than desensitization (Krupnick and Benovic, 1998; Lefkowitz, 1998).
We also reported on the function of the deglycosylated rec-eCGs produced from CHO-K1 cells (Min et al., 1996, 2004). However, we did not revealed the specific factors for the reduced biological activity in receptor cells. To better define the involvement of the biological activity of the rec-eCG stimulated cAMP synthesis, we have now analyzed the internalization of the rFSHR and rLH/CGR by deglycosylated rec-eCG. In the present study, we constructed a mammalian expression vector of rFSHR and rLH/CGR. The expressing vectors transfected to CHO-K1 cell and assessed the internalization of rFSHR and LHR by the bound rec-eCGβ/α.
MATERIALS AND METHODS
The expression vector pcDNA3 was purchased from Invirogen (CA, USA). CHO-K1 cells were obtained from the Japanese Cancer Research Resources Bank (Tokyo, Japan). Endonucleases were Boehringer Mannheim (MA, USA) and Takara (Osaka, Japan). Polymerase chain reaction (PCR) reagents were from Takara (Japan). Ham's F-12, CHO-S-SFM II, Geneticin, Lipofectamine 2000 and fetal bovine serum (FBS) were from Gibco BRL (MD, USA). The QIAprep-Spin plasmid kit was purchased from QIAGEN Inc. (Hilden, Germany). PMSG enzyme-linked im-munosorbent assay (ELISA) kit was from DRG (USA). The pCORON 1000 SP VSV-G tag expression vector, cAMP kit and CypHer 5-labeled anti-VSVG were from GE Healthcare Life Science (Buckinghamshire, UK). The oligonucleotides were synthesized by Green Gene Bio (Seoul, Korea). Fetal bovine serum was from Hyclone laboratories (Utah, USA). Centriplus Centrifugal Filter Devices were purchased from Amicon Bio separations (MA, USA). All other reagents used were from Sigma-Aldrich (USA) and Wako Pure Chemicals (Osaka, Japan).
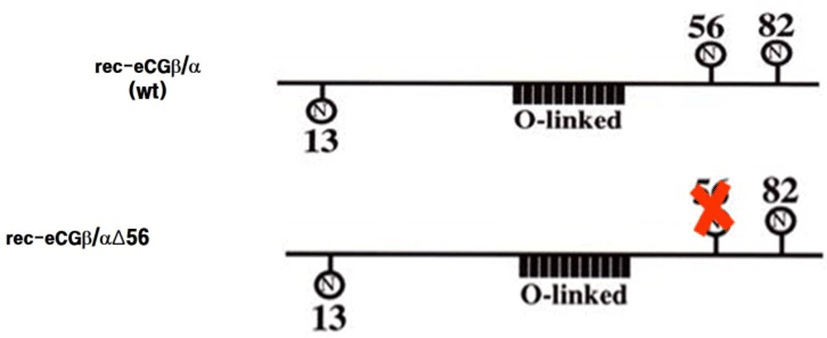
cDNAs encoding the tethered eCGβ/α were inserted in the pcDNA3 mammalian expression vector and used as templates to construct the mutants, in which Asn56 (AAC) at the glycosylated site of the α-subunit was substituted for Gln (CAG), as previously reported (Min et al., 2004). The epitope, myc-tag (Glu-Gln-Lys-Leu-Ile-Ser-Glu-Glu-Asp-Leu), inserted into the tethered eCGβ/α and mutant (eCGβ/ αΔ56) between the first and second amino acids in the β-subunit of the mature protein. Site-directed mutagenesis was performed using the three primers method in a single-step PCR (Min et al., 2004). The schematic diagrams of rec-eCGβ/αWT and eCGβ/αΔ56 are shown in Fig. 1. These fragments were digested with EcoR I and Sal I and ligated into the eukaryotic expression vector, pcDNA3. The plasmids were then purified and sequenced in both directions by automated DNA sequencing to ensure that the correct mutations had been introduced (designated as pcDNA3-eCGβ/α; β/αΔ56).
Cultured CHO-K1 cells were transfected into expression vectors using the liposome transfection method, as pre-viously described (Min et al., 2004). The transfected cells were cultured for 48 h in a serum-free medium (CHO-S-SFM-II) and then harvested and centrifuged at 15,000 rpm for 10 min. The supernatant was collected and stored at −20℃ until the assay. Six to 8 pools of stably transfected cells were selected by incubating the cells in a growth medium [Ham’s F12 medium with 10% FCS, penicillin (100 IU/mL), streptomycin (100 μg/mL) and glutamine (2 mM) supplemented with G418 (800 μg/mL)] for 2–3 weeks after transfection, as previously reported method (Min et al., 2004). The culture medium was centrifuged at 15,000 rpm for 10 min to remove cell debris. The supernatant was collected and concentrated in an Amicon Stirred cell concentrator and stored at −20℃ until the assay.
The tethered eCG proteins secreted by the transient transfected CHO-K1 cells and the stable cell lines into serum-free media were collected, and rec-eCG was quantified using the PMSG ELISA (enzyme-linked immunosorbent assay) kit, according to the supplier's protocol (DRG, USA). Briefly, the collected medium was dispensed into wells coated with a monoclonal antibody against unique antigenic site on the eCG molecule. The wells were incubated for 60 min at RT and 100 µL of conjugate was then added into each well. The substrate solution (100 µL) was also added, and the mixture was then incubated for 30 min at RT. Finally, 50 µL stop solution was added to stop the enzymatic reaction. The optical density (OD) of the product solution was read at 450 nm with a micro titer plate reader.
The cDNAs for rLH/CGR and FSHR were cloned and expressed in CHO-K1 cell lines, as previously described (Min et al., 1998; Min, 1999). The receptor cDNAs were subcloned into the eukaryotic expression vector, pCORON 1000 SP VSV-G tag (GE Healthcare Life Science, Buc-kinghamshire, UK), for transfection. Stably transfected cell lines were selected by G418 treatment, as previously described (Min et al., 2004, Seo et al., 2015). Briefly, cloned cells were cultured in standard 96-well microplates at concentrations of 105–106 cells/mL and incubated overnight. Cholera toxin was added and the mixture was incubated for 30 min. Finally, the cyclic AMP kit was used according to the supplier's protocol, and the OD was measured at 630 nm with a microplate reader. The establishment and pro-perties of a clonal cell line expressing vsvg-rLH/CGR-wt [designated as vsvg-rLH/CGR (wt-9)] and one expressing vsvg-rFSH-wt [designated vsvg-rFSHR (wt-7)] were studied based on cAMP responsiveness to an agonist, cholera toxin, as previously reported (Park et al., 2009).
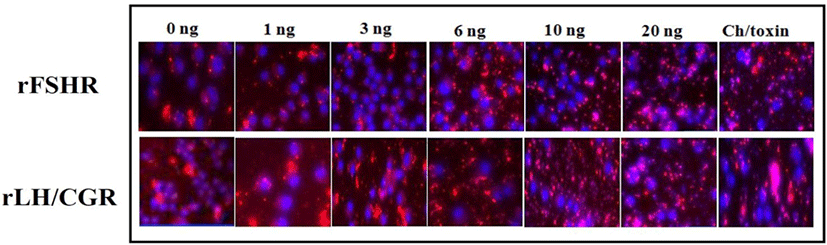
A novel assay technology, which relies on the intrinsic ability of cell surface receptors to internalize into acidic endosomal vesicles upon agonist stimulation, was recently reported (Lowe et al., 2015). This technology uses a novel, fluorogenic, pH-sensitive cyanine dye, CypHer 5. It is there- fore ideal for reporting the movement of receptors from the cell surface into the acidic internal endosomes. The rLH/CGR cells and FSHR cells were seeded into a 96-well tissue culture view plate (Perkin Elmer; 6005710, pre-coated with poly-D-lysine) at a density of 2×103 cells/cm2 in a complete Nutrient Mix F-12 Ham’s medium with G418 (700 µg/mL). The cells were incubated at 37℃ overnight until the cells were 20–40% confluent. The supernatant was removed and the cells gently washed twice with Kreb's Ringer Buffer (KRB) (120 mM sodium chloride, 25 mM HEPES, 4.8 mM potassium chloride, 1.2 mM potassium dihydrogen orthophosphate, 1.2 mM magnesium sulphate heptahydrate, and 1.3 mM calcium chloride) at pH 7.4. The culture medium was replaced with 70 µL/ well of KRB. KRB with CypHer5-labeled antibodies (2-5 µg/mL), 1μM Hoechst 33342, and 1% FBS was then added to the plate. After incubating at RT for 10 min, 30 µL of KRB containing the agonist (1, 3, 6, 10, and 20 ng/mL of rec-eCG and 500 ng/mL of cholera toxin) was added to the plate. The mixture was incubated at RT for 60 min, and 75 μL/well of ice-cold 8% paraformaldehyde was then added. After incubating on ice at 4℃ for 20 min, the supernatant was removed and the cells were gently washed twice with PBS. Finally, 100 μL/well of PBS was added. The Cy5-labeled receptors and the Hoechst 33342 were visualized with the In Cell Analyzer 3000 device (GE Healthcare Life Science, Buckinghamshire, UK).
RESULTS
The eCGβ/α and rec-eCGβ/αΔ56 vectors were transfected into CHO-K1 cells and the cells stably expressing genes were then selected with G418 treatment. The rec-eCGs secreted by the stably transfected cells into the serum-free medium were isolated and concentrated. The rec-eCGs were quantified using ELISA. The quantity of rec-eCG obtained from stably transfected cells was about 180-240 mIU/mL. The size of the rec-eCGβ/α protein was found to be approximately 40-45 kDa (data now shown), which was consistent with previous studies (Park et al., 2009, 2010; Jeoung et al., 2010). However, the size of the rec-eCG secreted by the mutant with the deglycosylated Asn56 in the α-subunit was decreased approximately 4-5 kDa.
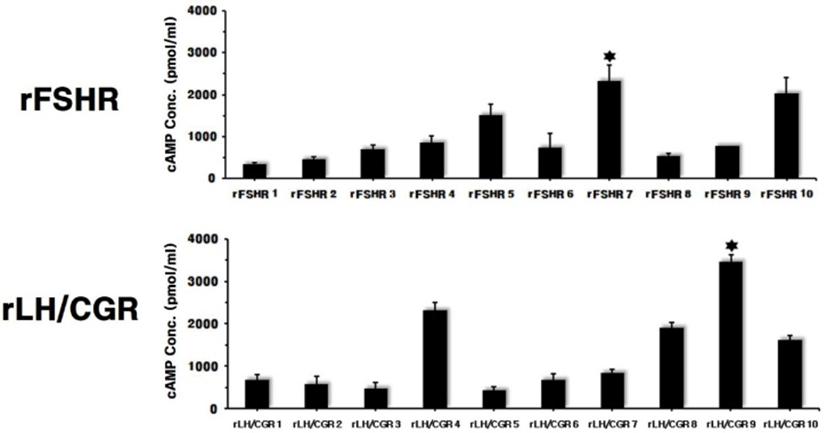
The mammalian expression vectors for rFSHR and rLH/ CGR were transfected into CHO-K1 cells. The 10 clones were then isolated by G418 treatment. The cAMP level was determined to evaluate the response for agonist stimulation. The most stimulated cells were rFSHR-7 and rLH/ CGR-9, as shown in Fig. 2. The maximal cAMP responses were 2,100 pmol/mL and 3,500 pmol/mL in the rFSHR and rLH/CGR, respectively.
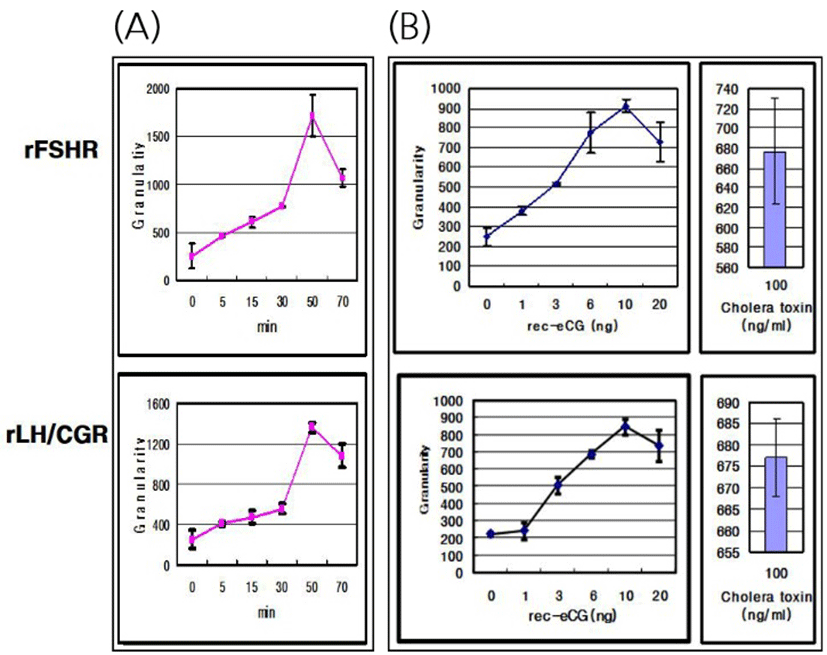
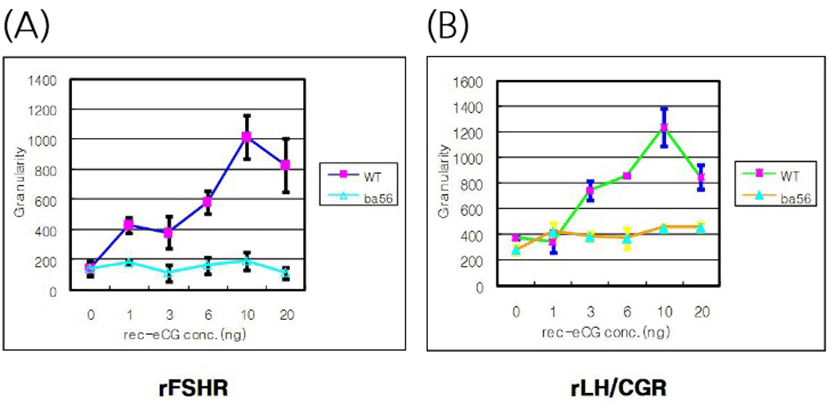
The effects of rec-eCGβ/α on the internalization of rFSHR and rLH/CGR in CHO-K1 cells were determined in a time-dependent manner (0–70 min). As shown in Fig. 3A, rFSHR rLH/CGR were most internalized at 50 min after incubating rec-eCGβ/α (10 ng/mL). We also measured the effect of rec-eCGβ/α on the internalization by receptors in a concentration-dependent manner (Fig. 3B), and found that the internalization increased proportional to the concentration of rec-eCGβ/α. The granularity values for both the receptors were found to be 800–900. The results of the agonist activation experiment also suggested that both the receptors were internalized in endosomes. Therefore, we used Cy5-labeled receptors to detect the localization after 60 min of incubation with rec-eCGβ/α (Fig. 4). Next, we analyzed the differences between the effects of rec-eCGβ/α and rec-eCGβ/αΔ56 on stably expressed these receptors (rFSHR-7 and rLH/CGR-9). The results of this analysis showed that the rec-eCGβ/αΔ56 in cells expressing rFSHR and rLH/CGR was completely flat; no agonist-stimulated internalization was induced in either cell type by eCGβ/αΔ56 (Fig. 5A, B). Together, these data suggested that rec-eCGβ/α induced receptor internalization in cells expressing rFSHR and rLH/CGR. However, the mutant with the eCGβ/αΔ56 did not induce this agonist stimulated internalization. Thus, the glycosylation site (Asn56) in the eCGα- subunit played a pivotal role in receptor internalization.
DISCUSSIONS
The results of the present study showed that rec-eCGβ/α induced the internalization of rFSHR and rLH/CGR in CHO- K1 cell lines that expressed those receptors in a time- and con- centration- dependent manner. We also showed that deglycosylation at Asn56 of the α-subunit of rec-eCGβ/α inhibited this agonist-induced internalization in both cell types.
Previous studies have demonstrated that rec-eCGβ/α was expressed in the baculovirus-Sf9 insect cell system, either as a single-chain with the C-terminal of the β-sub-unit fused to the N-terminus of the α-subunit or as non-covalently linked heterodimers with or without polyhistidine tags at various locations (Legardinier et al., 2008). Heterodimeric rec-eCG exhibited the same thermal stability as natural pituitary LH, and its advantages over the single-chain eCG included higher secretion, higher in vitro bioactivity, and lower risk of immunogenicity (Legardinier et al., 2008). From the labeling and pulse-chase experiments, the secretion of the eCGβ-subunit from CHO-K1 cells was inefficient (medium recovery of 16–25%) and slow (t1/2>6.5 h). This secretion of eCGβ-subunit resembled that of hLHβ-subunit, rather than that of the hCGβ-subunit (Cohen et al., 2015). In a previous study, rec-eCG was expressed in commercially available MimicTM insect cells that were stably transformed into Sf9 cells expressing five mammalian genes that encoded glycosyltransferases involved in the synthesis of complex-type monosialylated N-glycans. the rec-eCG produced from these MinicTM insect cells showed mammalian-like nonsialyl complex-type N-glycosylation (Legardinier et al., 2005). Natural eCG contains complex-type N-glycans terminating with both Siaα2,3Gal and Siaα2,6Gal. In previous studies, we had reported rec-eCG production in CHO-K1 cells (Min et al., 2004; Park et al., 2009, 2010; Jeoung et al., 2010). But, the specific glycosylation site is very important for the secretion of rec-eCG in CHO-K1 cells (in preparation). Our results accord to reports that rec-eCG was exhibited biologically activity (Min et al., 1996, 2004; Park et al., 2009, 2010). Taken together, these results indicated that rec-eCG produced from mammalian cells could be useful for improving the development of dominant and pre-ovulation follicles in non-equids.
We also stably transfected CHO-K1 cells with rFSHR and rLH/CGR cDNAs. In previous studies, rec-eCGβ/α was shown to have dual activity of both rFSHR and rLH/ CGR in not-equids, but only LH-like activity in equid species (Park et al., 2009; 2010). However, there have been no reports on the internalization of rec-eCGs in rFSHR and rLH/CGR. Our results demonstrated that receptors of rFSH and rLH/CG were internalized in a time- and concentration-dependent manner by rec-eCGs. However, when deglycosylated at the Asn56 of α-subunit, rec-eCG did not induce the internalization of those receptors. Our results support the data from a previous study that showed that the internalization and degradation of chemically deglycosylated hCG (dghCG) was slower than that of hCG in MA-10 cells, and that this effect was not due to its inability to stimulate cAMP production (Hoelscher et al., 1991). In fact, dghCG was not capable of uncoupling the LH/CGR in Leydig tumor cells (Rebois and Fishman, 1983). In contrast to hCG, addition of dghCG to 32P-labeled 293L cells resulted in little or no increase in the phosphorylation of LH/CG receptors (Hipkin et al., 1993). Based on the data presented, we suggested that the rate of the mutant rec-eCG, which was deglycosylated at Asn56 of the α-subunit, was not increased because of the conformation of the hormone-receptor complex. A previous study suggested that four serine residues in the C-terminal tail of rLH/CGR had distinct structural requirements for agonist-induced uncoupling and agonist-induced internalization (Lazari et al., 1998). A constitutive active mutant of hLH/CGR, hLH/CGR- L457R, was not routed to the lysosomes, but was recycled to the cell; hormone degradation was barely detectable (Galet and Ascoli, 2006). Similarly, the removal or simultaneous mutation of two residues (Cys699 and Leu683) rerouted internalized hCG to a degradation pathway and internalized hLHR to the lysosomes (Galet et al., 2004). Replacement of all of the Ser/Thr residues in the carboxyl-terminal tail and truncation of the domain containing these residues completely inhibited vasoactive intestinal polypeptide (VIP)-stimulated phosphorylation and receptor internalization (Langlet et al., 2005). This GPCR internalization has been shown to be agonist-specific and G protein regulated kinase (GRK)-dependent (Hossain et al., 2009). This type of feedback regulation is conventional, as it requires the activation of classic G proteins (Kohout and Lefkowitz, 2003; Lodowski et al., 2003) as shown in Fig. 6. In the presented study, we showed that deglycosylated rec-eCG did not induce agonist-stimulated internalization. Therefore, we suggested that the glycosylation site in rec-eCG played a pivotal role in the internalization of rFSHR and rLH/CGR. The established stable cells expressing rFSHR and rLH/CGR could be utilize agonist-stimulated receptor internalization when stimulated with rec-eCG mutants that were deglycosylated at other sites. Further studies are required to conclusively determine whether internalization is indeed responsible for the uncoupling of rFSHR and rLH/CGR. These activity will be analyze the oocyte ovulation in vivo using mice.