INTRODUCTION
The small intestine is a part of a digestive tract that is responsible for nutrient absorption. To maximize its function of absorption, small intestine has developed a folding structure called villi to increase the mucosal surface area. The villi, a small thin and finger-like projections of the small intestine, are composed of superficial simple columnar epithelium and deeper connective tissue which includes blood vessels, smooth muscles, nerves, and immune cells. The proliferation of the stem cell at the base of villi leads to regeneration of the epithelial lining every seven days (Cheng & Leblond, 1974; Sato et al., 2009). When stem cells finish mitosis, the daughter cells push up the epithelium and differentiation takes place as they are proliferating along the villus. The epithelial cells at the tips of the villus undergo apoptosis and are removed from the epithelium.
An adult human has an average small intestine surface area of 30 m2. The villi increase the surface area as much as 6.5 times (Helander & Fändriks, 2014). Nutrients from digested foods can be absorbed rapidly due to enormous surface area. Large loss of absorptive surface area inhibits the effective nutritional absorption and may result in life-threatening nutrient deficiency such as malabsorption or intestinal failure (Goulet et al., 2004). When significant portion of the small intestine is removed, the increase in cell proliferation and migration helps the remaining intestine to build longer villi (Yang et al., 2004). The villi development only occurs in the fetus, and thus the villi number is unchanged after the neonatal stage (Clarke, 1967; Forrester, 1972).
The development of villi starts at embryonic day (E) 14.5 in mice which is human fetus at 8–10 weeks. The development occurs by morphogenesis, changing the structure of the pseudostratified epithelium into villi with simple columnar epithelial lining. The human and mice share similar intestine morphology and gene expression, therefore villi development of mice can be applied to the counterpart of human intestine. In mice embryo, the villi development begins in the duodenum and then spreads to the distal portion of small intestine within 36 hours (Goulet et al., 2004), and this phenomenon appears to be similar in the human intestine (de Bakker, 2016).
Hh SIGNAL
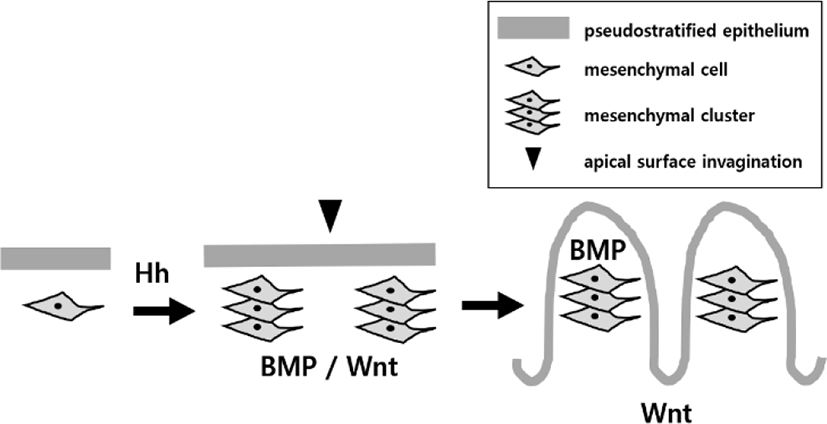
Villus development needs epithelial–mesenchymal interactions whose role has been highlighted for several decades. Epithelial–mesenchymal interactions is coordinated through signaling molecules such as hedgehog (Hh), platelet-derived growth factor (PDGF), bone morphogenetic protein (BMP) ligands, BMP signaling modifiers, Wnt and mechanical forces exerted by underlying tissue expansion and epithelial deformation (Wang et al., 2019). The proliferating epithelium secretes Hh and platelet-derived growth factor A (PDGFA). Hh signals bind mesenchymal cells, and help them to aggregate into clusters toward epithelial basement membrane (Madison et al., 2005; Kolterud et al., 2009; Walton et al., 2012).
Type of paracrine Hh signal, sonic hedgehog (Shh) and indian hedgehog (Ihh) signals are expressed by the nascent intestinal epithelium. The signals are then received by the mesenchymal cells, smooth muscle cells, endothelial cells, sub-epithelial, and myofibroblast (Ptch1-expressing subepithelial myofibroblast) that are located underneath the highly proliferative pseudostratified epithelium. The inhibition of the Hh signaling affects the villi patterning by expanding the desmin-positive smooth muscle progenitor cells and its mislocation (Madison et al., 2005). This expansion and mislocation increases the gene expression of the Tcf4/β-catenin which increases the proliferation and the formation of the precrypt structures on villus tips (Brittan & Wright, 2004).
Mice without Ihh show reduced proliferation in the intervillus region, indicating that Ihh is required to maintain the intestinal stem cells (Ramalho-Santos et al., 2000). Mice without Shh show an overgrowth of villi and there is a significant decrease in smooth muscle and the mice die after early somite stages (Ramalho-Santos et al., 2000). The requirements of Ihh and/or Shh for villi formation are further confirmed (Mao et al., 2010; Walton et al., 2012). Activation in Hh signaling leads to increase in size of villi clusters, and inhibition in Hh signaling prevents cluster to form (Madison et al., 2005; Kolterud et al., 2009; Walton et al., 2012). These results indicate that Hh signaling pathway is very important during villi development.
Mesenchymal cells express Foxf1 and Foxl1, Hh target genes (Madison et al., 2009). Inactivation of Foxf1 or Foxl1 lead to irregular villi (Kaestner et al., 1997; Ormestad et al., 2006), suggesting that the Hh function in the formation of villi is regulated partially by Foxf1 and Foxl1. It is also reported that the overexpression of the Hh signal can lead to cancer (Berman et al., 2003). The tumor is highly dependent on Hh ligand. The tumor growth is stimulated with autocrine Hh signaling instead of paracrine that was seen in villi formation (Berman et al., 2003).
The Hh signal helps to create mesenchymal clusters that are arranged in a specific pattern, but the mechanism of the cluster distribution is unknown (Walton et al., 2012). After the formation of the initial cluster, the position of the secondary, tertiary, and quaternary clusters will be established according to its previous specific patterning field. The formation of the subsequent cluster is positioned in the intervillus regions, between the two villi, when the region is large enough for the expansion of the patterning field. The intestinal villi clusters form first in the anterior, dorsal, and then towards posterior and lateral region (Breathnach, 1978).
Mesenchymal cells express many Hh target genes such as Gli1, Ptc1 (Ptch1) and PDGFRα, a receptor for the (PDGFA), before villi formation (Walton et al., 2012). Villus morphogenesis is partially coordinated through epithelial secretion which stimulates mesenchymal condensation, proliferation, and invagination of overlying epithelium to form villi (Karlsson et al., 2000). The cluster cells contain PDGFRα and the epithelial cells express PDGFA ligand. The loss of either the PDGFRα or PDGFA ligand does not change initiating clusters but inhibit subsequent clusters. The malfunction of the PDGFA ligand leads to deformed and sparse villi and the spacing between the clusters is controlled by the PDGFA ligand. These suggest both Hh and PDGF signals are coordinated to control this mesenchyme-induced process (Karlsson et al., 2000).
When epithelial stem cells isolated from adult mouse intestine are cultured, they fail to grow villi, indicating epithelial stem cells alone do not have an ability to develop to form villi without mesenchymal cells (Sato et al., 2009). There is intimately tight interdependence between mesenchymal cells and the epithelial cells.
BMP SIGNAL
The space between the clusters are usually 60–70 μm in mice (Walton et al., 2012). After the mesenchymal clusters are formed, villi emergence takes place (Walton et al., 2016). Several different types of BMP ligands are expressed by the Hh-responsive mesenchymal cells of clusters (Karlsson et al., 2000). The BMP ligands bind the cluster cells itself for regulating the cluster patterning such as the size and spacing of the clusters (Walton et al., 2016). The BMP signals are also used to regulate overlying epithelium, resulting in deformation and exit the cell cycle, and inhibit the cell proliferation at the tips of the villi (Wang, 2019).
A high concentration of BMP can inhibit cluster formation. Even after the cluster formation, a change in BMP concentration can have an effect on the cluster patterning (Walton et al., 2016). When BMP receptors are genetically deleted from the Hh-responsive mesenchymal cells or BMP inhibitor are added gradually, the cluster spot pattern turn into stripe, indicating the BMP has a function of regulating the cluster structure (Walton et al., 2016). This change in cluster pattern supports the Turing field, a reaction-diffusion model that shows the distribution and patterning of the mesenchymal clusters. This model was first suggested by a mathematician Alan Turing (Turing, 1952).
Wnt SIGNAL
The Wnt ligand is secreted by mesenchymal cells and epithelial cells (Theodosiou & Tabin, 2003; Wang et al., 2018). The Wnt signaling controls both the proliferation and differentiation in the crypt-villus axis and is used for crypt formation in intervillus regions (Van de Wetering et al., 2002; Madison et al., 2005).
When Wnt is interrupted, epithelial proliferation is significantly reduced due to decrease in Myc expression and increase in p21 (cell cycle inhibitor) expression (Madison et al., 2005). A Wnt transcriptional effector, Tcf4, is required for making and maintaining the crypt stem cell (Madison et al., 2005). Inhibition of the Tcf4 can lead to fewer villi and reduced proliferation in intervillus regions (Korinek et al., 1998). Before villi formation, thick pseudostratified epithelium is not able to express Wnt signals. After villi emerge, cluster mesenchymal cells express BMP, which repress Wnt signaling in the overlying epithelium on the clusters, confining Wnt signals to the intervillus regions (Chin et al., 2016; Nigmatullina et al., 2017).
With several BMP ligands and BMP signaling modifiers, Wnt molecules regulate cluster patterning and give the information to the overlying epithelium, which can deform and exit from the cell cycle (Wang et al., 2019).
PHYSICAL FORCE
In mice, overlying epithelial cells on the cluster receive several signals from underlying cluster and change shape, shortening apicobasaly. Cell shape change evokes intraepithelial compression force at the intervillus regions between clusters. Intraepithelial physical constraint and mitosis deform the overlying epithelium to initiate apical surface invaginations, changing thick pseudostratified epithelial layer into villi (Freddo et al., 2016). However, no direct evidence showing the relationship between physical force and signaling molecule has not been elucidated yet.
Unlike mammals, chick’s intestinal villi are formed by using physical force with the signaling molecules (Freddo et al., 2016). The physical force is generated by the smooth muscle layer which is located in the deep mesenchyme. As the smooth muscle cells differentiate, a compressive force is generated on the intestine. As the circular muscles, exterior longitudinal muscles, then the interior longitudinal muscles are differentiated in series, more and more physical constraint is given to the intestine (Shyer et al., 2013). The smooth muscle layer affects the epithelial structure to curve (Shyer et al., 2015). The curvature of the epithelium leads to zig-zag pattern formation and finally into villi structure (Shyer et al., 2013). At the ‘valley’ of the zig-zag, mesenchymal clusters are formed due to Hh signal produced by the epithelial cells (Shyer et al., 2015).
CONCLUSION
Inter-tissue communication can be possible for villi development, as epithelial-mesenchymal interactions are, on the background of close contact between two tissues, tightly coordinated through soluble signaling molecules, physical forces by expanding tissue and following epithelial deformation (Fig. 1). Overlying epithelium secretes Hh ligands including several signals to the underlying mesenchymal cells that aggregate into cluster, which express BMP affecting its own cluster patterning and deforming back overlying epithelium for initiating villi, and Wnt affecting cluster patterning and controlling the proliferation and differentiation of overlying epithelium for establishing crypt-villus axis. The deforming epithelium generates intraepithelial compression pressure to initiate apical surface invaginations, changing pseudostratified epithelial layer into villi. In chick, the physical force from the sequentially developing smooth muscle layers, causes the epithelium to deform, forming villi. The cellular and molecular drivers of the villus formation have been extensively studied, but it is not completely understood in mammals.
It is interesting to know a recent finding that the overexpression of the Hh signal can lead to cancer. The tumor growth is stimulated with autocrine Hh signaling instead of paracrine that was seen in villi development. Therefore, it is worth to aim future research on the treatment of gastrointestinal cancer by targeting the Hh signal.

