INTRODUCTION
The sex maturation of fish is controlled by reproductive endocrine system in the Brain-Pituitary-Gonad axis (BPG axis) (Davies et al., 1999; Weltzien et al., 2004; Zohar et al., 2010). The hypothalamus secretes GnRH, and these regulates secretion and synthesis of GTH in the pituitary. The regulators of BPG axis are the gonadotropins, the FSH and LH, these induces the synthesis of sex steroids in the gonad (Pankhurst, 1998). In females, FSH interacts with premature ovarian and stimulates oocyte growth, inducing LH is known for final gonadal maturation and ovulation. The reproductive endocrine system interacts with various environmental factors, such as photoperiod and water temperature. Particularly, photoperiod and water temperature are the major environmental factors to control sex maturation and the spawning period. Thus, recent research to control sex maturation of fish is more studied by investigating the relationship between these environmental factors and the endocrine system (Norberg et al., 2001; Schulz et al., 2006; Hala et al., 2011).
Olive flounder, Paralichthys olivaceus is potentially important and economically valuable aquaculture fish in Korea. Natural spawning takes place between February and June with March through May being the peak spawning season. Based on the flounder’s spawning season, the optimal environmental conditions for maturation and spawning have been studied and the results from artificial conditions to induce spawning were reported (Min, 1988; Hur, 1991). Although information on the reproductive biology in olive flounder have been compiled, the physiological information on controlling the gonadal maturation and spawning period by the environmental factors under artificial conditions is insufficient. Therefore, the present study investigated the effects of photoperiod on gonadal activity of the female olive flounder at resting season (September) in Jeju Island, South Korea. In addition, RT-PCR assays mRNA expression of GTH subunits (FSHβ, LHβ) in the pituitary gland under the artificial condition where photoperiod is controlled.
MATERIALS AND METHOD
Olive flounder (body weight 1,843–1,921 g, total length 54.8–55.4 cm) being reared at Wimi Fishery Co. Ltd., located in Seogwipo, Jeju, South Korea, were examined. The experimental fish were divided into two groups; the control group under a condition with natural photoperiod and the treatment group under an artificial condition with controlled photoperiod (15L: 9D). They were reared for approximately seven months from September 2011 to March 2012. Five fish from each group were randomly selected to compare gonadal development and GTH expression under different conditions for three times; one in September 2011, another in November 2011, and the third in March 2012. The experimental fish were anesthetized with 2-phenoxyethanol (Sigma Co.), and then gonads and pituitary gland were extracted. The extracted gonad of each specimen were weighted for calculation of the gonadosomatic index (GSI=gonad weight ×100/body weight). For the histological analysis, gonad samples were fixed in Bouin’s solution, embedded in paraffin, sectioned 5 μm thickness and stained with haematoxylin-eosin. The extracted pituitary samples were kept at –80°C until analysis.
The total amount of RNA from the pituitary samples was extracted using RNAiso Reagent (TaKaRa Bio, Kyoto, Japan). The total RNA was treated with RQ RNase-free DNase (Promega, Madison, WI, USA) to prevent genomic DNA contamination. The purity of the total extracted RNA from the pituitary was examined with the ratio of 1.7 and 2.1 for A260/A280. From 1 μg of the extracted total RNA, the reverse transcription reaction was performed to synthesize cDNA using the Transcriptor First strand cDNA Synthesis kit (Roche Diagnostics, Mannheim, Germany).
Primers for the experiments were produced by FSHβ (Gene bank, accession number AB042422) and LHβ (AB042423) of P. olivaceus that are listed on NCBI (Table 1). A qPCR was conducted with 20 ng of cDNA using SYBR green premix PCR kit (Takara) in CFX96™ Real-time System (Bio-Rad, Hercules, CA, USA). PCR was performed at 95°C after the initial denaturation. Afterwards, the PCR reaction was performed by 40 cycle of denaturation for 45 s at 94°C, annealing for 45 s at 58°C, and extension for 1 min at 72°C. Expression of the FSHβ and LHβ genes in each sample was normalized to the amount of the internal control β-actin gene.
RESULTS
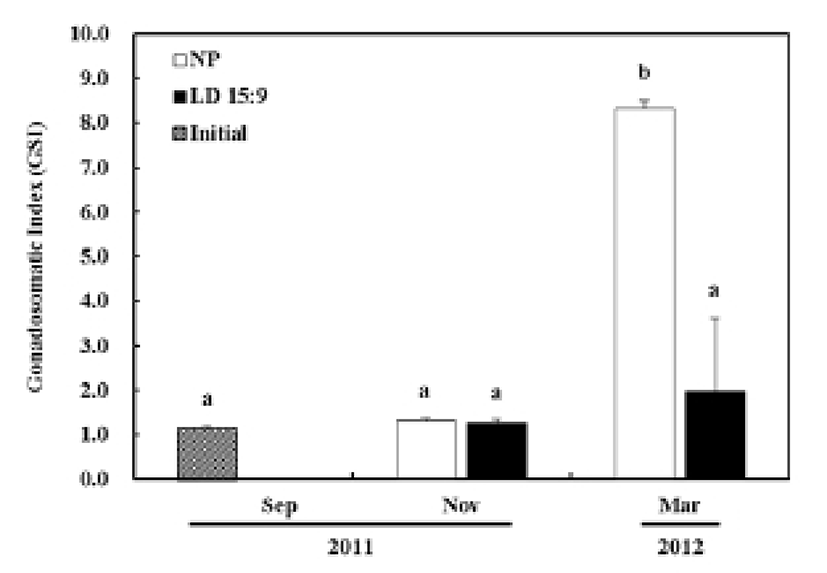
The GSI of olive flounder at the onset to the experiment (September 2011) was 1.2 ± 0.1. In the control group under the natural photoperiod, the GSI for November was 1.3 ± 0.1 and showed no significant statistical difference from the onset to the experiment. However, the last experiment, in March 2012, presented a drastically increased GSI at 8.3 ± 0.2, showing a significant statistical difference (P<0.05). In the treatment group under an artificial condition with controlled photoperiod (15L:9D), the GSI from the November experiment was 1.3 ± 0.1 and the GSI from the March experiment was 2.0 ± 1.7, representing no significant statistical difference (Fig. 1).
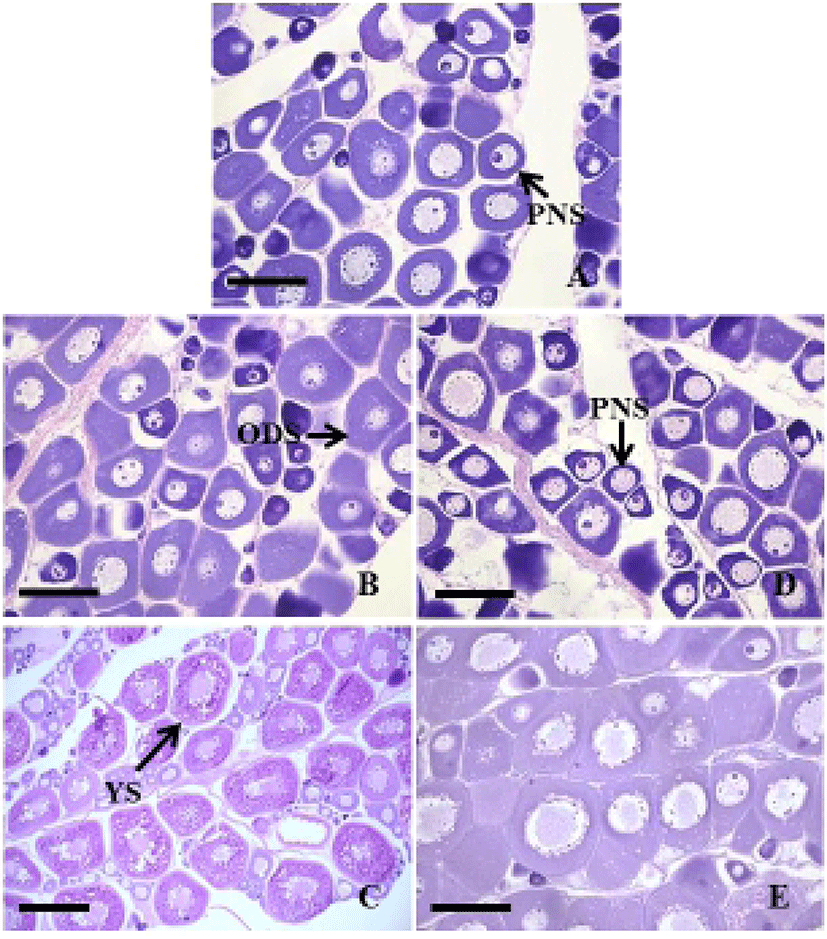
In September 2011, the ovaries were immature, mainly contained perinucleolus stage oocytes of 20 to 50 μm in diameter (Fig. 2A). In the control group, the ovaries from November were growing phase, contained the oil-droplet stage oocytes of 80 to 150 μm in diameter (Fig. 2B). In March 2012, the ovaries became mature as vitellogenic oocytes of 220 to 500 μm in diameter (Fig. 2C). In the treatment group, the ovaries from November were immature, mainly contained perinucleolus stage oocytes (Fig. 2D). In March 2012, the ovaries were still growing phase, contained the oil-droplet stage oocytes (Fig. 2E).
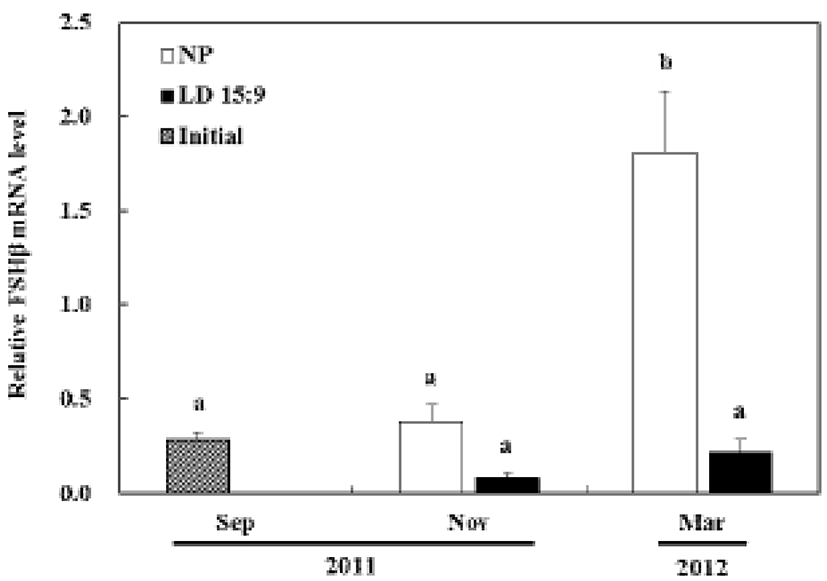
In the control group, the expression of FSHβ mRNA at the November did not show significant statistical difference from the first experiment in September. However, the March experiment showed drastically increased levels of FSHβ mRNA determining significant statistical difference (P<0.05). In the treatment group, the expression of FSHβ mRNA had no significant statistical difference throughout all of the experiments (Fig. 3A).
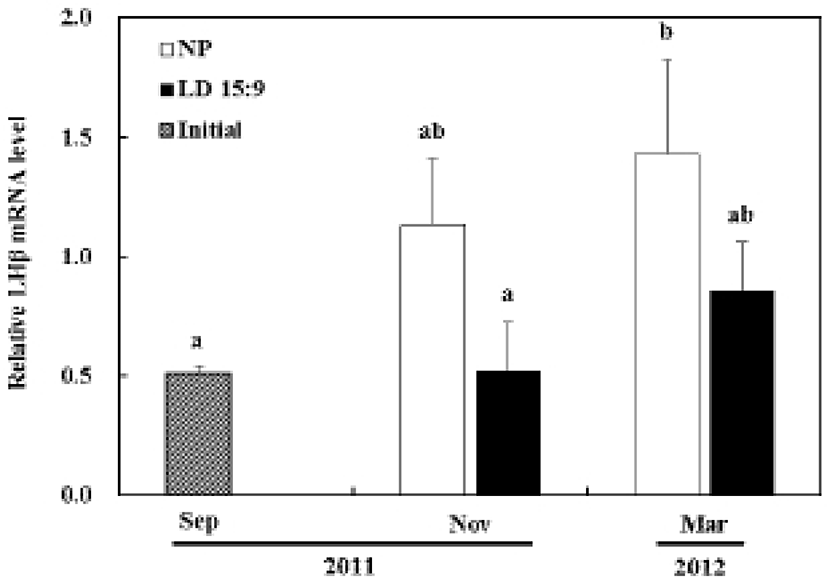
In the control group, the LHβ mRNA, compared with the first experiment, started increasing from November and showed statistical significance by March (P<0.05). However, in the treatment group, the LHβ mRNA had no significant statistical difference in expression until the last experiment (Fig. 3B).
DISCUSSION
The reproductive cycle of fish is species specificity, and photoperiod information and water temperature are known to be important factors for the maturation of gonads and spawning cycle (Huber & Bengtson, 1999; García-López et al., 2006). Based on this, many studies to regulate the reproductive endocrine system and spawning cycle using photoperiod are being performed (Begtashi et al., 2004; Unwin et al., 2005; Abdulfatah et al., 2011; Cowan et al., 2012). As for Nile tilapia, Oreochromis niloticus L., sex maturation occurs during a short photoperiod, when a long photoperiod switches to a short photoperiod. The results from experimental studies under the natural photoperiod and long photoperiod conditions of 24L:0D, 20L:4D, and 18L:6D show that the long photoperiod treated group suppressed gonadal development than the control group. GSI from 24L:0D treatment group was lower than other long photoperiod treatment groups, which did not have gonadal development (Rad et al., 2006). The Atlantic cod (Gadus morhua) under the 24L:0D photoperiod significantly decreased expression of FSHβ and LHβ during the sex maturation period (Almeida et al., 2011). In particular, blood concentration of sex steroid hormones such as testosterone, estradiol-17β and 11-ketotestosterone decreases because gonads were not developed (Cowan et al., 2011). Gilthead seabream, Sparus aurata, a marine species that spawns in the winter, also showed low GSI when treated under long photoperiod of 15.5L: 8.5D until the spawning season (Kissil et al., 2001). In this study, to sex maturation control of olive flounder by regulation of photoperiod, gonadal development and pituitary GTH mRNA expression were investigated. Photoperiod was treated each the different conditions, natural photoperiod and long photoperiod (15L:9D) conditions from September 2011 to March 2012. The results showed that the GSI of a control group under natural photoperiod was 8.3 ± 0.2 and the ovaries contained vitellogenic oocytes in March. However, the GSI of treatment group under long photoperiod was 2.0 ± 1.7 and the ovaries mainly contained peri nucleolus or the oil-droplet stage oocytes in March. Also, FSHβ and LHβ mRNA expression in pituitary decreased more in long photoperiod treatment group than in natural photoperiod. In particular, long photoperiod treatment from September 2011 to March 2012 inhibits the secretion of FSHβ and maintains early growing stage of gonadal development in P. olivaeus.
The GTH of fish shows a different secretion patterns depending on its spawning cycle. In the rainbow trout, Oncorhyncus mykiss, the FSHβ blood concentration increased between the immature and mature stages and that LHβ increased during mature and spawning stages (Breton et al., 1998). Such GTH secretion is regulated by the GnRH in hypothalamus. In the masu salmon (Oncorhynchus masou) under two different conditions of short photoperiod (8L:16D) and long photoperiod (16L:8D), a shot photoperiod treatment induced sex maturation but long photoperiod treatment delayed sex maturation. Also, GnRH mRNA expression increased according to its maturation stage (Amano et al., 1995). Recent studies have discovered new major factors or hormones influencing the neuroendocrine and the reproductive endocrine system. Among the various environmental factors affecting aquatic animals, photoperiodic change is the most important factor in the initiate and/or termination of reproductive activity. For example, melatonin is produced and secreted mainly in the pineal gland, which is a photosensory organ and stimulates various physiological functions such as growth, gametogenic activities, immune responses, and migration (Falcón, 2010). These melatonin secretion rhythm effects various hypothalamic neurotransmitter such as arginine vasotocin, neuropeptide Y, GnRH, kisspeptin, and dopamine (Zohar et al., 2010). Moreover, the synthesis and secretion of hypothalamic GnIH modulate by pineal melatonin, suggestion that GnIH could be implicated in the transduction of photoperiod information to other endocrine system involved in the control of reproduction (Tsutsui et al., 2007).
This study infers that long photoperiodic conditions regulates sex maturation of olive flounder due to the reduced secretion of FSHβ and LHβ mRNA in the pituitary by melatonin secretion rhythm and interaction of dopamine and GnIH in hypothalamus. But, further studies are needed to ascertain the interaction and secretion of hypothalamic neurohormones (kisspeptin, GnRHs) and other reproductive endocrine hormones by various photoperiodic conditions in this species.


