INTRODUCTION
Cytochrome P4501A (CYP1A), a drug metabolizing enzyme found in most vertebrates, has been known to be inducible by intrinsic and extrinsic factors. CYP1A reacts specifically to environmental pollutants including polycyclic aromatic hydrocarbons (PAHs) to solubilize them to excrete from the body, and some metabolic intermediates of the environmental pollutants have been proven to be carcinogenic. PAHs induce CYP1A gene after consecutive binding to cytosolic aryl hydrocarbon receptor (AhR) and AhR nuclear translocator (ARNT) in nuclear. On the other hand, vitellogenin (Vg) which is utilized as a nutriment during embryonic period of an oviparous animal is induced in the liver by estrogen (E2) and endocrine disruptors. Therefore the gene expression of CYP1A and Vg have been used as a biomarker for aquatic pollutants (Goksoyr, 1995; Marin & Matozzo, 2004; Sarkar et al., 2006).
Benzo[a]pyrene (B[a]P), a carcinogenic substance of PAHs, has been reported to induce CYP1A activity in many fish species (Padros et al., 2003; Elskus, 2004). In addition, tributyltin (TBT), an aquatic pollutant as an organic tin compound, has been used as an antifoulant of under water part of ship and marine structures to prevent corrosion and adhesion of shellfish (Evans et al., 2000). Since the beginning of 2000's TBT has been banned on use in most of developed countries. However, large amounts of TBT has been found in fishes and sea-floor sediments near shipyards (Fent et al., 1996; Michel et al., 2001; Birchenough et al., 2002; Brian et al., 2003; Gopalakrishnan et al., 2011). TBT is classified as an endocrine disruptor. It induces imposex in gastropoda such as spiral shell fish and turbo, which brings about rapid decline in their population (Ellis et al., 1990; Shim et al., 2000; Santos et al., 2002). TBT has been found to effect on breeding disturbance and sexual transformation of fish to male (McAlister & Kime, 2003; Shimasaki et al., 2003; Nakayama et al., 2004). TBT has also been reported to control the hepatic expression of estrogen receptor (ER), Vg and zona radiata of Atlantic salmon (Mortensen & Arukwe, 2007). In the same study the expression of AhR, ARNT and AhR repressor (AhRR) was different as the TBT concentration. In addition, decrease in CYP1A activity was reported in channel catfish and Atlantic salmon (Rice & Roszell, 1998; Mortensen & Arukwe, 2007).
Although numerous studies on the effects of environmental pollutants on the activities of CYP1A and its isomers have been reported, researches on the effects of TBT, one of the most dangerous aquatic pollutants, on the expression of CYP1A, AhR and Vg genes have been limited to a few fish species. Therefore, in the present study the expressions of CYP1A, AhR and Vg genes as affected by TBT treatment were investigated using cultured eel hepatocytes.
MATERIALS AND METHODS
Eels (Anguilla japonica) weighing 250–300 g were purchased from a local hatchery and kept in plastic tank containing 25°C fresh water. They were not fed during experimental period, and immaturity of genital glands was confirmed by dissection at the end of experiment.
Benzo[α]pyrene, estradiol-17β and tributyltin chloride (TBT) were purchased from Sigma Chemical Co (St Louis, MO USA). They were dissolved in DMSO before injection to culture media.
For in vivo induction of CYP1A and vitellogenin eels were given single intraperitoneal injections of Benzo [α]pyrene (10 mg/kg BW) or estradiol-17β (10 mg/kg BW) dissolved in DMSO. Fish were sampled after 2 days the injection and hepatocytes were isolated as described below.
Hepatocytes were prepared according to the procedure of Kwon & Mugiya (1994). Eels were anesthetized with 0.1% 2-penoxyethanol. The liver was exposed and perfused in situ with Ca2+-free Ringer solution (120 mM NaCl, 4.7 mM KCl, 1.25 mM KH2PO4 and 23 mM NaHCO3, pH 7.4) via the hepatic portal vein for 10 min. The perfusion was followed with the Ringer sloutin containing collagenase (0.35 mg/ml, Type IV, Sigma) at room temperature (ca 25°C) for 25 min. The liver was finally perfused with 30 ml of Ca2+-and Mg2+-free Ringer solution containing 2 mM EDTA. After perfusion, the liver was freed from the gall bladder and transferred to a beaker containing the Ca2+-free Ringer solution (30 ml). It was minced with scissors and pipetted gently. The cell suspension was filtered with a nylon gauze into a 50 ml conical tube and the filtrate was resuspended with Ca2+-free Ringer solution and washed three times by centrifugation. Then, the cell precipitate was resuspended in a small volume of the Ringer solution. Cell yield and viability were determined by the Trypan Blue exclusion test. Cells were plated into a 60 mm Petri dish (Falcon) at a density of 3×105 cells/dish. L-15 medium (Sigma) containing 0.2 μM bovine insulin (Sigma), streptomycin (100 μg/ml), and penicillin (70 μg/ml) was used for cell culture. All incubations were carried out in 3 ml of the medium at 25°C under 5% CO2. The media containing the chemicals were changed every day. All incubations were replicated at least twice.
Hepatocytes were homogenized with 0.5 ml Trizol reagent (Invitrogen Inc.) on ice and incubated for 15 min at room temperature. After centrifugation (12,000×g, 20 min, 4°C) to remove cell debris, 140 μl of chloroform was added to supernatant. RNA from aqueous phase was precipitated by mixing with isopropanol and incubating samples on ice for 2 hr. After centrifugation (12,000×g, 15 min, 4°C), RNA pellet was washed with 75% ethanol and air dried. RNA dissolved in nuclease-free water was determined for concentration and purity using GeneQuant (Biochrom Ltd., Cambridge, England). RNA samples were stored at –80°C before use.
cDNA was synthesized from the isolated total RNA using Oligo(dT)15 primer (Promega) and M-MLV reverse transcription (Promega). Primers targeting CYP1A, Vg, AhR and GAPDH genes (Choi, 2012) were used in RT-PCR as shown in Table 1. Constructed cDNA was amplified with Go Taq Green Master Mix (Promega) and 0.2 M of each PCR primer. PCR μ procedure included one cycle of 2 min at 95°C, 30 cycles of 40 sec at 95°C, 40 sec at 65°C, 40 sec at 72°C and a final extension step of 7 min at 72°C by using a thermal cycler (Thermocycler, Biometra). The PCR product was separated on an 1% agarose gel (in TBE buffer, 30 μg/ml ethidium bromide solution) and visualized using Image Analysis System (Kodak).
RESULTS AND DISCUSSION
Changes in gene expressions of CYP1A and AhR by B[a]P (10-6-10-5 M) and TBT (10-9-10-5 M) was investigated using RT-PCR in cultured eel hepatocytes which was treated previously in vivo with B[a]P to induce CYP1A and AhR genes. As shown on Fig. 1, the expression of CYP1A gene by B[a]P was increased in a concentration-dependent manner as compared with DMSO control (p<0.05). Gene expression of CYP1A by B[a]P, which has been reported in olive flounder (Paralichthys olivaceus) and rainbow trout (Oncorhynchus mykiss) (Malmström et al., 2004), was used as a control for studies on changes in CYP1A expression by TBT. When eel hepatocytes were treated with TBT (10-9-10-5 M), decrease in CYP1A was observed at high concentration of TBT (10-6 M and 10-5 M) while showing no changes at low concentration (10-9-10-7 M) (p<0.05). Changes in the expression of CYP1A by B[a]P and TBT were not observed to be different between 24 hr and 48 hr of incubation (data not shown). These results indicate TBT to act as a suppressor to CYP1A expression in cultured eel hepatocytes. In an in vivo study with Atantic salmon (Salmo salar) the expression of CYP1A was suppressed with high concentration of TBT and stimulated with low concentration (Mortensen & Arukwe, 2007). Meanwhile, in the present study with eel hepatocytes the expression of CYP1A was suppressed with high concentration of TBT as in Atlantic salmon, while showing no differences from control at low concentration.
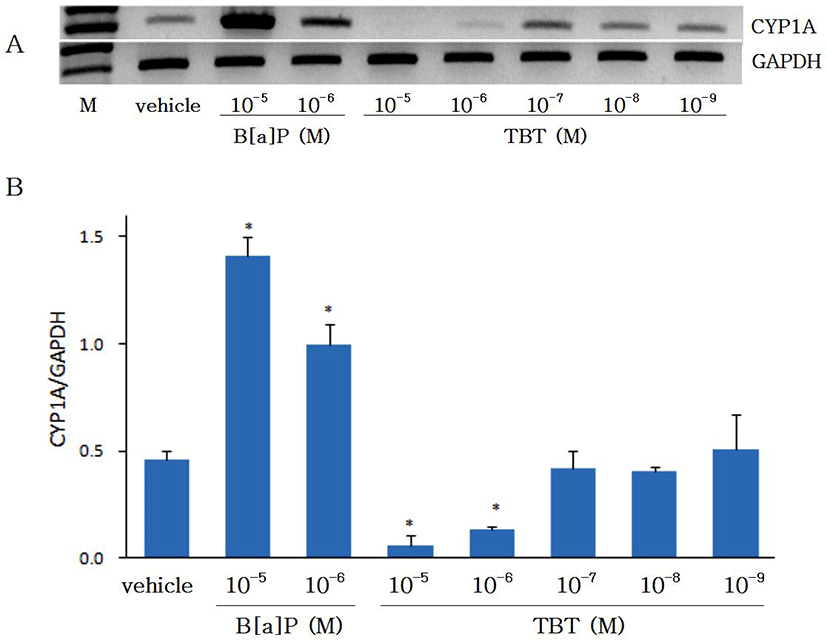
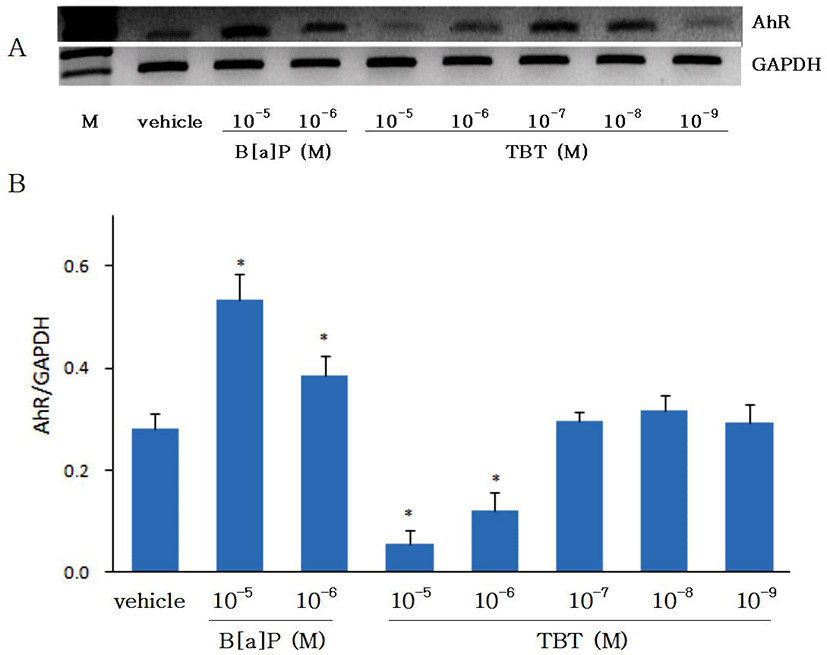
Effects of B[a]P and TBT on gene expression of AhR are shown on Fig. 2. Gene expression of AhR was increased with B[a]P. As CYP1A has been known to be induced by xenobiotics such as PAHs including B[a]P which binds to the AhR and thereby induce the CYP1A (Hahn, 1998), in the present study with eel hepatocytes the gene expressions of both CYP1A and AhR were shown to be increased by B[a]P. The gene expression of AhR was significantly suppressed (p<0.05) by TBT at 10-6 M and 10-5 M, but at 10-9-10-7 M it did not show any differences from control group. The expression of AhR at 24 hr was not different from 48 hr of incubation (data not shown). In an in vivo study with Atlantic salmon, although hepatic mRNA expression of AhR was not significantly changed by treatment of TBT, it was higher in fishes treated with low concentration of TBT (50 ul/L) than in fishes treated with high concentration of TBT (250 ul/L) (Mortensen & Arukwe, 2007). The results on the control of AhR gene expression by TBT in the present study with eel hepatocytes are not in accord with the in vivo study with Atlantic salmon. Further studies in other fish species are needed to prove whether the discordance was caused by in vivo and in vitro difference or species difference.
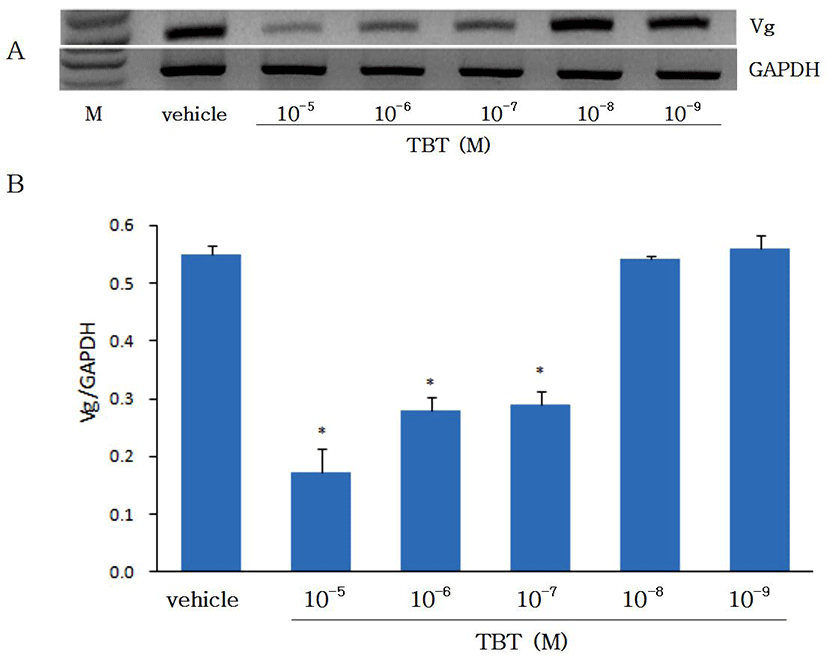
Effects of TBT on vitellogenin (Vg) gene expression were studied in cultured eel hepatocytes. Hepatocytes which were isolated from eels treated previously with estradiol-17β (E2) in vivo were cultured for 4 days with TBT. Differences in the Vg mRNA level after TBT treatment were estimated by RT-PCR (Fig. 3). At both 2 and 4 days of culture the expression of Vg mRNA in hepatocytes treated with 10-7-10-5 M TBT was significantly decreased compared with control cells, while when treated with low concentration (10-9-10-8 M) no difference in Vg expression was observed. Endocrine disruptors acting like E2 have been known to increase Vg gene expression (Jung et al., 2012). However, despite the TBT is classified as an endocrine disruptor it suppressed the expression of Vg gene in the present study. The suppression of Vg gene expression by TBT in this study is coincide well with the in vivo result with Atlantic salmon (Mortensen & Arukwe, 2007). Although Vg gene has been known to be expressed through activation of estrogen receptor (ER) in the presence of E2, activation of ER with TBT has been shown to be different between ER isomers. The expression of ER was α elevated, while ERβ decreased (Mortensen & Arukwe, 2007). Therefore, whether the suppression of Vg expression with TBT was through ER is not clear. As the present study with eel hepatocytes did not prove the point, more indept studies on the expression of Vg and ER are needed.
In summary, the gene expressions of both AhR and CYP1A were shown to be suppressed by TBT, and the suppression of CYP1A was thought to be through AhR. Expression of Vg gene was also observed to be suppressed by TBT. Therefore, as in the present results, the expressions of CYP1A, AhR and Vg in eel hepatocytes would be suggested to be valuable tools for detecting and studying endocrine disruptors and environmental pollutants such as PAHs, PHHs and PCB.

