INTRODUCTION
Generally, first resumption and completion of meiosis in mammalian oocytes are essential process for capacity of fertilization and developmental competence after oocytes maturation (Eppig, 1996). In addition, nuclear and cytoplasmic maturation of oocytes are involved with activation of oocyte, fertilization process, and embryo development before implantation (Eppig et al., 1994), and it require sufficient nutrients from follicular fluid and cells (Steeves & Gardner, 1999; Sutton-McDowall et al., 2005). Moreover, a variety of growth factors and hormones that are secreted from follicular cells stimulated both of nuclear and cytoplasmic maturation of oocytes (Mattioli et al., 1988; Procházka et al., 2000).
For successful oocyte maturation and subsequent development, cellular organelles in cytoplasm of oocytes undergo a physiological changes during maturation. Especially, the Golgi apparatus and endoplasmic reticulum (ER) are closely related with cytoplasmic and nuclear maturation. The Golgi apparatus, also known as Golgi complex, is involved with various intracellular trafficking events and ER plays role in protein folding, degradation, and store of calcium ion (Mao et al., 2014). These organelles of oocytes are redistributed in cytoplasm of oocytes and undergone the structural changes during maturation. And interaction between Golgi apparatus and ER is an important for both of cytoplasmic and nuclear maturation. Moreno et al. (2002) had reported that treatment of brefeldin A, inhibitor of membrane trafficking, during in vitro maturation of mouse oocyte blocked nuclear maturation and this study indicated that membrane trafficking from ER to the Golgi apparatus is closely associated with oocyte maturation. As a one of transport protein, transport protein Sec61 subunit beta (Sec61β) is an important factor of the protein translocation of ER membrane and coatomer protein complex subunit gamma 2 (COPG2), a one of type-1 COP, is involved with retrograde transport of protein from Golgi apparatus to ER (Mendis et al., 2008). These reports suggested that Sec61β and COPG2 are concerned with oocyte maturation through regulation of protein transport between Golgi apparatus and ER, however, roles of transport protein on oocyte maturation were not fully understood.
As a one of growth factor, epidermal growth factor (EGF) plays an important role in cell growth, proliferation, and differentiation by stimulation of mitosis in various type of cells (Yarden, 2001). And it also induces resumption of meiosis in mammalian oocytes (Bolamba et al., 2006; Lindbloom et al., 2008). This growth factor was contained in mammalian follicular fluids including pigs (Conti et al., 2006). Uhm et al. (2010) had reported that nuclear maturation of porcine oocytes were stimulated by action of EGF instead of follicle-stimulating hormone (FSH). Stimulatory effects of EGF on cellular process were generated by binding into its specific receptor (EGFR) and interaction between EGF and EGFR induced intra-cellular signal transduction, including mitogen-activated protein kinase (MAPK) and extracellular signal-regulated kinase (ERK)(Filardo et al., 2002). Because a variety of signal pathways are related with oocyte maturation, biolo-gical function of EGF is an important for successful matu-ration of mammalian oocytes. Therefore, we expected that EGF stimulates porcine oocyte maturation via regulation of transport protein, and the aim of this study was to investigate stimulatory effect of EGF on nuclear maturation and expression of Sec61β and COPG2 in porcine oocytes.
Materials and Methods
All procedures that involved the use of animals were approved by the Kangwon National University Institutional Animal Care and Use Committee (KIACUC-09-0139). Porcine ovaries were collected from slaughtered gilts at local slaughter house and transferred to the laboratory in 0.9% (w/v) sterilized saline within 2 h. The cumulus- oocyte complexes (COCs) were aspirated from antral follicles with 3-6 mm in diameter using 10 cc syringe with 18-gauge needle. After aspiration, COCs with homogeneous cytoplasm and compact cumulus layer were selected. To investigate effect of EGF on nuclear maturation of porcine oocyte, COCs were maturated in medium-199 (Invitrogen, MA, USA) containing 10% (v/v) porcine follicular fluid (pFF), 10 IU/mL human chorionic gonadotropin (hCG; Intervet), 10 ng/mL luteinizing hormone (LH; Sigma-Aldrich, St. Louis, MO, USA), 10 ng/mL follicle stimulating hormone (FSH; Sigma-Aldrich) with or without 10 ng/mL EGF (Sigma-Aldrich) at 38.5℃, 5% CO2 condition for 22 h (IVM-Ⅰ) and subsequently maturated using hormone-free medium-199 containing 10% (v/v) pFF with or without 10 ng/mL EGF for 22 h (IVM-Ⅱ).
Aceto-orcein stain method was used to evaluate nuclear maturation stage of porcine oocyte. Incubated COCs were treated by 0.1% (v/v) hyaluronidase. Cumulus cells surrounding zona pellucida of oocyte were removed by gentle pipetting using pasteur pipette. Denuded oocytes were fixed in acetic alcohol solution (acetic acid : ethanol; 1 : 3; v/v) for 48 h, at room temperature (RT), and nuclear was stained by 1% (w/v) aceto-orecein at RT for 7 min. The morphology of nuclear was observed under light microscope. The oocytes at germinal vesicle (GV), germinal vesicle breakdown (GVBD), metaphaseⅠ(MⅠ), and telophaseⅠ(TⅠ) phase were decided as immature oocyte and MⅡ stage oocytes were classified as mature oocyte.
Mature oocytes, which were incubated with or without EGF during IVM stage, were denuded and fixed in 4% (v/v) paraformaldehyde solution at 4℃ for 1 h. For permeabilization of oocytes, PBS containing 0.5% (v/v) Triton X-100 (Sigma-Aldrich) was used. Then, blocking process of oocytes was carried out using blocking solution [PBS containing 0.1 M glycine, 0.01% (v/v) Triton X-100, 1% (w/v) non-fat dry milk, 0.5% (w/v) bovine serum albumin, and 0.02% (w/v) sodium azide] at RT during overnight. Immunofluorescence was conducted using goat anti-COPG2 (sc-23204, 1:500, Santa Cruz, USA), EGFR (sc-31156, 1:1000, Santa Cruz), rabbit anti-Sec61β (PA3-015, 1:1000, Thermo Fisher scientific, Waltham, MA, USA), and GM130/GOLGA2 (maker of Golgi apparatus, NB110-57012, 1:100, Novus Biologicals, Littleton, CO, USA) at 4℃ during overnight and oocytes were incubated with goat anti-rabbit IgG (H+L)-Alexa Fluor 546 (A11010, 1:1,000, Invitrogen) and donkey anti-goat IgG (H+L)-Alexa Fluor 488 at 4℃ during overnight. The nuclear was counterstained by 1 ng/mL Hoechst 33342. For measurement of fluorescent intensity, immunofluorescence was conducted to 3 replication (40-50 oocytes per replication) and fluorescence images were captured using confocal laser scanning microscope (Olympus LX70 FV00 05-LPG-193, OLYMPUS, Japan) at same exposure time and photo multiplier tube (PMT). Fluorescence intensity was ana-lyzed by Multi gauge software ver 3.0 (Fuji photo Film, Japan).
All numerical data representing each parameter were analyzed using the Statistical Analysis System Software (SAS, version 9.3). Data are presented means ± SEM and Duncan’s multiple range tests. Comparisons among treatment groups were conducted using a generalized linear model (GLM) in the SAS package. A value of P<0.05 was considered to indicate a statistically significant difference.
Results
The effect of EGF on nuclear maturation of porcine oocytes during IVM were presented in Table 1. Treatment of EGF during both of IVM stage enhanced the nuclear maturation rate of oocyte compare with other groups (P<0.05). However, the portions of matured oocytes were lower in treatment groups that are absence of EGF during IVMⅠor IVMⅡstage. And it was lowest in EGF absence group among the all treatment groups (P<0.05). In non-EGF stimulation group, immature oocytes were arrested mainly at GV and GVBD stage, especially, proportion of GV stage oocytes were higher than EGF-treated group during both IVM stage.
The data are present as mean ± SEM from 3 replicates.
a Immature oocytes include germinal vesicle, germinal vesicle breakdown, metaphase, and telophaseⅠphase oocytes.
b Mature oocytes include only metaphaseⅡ oocytes.
c-e Different superscript in the same column indicate significant difference (P<0.05).
* A period that is first 22 hour at in vitro maturation (IVM).
** Subsequent maturation stage for 22 hour after IVMⅠ.
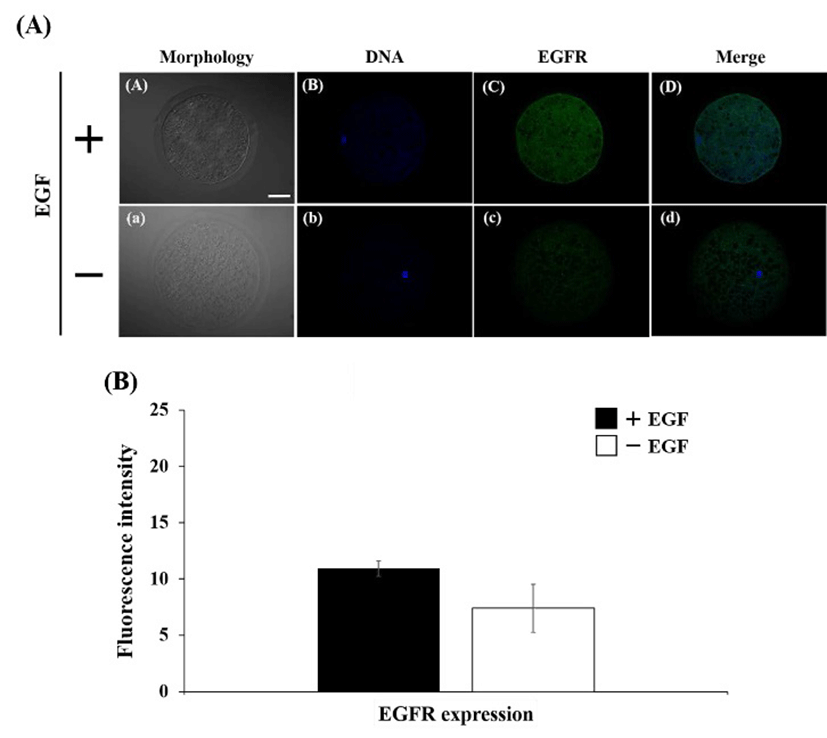
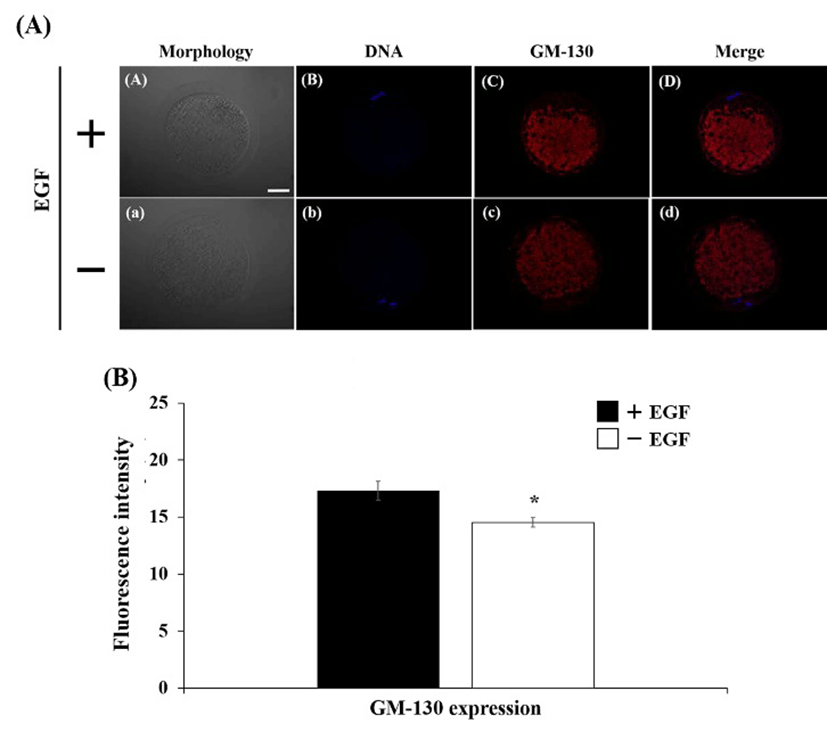
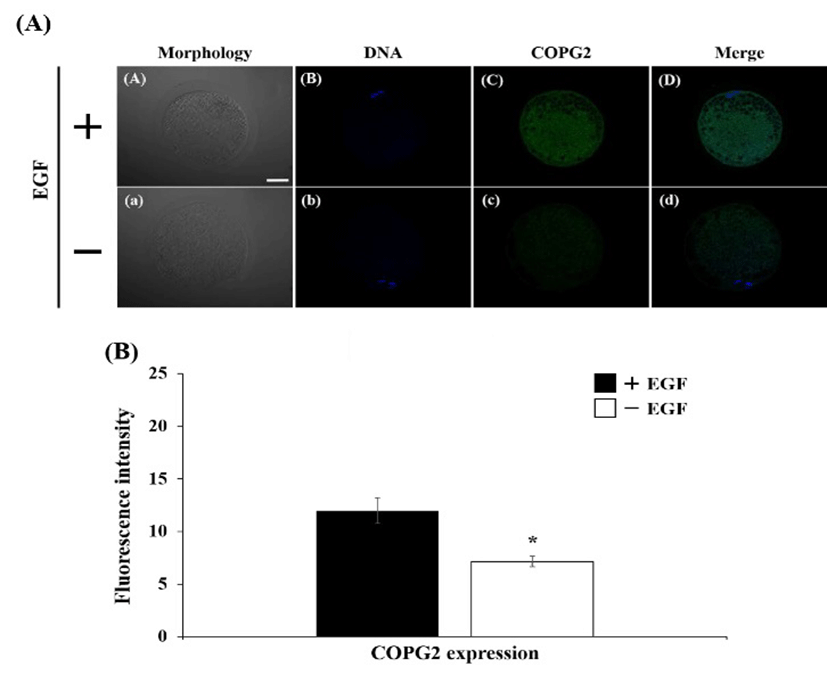
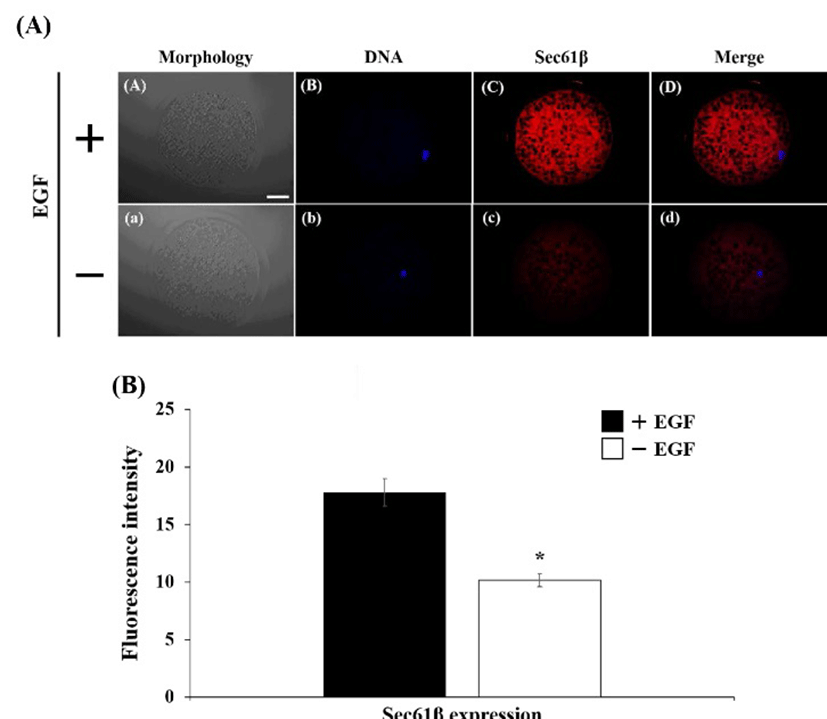
The expression of EGFR was detected on cytoplasm in both of EGF-treated and EGF non-treated oocyte (Fig. 1A). Fluorescence intensity in EGF treatment group was higher than non-treatment group, however, there was no significant difference (Fig. 1B). The Golgi apparatus was placed mainly in cytoplasm, whereas it was not located in surrounding chromosome region (Fig 2A). The expression level of the Golgi was stimulated by EGF treatment during maturation compare with non-EGF group (Fig. 2B, P< 0.05). Similar with EGFR expression, the COPG2 protein was detected on cortical area and cytoplasm (Fig. 3A). And fluorescent intensity of this protein was enhanced by EGF stimulation as well as Golgi apparatus expression (Fig. 3B, P<0.05). Sec61β in both of treated and non-treated oocyte was distributed in cytoplasm and fluorescence intensity was increased in EGF-treated oocyte (Fig. 4, P<0.05).
Discussion
This study was conducted to confirm the change of nuclear maturation of porcine oocytes by stimulation of EGF during IVM and we expected that it was mediated transport protein including COPG2 and Sec61β. Nuclear maturational rate was enhanced by EGF treatment during both IVM stage and expression of Golgi apparatus, COPG2, and Sec61β were up-regulated in cytoplasm by EGF stimulation.
Signal transduction by interaction between EGF and its specific receptor plays a crucial role in maturation process of mammalian oocytes. EGF and EGF-like ligands, such as amphoregulin, epiregulin, and transforming growth factor-α, shared and bind to the EGFR (Shimada et al., 2006). And these action improved the maturation of oocytes in various species including cows (Lonergan et al., 1996), mice (De La Fuente et al., 1999), monkeys (De Prada et al., 2009), and pigs (Coskun & Lin, 1995). Coskun and Lin (1995) had reported that stimulation of porcine oocyte maturation by EGF is independent of the cyclic AMP (cAMP) and it might be related with protein kinase C (PKC) pathway. In this present study, nuclear maturation of porcine oocytes were enhanced by EGF stimulation at IVM stage, and expression of EGFR was increased in cytoplasm of EGF-treated oocytes. In the goat COCs, EGFR was localized on both of cumulus cells and oocyte, and expression of protein was higher in mature oocytes than immature oocytes (Gall et al., 2004). Also, oocyte maturation was mediated by phosphorylation of MAPK through action of EGF and EGFR (Gall et al., 2005). Therefore, porcine oocyte maturation might be stimulated by a variety of signal pathway that are induced by action of EGF. And action of EGF might influence to change of organelle in oocyte such as Golgi apparatus and ER.
During process of oocyte maturation, the Golgi apparatus undergoes fragmentation and it is accumulated in the central part of the oocyte as dotted structure (Moreno et al., 2002). Moreover, Payne and Schatten (2003) had reported that the Golgi apparatus was translocalized into the ER during oocyte maturation of cow. Although the function of Golgi apparatus on oocyte maturation had been reported, regulation of this organelle by growth factors is not fully understood. In this results, fluorescence intensity of GM-130, a marker of Golgi apparatus, was significantly higher in EGF-treated oocytes than EGF non-treated oocytes. These result show that expression of Golgi was stimulated by action of EGF, however, regulatory mechanism of Golgi apparatus by EGF stimulation during oocyte maturation is required more studies.
Sec61 complexes are transport protein that act as ER membrane protein translocator. And they are divided into three type subunit: alpha, beta, and gamma (Samuelson et al., 2000). Greenfield and High (1999) had reported that Sec61 complexes were located in both of the ER and ER-Golgi intermediate compartment in COS1 cells, which are African green monkey kidney SV40 transformed cell line. And they suggested that Sec61 complexes were maintained in the ER membrane by a retrieval mechanism. These report indicated that Sec61 proteins play a potential role in transport between the ER and Golgi apparatus. In results of present study, expression of Sec61β protein was increased by EGF stimulation and localized in cytoplasm as well as GM-130 and EGFR expression. The relationship between
Sec61β and oocyte maturation was not reported. However, Sec61 complexes play a role in transport between Golgi apparatus and the ER that is closely related with oocyte maturation. Wang et al. (2010) reported that EGFR level in the nucleoplasm was reduced by knocking-down of Sec-61β. These reports suggest that Sec61β protein might influence to oocyte maturation via regulation of EGFR expression and mediation of transport between Golgi apparatus and the ER.
The relationship between expression of COPG2 and oocyte maturation was not reported as well as Sec61β protein. Cui et al. (2007) reported that expression of COPG2 mRNA was higher in 4-cell stage embryo and blastocyst than morula embryo. In in vitro produced bovine embryos, COPG2 mRNA was detected and it was expected that this gene function in embryonic developmental processes (Ripamonte et al., 2011). These effect of COPG2 on embryo development might be attributed by transport from Golgi apparatus to ER. Because transport between Golgi apparatus and ER is an important factor during oocyte maturation, COPG2 protein might be expected to play an important role in oocyte maturation processes. Despite of several studies associated with protein expression of ER/Golgi transport protein during oocyte maturation, transcriptional regulation of transport proteins were not reported.
In conclusion, we found that nuclear maturation of porcine oocytes were stimulated by EGF and stimulatory effect of EGF regulated expression of Golgi apparatus, Sec61β, and COPG2. Especially, EGFR expression was down-regulated by absence of EGF stimulation and it might be related with expression of Sec61β protein that was regulated by EGF. This present study suggests that stimulatory effect of EGF is an important factor in porcine oocyte maturation and stimulation by EGF in oocytes could influenced not only intracellular signal transduction but also interaction between Golgi apparatus and ER that in mediated by transport protein.

