INTRODUCTION
Fish cytotaxonomy refers to the study of phenetic and/or phylogenetic relationships among species based on comparisons of chromosome number and morphology (Gold, 1979; Kim & Park, 1990; Park et al., 1999, 2017). Cytogenetic studies of fish from coastal areas around Korea are limited to saddled weever, Parapercis sexfasciata, marbled rockfish, Sebastiscus marmoratus and marbled sole, Pleuronectes yokohamae (Park & Lee, 1996), and black plaice, Pleuronectes obscurus, gizzard shad, Konosirus punctatus and perch sculpin, Pseudoblennius percoides (Kim et al., 2007).
The chicken grunt, Parapristipoma trilineatum inhabits rocky shores and seasonal offshore-inshore migrations in limited areas (Watanabe& Okazaki, 2000). This species was cultured and released for fishery in Japan (Watanabe & Okazaki, 2000).The small yellow croaker, Larimichthys polyactis has received considerable attention because of its great commercial importance in both Korea and China (Han et al., 2009; Kim et al., 2012a). It is geographically distributed in the Bohai Sea, Yellow Sea and East China Sea (Han et al., 2009; Kim et al., 2012a).The brown croaker, Miichthys miiuy has adapted well to the western coastal waters of Korea, which are characterized by high turbidity resulting from strong tidal currents and low water temperature in winter (Park et al., 2006, 2007a, 2007b; Kim et al., 2012b).
The chicken grunt, small yellow croaker and brown croaker, an important species in fisheries, are thought to be a suitable candidate for use in commercial-scale aquaculture (Choi et al., 2002; Park et al., 2006, 2007a, 2007b; Kim et al., 2012b). Given the commercial importance of 3 species, especially in the aquaculture industry, information on aspects of their biology are of great interest.
Karyotypic study is a part of the analysis of species divergence and evolution (Hartley &Horne, 1984; Park et al., 1995). So the karyotypic analysis of the order Perciformes was useful method of classifying these species exhibiting various sexual color and dimorphism at sexual maturation period, and further more distinguishing sex in each species (Park et al., 1995). Considering that the knowledge of its cytogenetic traits is currently insufficient, the objective of this study is to clarify the cytogenetic features of these species. This report presents data from karyotypic analysis of 3 species of Family Perciformes from Korea.
MATERIALS AND METHODS
We declare that all experiments in this study comply with the current laws of Korea (Ordinance of Agriculture, Food and Fisheries, No.1 - the law regarding experimental animals, No. 9932) and the Ethical Guidelines of Korea Maritime and Ocean University, Korea. The analysis was performed on preparations obtained from specimens of chicken grunt, P. trilimeatum(from Jeju island of Korea), small yellow croaker, L. polyactis(from Yellow Sea), and brown croaker, M. miiuy(from southern water in Korea) raised in the Fishery Genetics and Breeding Sciences Laboratory Aquarium, Korea Maritime and Ocean University, Korea.
Chromosome preparations were obtained from 22 samples of each species (10 females and 12 males weighing 300-400 g) using the standard kidney direct method (Kim et al., 1982; Im et al., 2001; Park et al., 2005). Sex was determined by gonadal inspection. An intraperitoneal injection of 0.05% colchicine (Sigma-Aldrich, St.Louis, Missouri, USA) was given to each specimen (1-10 µg/g body weight) 4 hrs before sacrificing. Kidney tissue was minced in 0.075 M KCl (Sigma-Aldrich, St.Louis, Missouri, USA). The suspensions were centrifuged, discarded, and cell sediments were fixed in three successive changes of fresh methanol-acetic acid solution (3:1). Chromosome slides were made by the conventional air-drying technique. Detailed procedures for the preparation are provided by Im et al. (2001) and Park et al. (2003). The final suspension was dropped on clean dry slides and placed on a 60°C slide warmer. Slides were prepared using routine air-drying method and stained with Giemsa solution (Gurr's R-66, BDH, England).
At least 20 countable metaphases from each specimen were observed using an optical microscope (CH 130, Olympus, Japan) to determine chromosome number and analyze the karyotype. Well-spread chromosomes at metaphase were selected and photographed. Karyotypes were prepared according to the procedures of Levan et al. (1964).
RESULTS AND DISCUSSION
Chromosome number and variability in chromosome number distinguish certain major taxonomic groups of fish (Gold, 1979; Park & Lee, 1996; Park et al., 1995, 1999, 2000, 2017). Chromosome number and karyotype have unique numerical forms in each species and can provide useful data with which to identify species (Gold, 1979). To our knowledge, this is the first report of the karyotypes of chicken grunt, P. trilimeatum, small yellow croaker, L. polyactis, and brown croaker, M. miiuy.
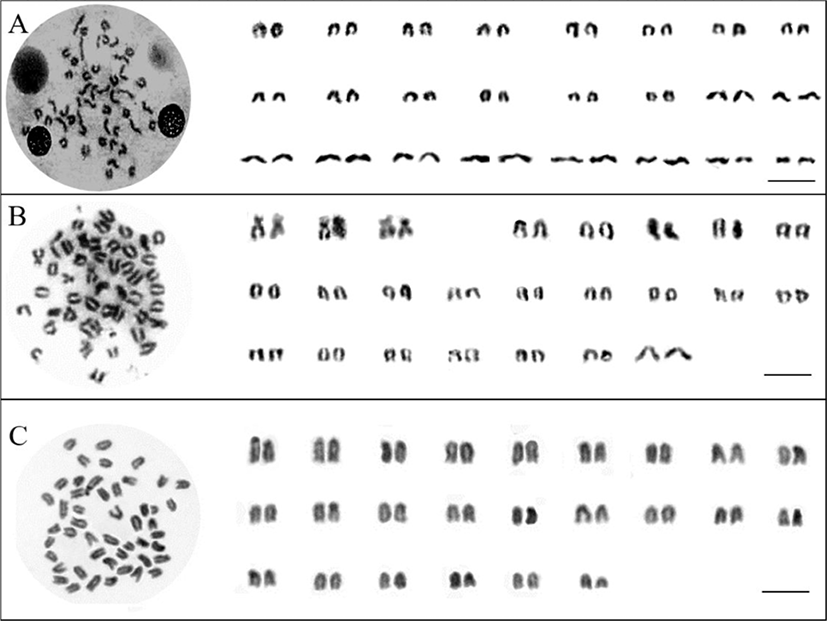
The karyotype and the chromosome distribution frequency of chicken grunt, small yellow croaker, and brown croaker are shown in Table 1 and Fig. 1. The chromosome number of chicken grunt, small yellow croaker and brown croaker was 2n=48, respectively. The fundamental number (FN) of chromosomes is very important when comparing species within a genus and indicates the progenitor type from thich the acrocentric chromosome number increase (Ohno, 1974; Kim et al., 2004). The FN increases with further differentiation of species (Arai, 1983). Chicken grunt and brown croaker consisted of 24 pairs of telocentrics. The FN of chicken grunt and brown croaker was48. The karyotype of small yellow croaker was 3 pairs of submetacentrics and 21 pairs of telocentrics. The FN of the small yellow croaker was 54.No evidence of polymorphism, including aneuploidy or sex-related heteromorphic chromosomes, was found in any specimens examined.
| Species | Chromosome number | ||||||
|---|---|---|---|---|---|---|---|
| 42 | 44 | 46 | 48 | 49 | 50 | 52 | |
| Parapristipoma trilineatum | 18 | 70 | 88 | 211 | 53 | 0 | 0 |
| Larimichthys polyactis | 0 | 75 | 92 | 242 | 31 | 0 | 0 |
| Miichthys miiuy | 0 | 18 | 31 | 378 | 13 | 0 | 0 |
As shown in Table 2, in the family Sciaenidae, both Johnius carutt a and J. vogleri contain 48 acrocentric chromosomes and 48 FN; an exception is Bairdiella chrysura (2n=52 and AN=46; Table 2; Ojima, 1983). In the family Epinephelidae, chromosome number of Epinephelus akaara, Epinephelus alexandrinus, Epinephelus awoara, Epinephelus caninus, Epinephelus fasciatomaculosus, and Epinephelus fasciatu s was 48 acrocentrics, and the FN was 48 (Table 2; Martinez et al., 1989; Rodriguez-Daga et al., 1993; Wang et al., 2004, 2012; Minglan et al., 2014). Epinephelus bruneus consists of one pair of metacentrics, 2 pairs of submetacentrics, and 21 paris of acrocentrics, and the FN was 54 (Table 2; Minglan et al., 2014). The FN of E. bruneus and small yellow croaker was not different. However the karyotype formula of E. bruneus (2 m+4 sm+42a) and small yellow croaker (6 sm+42a) was different. In comparison with previous studies, the karyotype shown here coincided with 10 species of previous studies in chromosome number, although it differed somewhat in the chromosome complement (Table 2).
| Species | Chromosome no. | Fundamental no. | Reference |
|---|---|---|---|
| Johnius carutta | 48 | 48 | Ojima (1983) |
| Johnius vogleri | 48 | 48 | Ojima (1983) |
| Bairdiella chrysura | 52 | 46 | Ojima (1983) |
| Epinephelus akaara | 48 | 48 | Wang et al. (2004) |
| Epinephelus alexandrinus | 48 | 48 | Martinez et al. (1989) |
| Epinephelus awoara | 48 | 48 | Wang et al. (2012) |
| Epinephelus caninus | 48 | 48 | Rodriguez-Daga et al. (1993) |
| Epinephelus bruneus | 48 | 54 | Minglan et al. (2014) |
| Epinephelus fasciatomaculosus | 48 | 48 | Minglan et al. (2014) |
| Epinephelus fasciatus | 48 | 48 | Minglan et al. (2014) |
| Parapristipoma trilineatum | 48 | 48 | Park & Gil (present study) |
| Larimichthys polyactis | 48 | 54 | Park & Gil (present study) |
| Miichthys miiuy | 48 | 48 | Park & Gil (present study) |
This previous report and our current investigation can be used as a basis for continued cytogenetic studies of coastal fish around Korea for species classification or maintenance, as well as to provide basic data for the commercial production of new species. Further evaluation based on a classification and evolutionary approach in the family Sciaenidae should be considered. Moreover, further extensive studies are needed to obtain more complete information on the evolutionary direction and detailed genetic relationships among Perciformes by performing chromosome banding, detecting nucleolus-organizer regions, and analyzing chromosome in situ hybridization.

