INTRODUCTION
Stem cells are undifferentiated cells characterized by their capacity for long-term self-renewal and multilineage differentiation under defined microenvironmental conditions. In 1998, James Thomson at the University of Wisconsin–Madison first derived and propagated human embryonic stem cell (ESC) from the inner cell mass (ICM) of blastocysts, establishing stable pluripotent cell lines and defining their core biological properties (Thomson et al., 1998). This landmark achievement provided a foundation for investigating early human developmental processes, pluripotency regulation, lineage specification, and translational applications targeting intractable and degenerative diseases (Temple, 2023). Over the past two decades, the field of regenerative medicine has advanced substantially, driven by improvements in stem cell isolation, in vitro expansion, direct differentiation protocols, genomic and epigenetic characterization, and functional validation (Rai et al., 2020). Stem cells derived from embryonic, fetal, and adult tissues as well as induced pluripotent stem cell (iPSC) have been extensively investigated for their therapeutic potential. These advances have facilitated the initiation and expansion of global clinical trials evaluating stem cell–based therapeutics across diverse indications, including neurodegenerative, cardiovascular, metabolic, and musculoskeletal disorders. Concurrently, increasing investment from pharmaceutical and biotechnology sectors has accelerated the translation of stem cell technologies into clinically applicable products. With ongoing progress in manufacturing standardization, safety assessment, and regulatory frameworks, stem cell–based therapeutics are increasingly positioned as a transformative platform for next-generation regenerative medicine.
The global stem cell therapeutics market was valued at approximately USD 297 million in 2022 and was projected to grow at a compound annual growth rate (CAGR) of 25.5%, reaching an estimated USD 558 million by 2027. In Korea, the domestic stem cell therapeutics market has likewise been expected to expand significantly, with projections estimating a market size of approximately USD 2.6 million in 2024, reflecting continued industrial growth and commercialization efforts (Bahari et al., 2023). In response to this rapid expansion, many countries have increased investment in the development of stem cell–based therapies while actively promoting commercialization strategies linked to advanced biopharmaceutical technologies. Through the establishment of multifaceted innovation ecosystems and the strengthening of collaborative research networks among leading global stem cell research institutes and universities, efforts are underway to enhance safety evaluation frameworks and develop next-generation platform technologies. These initiatives are intended to accelerate technology transfer and facilitate market entry (Takahashi et al., 2023). Within this competitive environment, technologically advanced countries emphasize securing core intellectual property (IP) rights related to stem cells and stem cell–based therapeutics, alongside implementing strategic patent portfolio management, litigation preparedness, and freedom-to-operate analyses. In contrast, compared with leading nations, Korea continues to face challenges in securing foundational IP rights and establishing comprehensive defensive strategies to protect core stem cell technologies (Hernández-Melchor et al., 2023).
Patent information analysis can generally be classified into three major methodological approaches (Sharma & Tripathi, 2017; van Raan, 2017; Yamada, 2022). First, the exploratory research method focuses on quantitative assessments, including the current status of patent holdings, temporal changes in application volume, and various index-based evaluations. Second, the qualitative analysis method involves arranging technological developments in chronological order, conducting patent network analyses through matrix construction and technology gap identification, and examining individual patents to assess claim scope and the breadth of rights. Third, the normative research method entails in-depth analysis of the technical content and legal scope of individual patents, followed by systematic organization and synthesis of the findings. Although a substantial volume of IP data is generated annually through continuous patent filings and registrations worldwide, key patent analysis indicators are commonly categorized as follows: 1) technological competitiveness (“Technological Value”) includes citation index, technology strength index, identification of core patents, citation ratio, patent longevity index, number of joint applications, scientific linkage, technology innovation cycle, alignment with technological trends, patent share, patent activity index, and patent grant rate. 2) Marketability (“Market Value”) includes applications per applicant, inventions per inventor, examination request productivity, number of Patent Cooperation Treaty (PCT) applications, number of examination requests, examination request rate, patent maintenance rate, and patent competitiveness index. 3) Impact (“Spillover Effect”) includes technology dependency, technology impact index, knowledge diffusion and absorption index, and market diffusion potential. 4) Activity (“Activity Level”) includes number of applications, application growth rate, number of registrations, number of examination requests, patent grant rate, number of valid patents, and average remaining patent term. 5) Legal Strength (“Rights Strength”) includes the number of independent and dependent claims, breadth of claim scope, and overall robustness of patent protection. Given the rapid accumulation of patent data—particularly in fast-evolving technological domains—there are inherent limitations in conducting comprehensive and real-time IP trend analyses. Accordingly, both domestically and internationally, there has been increasing emphasis on the development of patent maps to efficiently analyze patterns in stem cell–related IP. Patent maps systematically organize IP information across multiple dimensions—such as stem cell types, isolation methods, characterization techniques, marker analyses, differentiation protocols, and disease-specific applications filed globally—thereby facilitating identification of technological trends and competitive positioning (Hernández-Melchor et al., 2023; Xu et al., 2025). These patent maps are widely utilized from the early stages of stem cell research through R&D planning, technology commercialization strategy development, and preparation for potential patent infringement disputes among competing entities.
In recent years, stem cell–based therapeutic technologies have advanced rapidly, driven by increasing specialization within the stem cell field and the expansion of target disease indications. In particular, convergence with broader biotechnology platforms—including gene editing, biomaterials, and advanced bioprocessing technologies—has accelerated innovation and diversified therapeutic applications. Since 2015, stem cell therapeutics have increasingly demonstrated commercial feasibility as safety and efficacy have been validated through preclinical and clinical studies supported by IP–based technological frameworks Mendicino & Weber, 2015). Furthermore, technology life cycle analyses centered on IP portfolios have highlighted the strategic importance of commercialization models built upon proprietary stem cell platform technologies. Nevertheless, prior research has largely been limited to fragmented analyses of domestic and international patent trends. Given the technical complexity and high degree of specialization within the stem cell field, significant challenges remain in identifying truly foundational or “core” patents that drive technological advancement (Hernández-Melchor et al., 2023; Li & Fu, 2025). Moreover, insufficient integrative analyses linking IP-based translational research with clinical trial progression have resulted in an incomplete understanding of the critical determinants required to translate stem cell–related IP into commercially viable therapeutic products. Consequently, substantial limitations persist in systematically identifying and evaluating the key linkage factors necessary for successful technology commercialization based on stem cell–related IP (Mendicino & Weber, 2015; David et al., 2018; Malik, 2021). Accordingly, the objective of this study was to systematically analyze the characteristics of stem cell–based IP, focusing on ESCs, ASCs, and iPSCs. Furthermore, this study examined the correlations among scientific publications, IP filings, and clinical trial activities associated with each stem cell type between 2000 and 2020. In addition, we sought to comparatively evaluate the relationships between IP characteristics and key determinants influencing the commercialization of stem cell–based therapeutics. Through this integrative analysis, the study aimed to identify linkage patterns among research output, patent activity, and clinical translation, thereby providing strategic insights into the commercialization potential of stem cell technologies.
Stem cells are undifferentiated cells that function as the fundamental cellular units of multicellular organisms. They are present throughout all stages of human development—including the fertilized oocyte, embryo, fetus, and adult tissues—and are defined by two key properties: self-renewal, the capacity for sustained proliferation while maintaining an undifferentiated state, and differentiation, the ability to generate specialized cell types that contribute to tissue development, homeostasis, and repair. Modern human stem cell research advanced significantly in 1998 when James Thomson at the University of Wisconsin–Madison successfully derived and propagated human ESC from the ICM of blastocysts approximately seven days after in vitro fertilization (Thomson et al., 1998). The establishment and biological characterization of stable pluripotent ESC lines marked a pivotal milestone, providing an experimental platform for investigating early human development, pluripotency regulation, and regenerative applications. The defining biological feature of stem cells is their dual capacity for long-term self-renewal and multilineage differentiation, both in vitro and in vivo. Based on their differentiation potential, stem cells are broadly classified into ESCs (pluripotent), adult (somatic) stem cells (multipotent), and iPSC. iPSC, generated through the ectopic expression of defined transcription factors that reprogram somatic cells into a pluripotent state, have substantially expanded the conceptual and translational scope of stem cell biology (Herberts et al., 2011; Avinash et al., 2017; Khandia et al., 2024). In addition, accumulating evidence suggests that certain malignancies harbor a subpopulation of tumor-initiating cells, commonly referred to as cancer stem cells, which exhibit stem cell–like properties (Fig. 1).
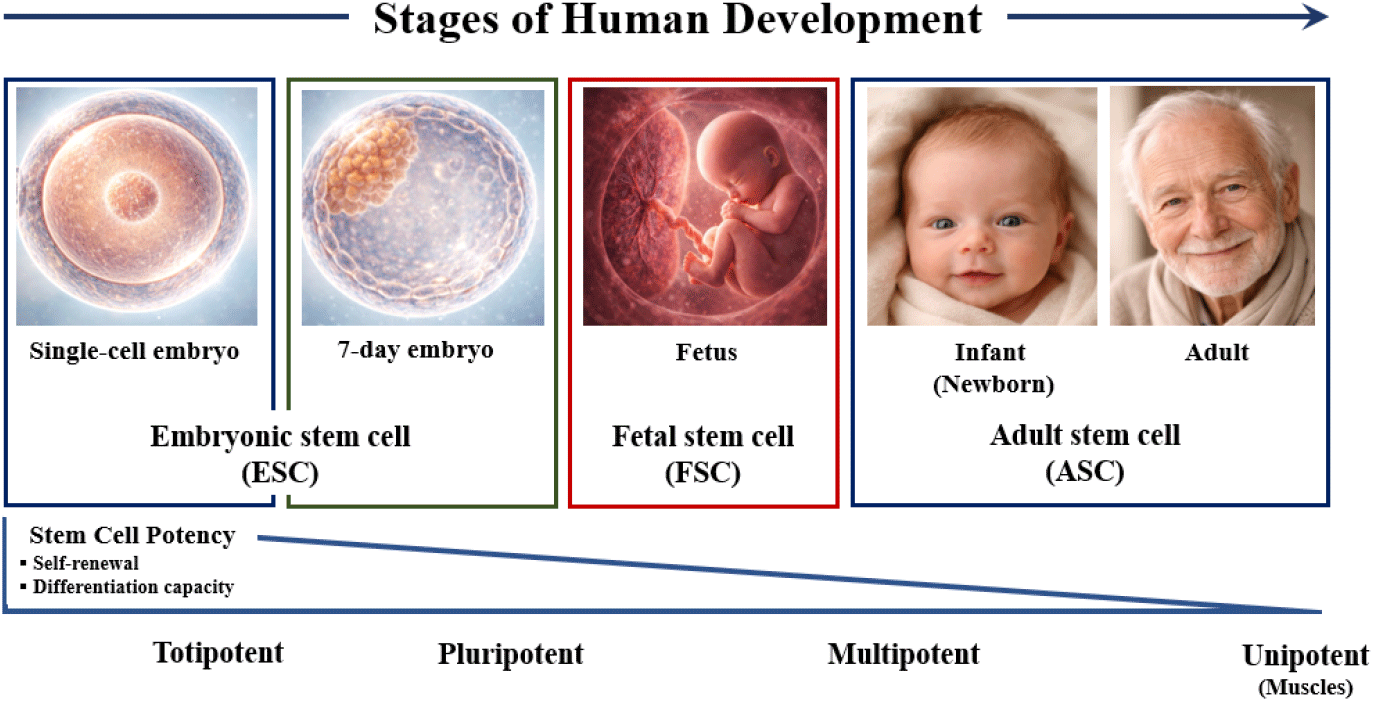
A critical determinant in categorizing stem cell populations is their differentiation potential, which varies across developmental stages and cell types (Tárnok et al., 2010; Kirkeby et al., 2025a). 1) Totipotent cells, exemplified by the zygote and early cleavage-stage blastomeres, possess the highest developmental capacity, able to generate all embryonic and extraembryonic lineages, including the placenta, thereby giving rise to a complete organism. 2) Pluripotent stem cells (PSCs), including ESC and iPSC can differentiate into derivatives of all three germ layers—ectoderm, mesoderm, and endoderm—but cannot independently form a full organism. ESC lines were first derived from the ICM of human blastocysts by James Thomson in 1998, establishing the foundation for human pluripotent stem cell research. iPSC were subsequently generated by Shinya Yamanaka in 2006 via reprogramming of somatic cells using four transcription factors—Oct4, Sox2, Klf4, and c-Myc—demonstrating pluripotency equivalent to ESC while circumventing ethical constraints associated with embryo use. iPSC have since been successfully derived from human somatic cells, expanding the toolkit for disease modeling, drug discovery, and regenerative therapies (Takahashi & Yamanaka, 2006). 3) Multipotent stem cells, such as hematopoietic stem cells (HSCs) and mesenchymal stem cell (MSC), exhibit lineage-restricted differentiation potential. HSCs generate all blood lineages, whereas MSCs can differentiate into mesodermal derivatives including bone, cartilage, and adipose tissue, making them valuable for tissue repair and regenerative applications. 4) Unipotent stem cells, including muscle satellite cells, are restricted to producing a single differentiated cell type. Differentiation potential therefore provides a framework for categorizing stem cells into totipotent, pluripotent, multipotent, and unipotent populations, informing both basic research and translational applications (Table 1). Understanding these distinctions is critical for designing stem cell–based therapies and for selecting appropriate cellular sources for disease modeling and regenerative interventions.
Stem cells constitute a cornerstone of regenerative medicine, particularly for degenerative and intractable diseases lacking effective conventional therapies. By harnessing the body’s intrinsic regenerative capacity, stem cell–based technologies are shifting the therapeutic paradigm from standardized treatment toward personalized, patient-specific interventions, thereby redefining the future landscape of healthcare. In the 21st century and the era of the Fourth Industrial Revolution, stem cell technologies have emerged as a central driver of the regenerative medicine industry. Beyond the development of tissue-specific cell therapies for rare and refractory conditions, these technologies are increasingly integrated with interdisciplinary research platforms platforms (Hussen et al., 2024; Li & Fu, 2025), including: 1) organoid systems for modeling human development and disease pathogenesis. 2) Genetic disease modeling to study cellular and molecular mechanisms. 3) Drug screening platforms for toxicity and efficacy evaluation. 4) Convergent approaches for the discovery of novel therapeutics.
This integration has substantially expanded the potential applications of stem cell technologies, both in basic research and translational medicine. Consequently, leading research institutions and market analyses predict that the technological and economic impact of stem cell–based therapeutics will sustain a high CAGR relative to conventional pharmaceuticals. For example, institutions such as Massachusetts Institute of Technology have identified stem cell manipulation technologies, ASC–based therapeutics, and regenerative medicine platforms as among the most promising areas for future technological development (Hussen et al., 2024). The following sections describe the characteristics of IP associated with each major type of stem cell and their implications for research, commercialization, and clinical translation (Table 2).
IP, intellectual property; ICM, inner cell mass; HLA, human leukocyte antigen; GMP, Good Manufacturing Practice.
The modern era of ESC research began in 1998, when Dr. James Thomson developed methods to efficiently isolate and culture the ICM from human blastocysts. Following this breakthrough, numerous patents were filed covering culture methods, media compositions, and protocols for differentiation into diverse cell types representing the three germ layers. Intellectual property activity rapidly expanded in areas including 1) introduction and expression of genes to modify ESCs for therapeutic applications targeting genetic diseases. 2) Somatic cell nuclear transfer (SCNT) technologies. 3) Genetic manipulation strategies for clinical use. Patents in the ESC field primarily focus on generating specific cell types to replace damaged tissues in rare or refractory diseases (Abusalah et al., 2024). There are key domains include 1) directed differentiation and lineage specification methods, 2) compositions and media formulations promoting differentiation, and 3) scaffolds, devices, and assays to enhance and evaluate differentiation efficiency. However, a critical translational challenge for ESC-based therapies is the tumorigenic potential of residual undifferentiated cells. Consequently, ESC related patents targeting purification and safety-enhancing technologies have become central (Hayakawa et al., 2015) including 1) surface marker analyses to identify undifferentiated cells, 2) selective elimination via undifferentiated cell–specific suicide genes, and 3) cell sorting and purification strategies to maximize the purity of fully differentiated populations. Such marker analysis and separation technologies are currently regarded as core technical areas in the ESC IP landscape (Hernández-Melchor et al., 2023). In addition, transplantation of ESC-derived cells may provoke immune rejection. To address this, patent filings increasingly focus on immune-evasion strategies, particularly gene knock-in/knock-out approaches using CRISPR-Cas9 genome editing (Lei et al., 2024). These efforts reflect the growing emphasis on improving the safety, clinical applicability, and translational potential of ESC-derived therapeutics.
ASCs represent a cornerstone of regenerative medicine, encompassing HSCs from bone marrow, MSC from connective tissues, and organ-specific progenitors such as neural, hepatic, muscle, and periodontal stem cells (Resnik, 2002; Riaz et al., 2024). Intellectual property in the ASC field has evolved alongside advances in isolation, expansion, and clinical application, reflecting both foundational and translational innovations.
Early patent activity in HSC focused on methods for ex vivo expansion, proliferation, and lineage-specific differentiation, often mediated through growth factors or cytokine combinations. Patents also covered techniques for isolation and fractionation using cell surface markers and gene expression profiling to obtain enriched HSC populations (Mitchell, 2020). However, recent trends have shifted toward addressing limitations in large-scale HSC production. Current patents increasingly emphasize bioprocess optimization, including scalable culture systems, automated manufacturing platforms, and standardized protocols to ensure reproducibility and clinical-grade quality. These innovations aim to enhance the translational potential of HSC-based therapies for hematologic disorders, immune deficiencies, and regenerative applications.
Early MSC patents primarily focused on composition-of-matter claims, covering the identification of tissue-specific markers, isolation procedures, and culture methods optimized for proliferation and differentiation potential (Jayaraman et al., 2021; Strecanska et al., 2025). Subsequent patents addressed culture medium formulations, three-dimensional scaffolds, and biomaterial supports to maintain stemness and lineage-specific differentiation. With the expansion of global clinical trials, recent MSC IP has increasingly focused on therapeutic applications, including 1) immunomodulatory and anti-inflammatory properties of MSC, 2) combination therapies integrating MSCs with pharmaceuticals or bioactive scaffolds, and 3) tissue engineering strategies for cartilage, bone, and musculoskeletal repair. Emerging patents also highlight methods for purifying MSC populations, enhancing engraftment efficiency, and reducing immunogenicity. Collectively, these IP activities demonstrate a strong emphasis on translating MSC research into commercially viable therapeutics (Hinsenkamp et al., 2022; Opretzka et al., 2024).
iPSC were first developed in 2006 by Dr. Shinya Yamanaka through the introduction of four transcription factors—Oct4, Sox2, Klf4, and c-Myc—into somatic cells, reprogramming them to a pluripotent state (Yamanaka, 2007). This breakthrough prompted extensive patent activity, with filings targeting reprogramming methods, vector systems, and applications in regenerative medicine. Also, iPSC is artificially generated cell developed to overcome the limitations of ESCs, including 1) the use of human oocytes, 2) ethical concerns related to embryo destruction, and 3) immune rejection following transplantation. iPSC are created by reprogramming differentiated somatic cells—typically fibroblasts—through the regulation of specific gene expression, thereby conferring key characteristics of ESCs, such as unlimited self-renewal and pluripotency (Okita et al., 2007). Globally, Japan has played a leading role in iPSC technology and therapeutic development. With strong governmental support, Professor Shinya Yamanaka’s research group initiated the world’s first clinical trial using iPSC-derived cells for an ophthalmic disease (Mandai et al., 2017).
Intellectual property related to iPSC increased rapidly between 2006 and 2018, particularly in the United States and Japan (Yamanaka, 2012; Lyu et al., 2024). However, a noticeable decline in patent activity has been reported in recent years. In the early phase of iPSC research, the field attracted substantial academic and university-based research efforts because iPSC were considered a promising alternative to ESCs. Nevertheless, a critical step in therapeutic development—involving viral vector–based overexpression of reprogramming genes—raised significant safety concerns due to risks of tumorigenicity and oncogenesis. As a result, patent filings have declined, particularly in areas related to viral gene delivery technologies, differentiation control methods, and safety issues associated with tumor formation. In the iPSC field, IP has increasingly shifted toward disease-specific cell technologies. Compared with patents covering iPSC generation techniques, a larger proportion of IP filings focus on differentiation technologies for patient-specific cell production, personalized disease treatment, and drug screening platforms targeting specific therapeutic compounds.
Therapeutically, major areas of iPSC-related patent activity include treatments for neurological disorders such as Parkinson’s disease and stroke, rare intractable vascular diseases, and regenerative applications involving differentiation into cardiomyocytes for damaged myocardium and muscle cells for degenerative muscle disorders (Meneghello et al., 2015). More recently, Professor Shinya Yamanaka has advanced research on “universal iPSC,” which are designed not only for autologous use but also for allogeneic transplantation into other patients. By modulating the expression of immune regulatory genes—particularly human leukocyte antigen (HLA) haplotypes—to evade T cell–mediated immune responses, preclinical animal studies have demonstrated reduced immune rejection (Bogomiakova et al., 2023). Consequently, research and IP related to gene-editing technologies aimed at developing commercially viable and broadly applicable iPSC-based therapeutics continue to expand. Overall, the iPSC IP landscape mirrors that of ESC in its focus on controlling pluripotency, enhancing safety, and enabling translational potential (Hu, 2014; Escribá et al., 2024). However, iPSC offer distinct advantages in personalized medicine applications due to their patient-specific origin, which has further fueled innovation and patent activity in this field (Li & Fu, 2025).
Allogeneic MSC–based therapeutics currently represent one of the most actively developed classes within the global cell therapy sector (Watanabe et al., 2021; Jovic et al., 2022). Biotechnology companies, including Systemix and Osiris Therapeutics, have been at the forefront of MSC-based therapeutic development. The development of stem cell therapeutics is inherently resource- and time-intensive. Following comprehensive stem cell characterization, preclinical validation of safety and efficacy is required in both in vitro and in vivo models, preceding multi-phase clinical trials. Given the substantial investment involved, the identification and protection of core technological capabilities through IP is critical for successful commercialization. Well-positioned IP rights provide not only legal protection but also a foundation for technology transfer, strategic collaborations, and investor confidence. Between 2017 and 2025, IP filings related to stem cell therapeutics are projected to grow at an average annual rate of approximately 24.4%, with IP pertaining to manufacturing processes for clinical-grade stem cells expected to expand at an even higher rate of 34.9% per year (Lechanteur et al., 2021; Sawarkar et al., 2022; Kirkeby et al., 2025b). This trend underscores the increasing importance of scalable production technologies relative to stem cell acquisition methods. Consequently, commercialization has been especially active in the ASC sector, particularly involving MSC and umbilical cord blood–derived stem cells. Major applications of stem cell–based therapeutics include regenerative medicine and drug development for aging-related diseases, cardiovascular and neurological disorders, musculoskeletal conditions, and autoimmune or degenerative diseases. Despite these promising projections, the field continues to face critical challenges, including high development costs, prolonged regulatory pathways, safety concerns such as tumorigenicity and immune rejection, and the need for robust, scalable manufacturing systems compliant with stringent regulatory standards (Malik, 2021; Hussen et al., 2024). Sustained technological innovation and strategic IP management are therefore essential to ensure long-term success in stem cell therapeutic commercialization.
In particular, North America—including the United States, which accounts for approximately one-third of the global stem cell therapeutics market—is expected to maintain steady growth in the commercialization of technologies emphasizing safety validation and regulatory compliance. In Europe, rising interest in stem cell–based therapies for cardiovascular diseases, coupled with strengthened safety regulations, is driving market expansion. In contrast, Asia—particularly Korea and China—is projected to experience the most rapid growth in stem cell–based technology commercialization (Wong & Mahalatchimy, 2018; Hernández-Melchor et al., 2023; Wang, 2024). This trend is fueled by factors such as aging-related medical tourism, increased healthcare investment, and supportive regulatory environments. Commercialization in Korea has progressed notably. In 2011, Pharmicell (formerly FC Pharmicell) received approval from the Ministry of Food and Drug Safety (MFDS) for Hearticellgram-AMI, an autologous bone marrow–derived MSC therapy for acute myocardial infarction, representing the world’s first government-approved commercial stem cell therapeutics. Subsequently, additional stem cell therapies have gained approval in Korea, including an allogeneic umbilical cord blood–derived stem cell therapy for knee cartilage defects developed by Medipost (Cartistem), an autologous adipose-derived MSC therapy for Crohn’s fistula developed by Anterogen (Cupistem), and an allogeneic umbilical cord blood–derived stem cell therapy for amyotrophic lateral sclerosis developed by Corestem (NeuroNata-R). With four approved stem cell therapeutics, Korea is recognized as one of the fastest countries to achieve commercialization of stem cell–based technologies (Tan et al., 2025). Currently, approximately 27 cell therapy products have received approval for clinical trials in Korea, with MSC–based therapies—primarily in Phase I/II trials—constituting the majority of ongoing developments.
In stem cell–based technology commercialization, MSC represent the most active cell source, accounting for approximately 64.6% of total therapeutic development and commercialization efforts. The dominance of MSC-based therapies is largely attributable to their relative ease of isolation from autologous tissues, including bone marrow and adipose tissue. Alongside HSC separation and production technologies, MSC are being extensively developed as therapeutic agents for a wide range of degenerative diseases (Jovic et al., 2022) Currently, over 60 companies are actively engaged in MSC-based therapeutic development, with the global market projected to exceed USD 15.5 billion by 2024, reflecting substantial growth. In particular: autologous MSC therapies show a CAGR of approximately 10.5%, with a reported market share of 42.1% as well as MSC-based products constitute the majority of both autologous and allogeneic stem cell therapies under development. In contrast, allogeneic stem cell therapies have recently gained greater recognition as next-generation stem cell products (Li & Fu, 2025). This shift is largely due to the limitations of autologous stem cells, including 1) reduced stem cell activity associated with aging donor cells, 2) prolonged in vitro expansion periods, and 3) increased production costs due to supplementation with multiple growth factors to compensate for reduced cell potency. Consequently, allogeneic MSC therapies have experienced marked growth, with their market share rising to approximately 53.6%, surpassing that of autologous MSC therapies (Zhu et al., 2025).
When categorized by treatment type, allogeneic stem cell therapies represented the largest market segment, valued at approximately USD 7.4 billion (54%) in 2019. Autologous stem cell therapies also maintained a substantial market presence, with an estimated value of USD 5.8 billion (Table 3). Allogeneic therapies utilize stem cells from donors whose HLA are matched to the recipient. Advantages such as scalable production, standardized manufacturing, and immediate availability have positioned allogeneic stem cells as the largest and most actively pursued segment, with numerous products in development and commercialization pipelines. Autologous therapies, in contrast, rely on a patient’s own cells, minimizing immune rejection and offering a highly personalized treatment approach. Demand for these therapies continues to rise alongside the increasing prevalence of chronic diseases, supporting sustained market growth in this segment. Syngeneic therapies, which involve stem cells from genetically identical individuals (e.g., twins), occupy a niche market due to limited donor availability, resulting in a comparatively small share of the stem cell therapeutics market (Bahari et al., 2023).
CAGR, compound annual growth rate.
In contrast, ESC have the theoretical potential to provide an unlimited supply of transplantable cells for patients with degenerative diseases. However, several technical and regulatory challenges have limited their commercialization compared with MSC. These challenges include 1) complexity in large-scale culture and manufacturing processes, 2) accumulation of genetic mutations during long-term culture, 3) difficulties in precise differentiation control, 4) ensuring high-purity cell selection and recovery, and 5) risk of immune rejection following transplantation. Due to these limitations, ESC-based therapeutic commercialization accounts for only approximately 16% of stem cell technology commercialization efforts, significantly lower than that of MSC-based therapies. Similarly, iPSC currently represent an estimated 11% of commercialization activity (Hayakawa et al., 2015; Kirkeby et al., 2025a). Although iPSC offer advantages such as bypassing ethical concerns associated with embryo use, technical hurdles remain, including improving reprogramming efficiency and addressing tumorigenicity risks. Nevertheless, continued advancements in gene regulation and genome editing technologies are improving production efficiency and safety profiles. As these technical barriers are progressively resolved and related IP portfolios expand, iPSC-based technologies are expected to demonstrate a relatively rapid growth rate in future commercialization.
MSC remain the most extensively developed cell source in the stem cell therapeutics sector, accounting for approximately 64.6% of global therapeutic development activities. This dominance is largely driven by their relative ease of isolation from autologous tissues such as bone marrow and adipose tissue, and by the strong translational momentum provided by ongoing clinical trials. In addition to HSC separation and production technologies, MSCs are being actively developed as therapeutic agents for a broad range of degenerative diseases. Currently, more than 60 companies are engaged in MSC-based product development, with the market projected to exceed USD 15.5 billion by 2030, reflecting significant growth potential. Despite this positive outlook, several challenges constrain the broader commercialization of MSC therapies. Both autologous and allogeneic MSC products face; 1) strict regulatory requirements, 2) ethical considerations depending on cell source, 3) high treatment costs, and 4) complex manufacturing and quality control processes (Strecanska et al., 2025). These limitations can hinder rapid market expansion. Nevertheless, investment in MSC-based regenerative medicine continues to grow, supported by a competitive global market and limited threat from substitute technologies. Regulatory momentum has increased in recent years, particularly following approvals of MSC-based therapies by the U.S. Food and Drug Administration (FDA). These approvals have catalyzed research and development for MSC applications in osteoarthritis, tissue regeneration, and other degenerative conditions, reinforcing the continued expansion of the stem cell therapeutics landscape (Malik, 2021; Bahari et al., 2023).
METHODS
In this study, data were systematically collected from multiple specialized databases to evaluate IP, scientific publications, and clinical research activity in the stem cell field.
Patent searches were performed using the Korea Intellectual Property Rights Information Service (KIPRIS, https://www.kipris.or.kr) and the WIPSON patent information search service (WIPS, https://www.wipson.com), which provide comprehensive coverage of domestic and international patent filings related to stem cell technologies.
Peer-reviewed, SCI-indexed publications were identified through PubMed (https://ubmed.ncbi. nim.nih.gov), the National Institutes of Health (NIH) bibliographic database, to capture research outputs on ESC, ASC, and iPSC.
Information on clinical trials was obtained from ClinicalTrials.gov, a publicly accessible registry maintained by the NIH (https://www.clinicaltrials.gov), which provides detailed records of study protocols, trial phases, and participant recruitment status for stem cell–based therapeutics.
This multi-source approach enabled a comprehensive assessment of the current landscape of stem cell research, IP activity, and clinical translation, facilitating correlation analyses between patent activity, scientific output, and clinical trial progression.
A comprehensive patent search was conducted for a 20-year period from 2000 to 2020 using the keywords “embryo-derived stem cells (ESC)”, “induced pluripotent stem cells (iPSC)”, and “adult stem cells (ASC)”. The search targeted both published and registered patents within the KIPRIS (https://www.kipris.or.kr) database. Patents were retrieved if the keywords appeared in the title, abstract, or claims, encompassing filings that had been submitted, registered, and publicly disclosed. The search period was determined based on the standard 20-year patent term, with the timeframe defined from January 1, 2000, to December 31, 2020, using the filing date as the reference. Domestic patents were restricted to those for which the priority country was listed as “KR.” No additional noise-filtering procedures were applied during the initial retrieval. However, patent families representing identical technologies, as well as patents with highly similar titles or those filed as divisional or duplicate applications, were removed prior to analysis to prevent redundancy. This approach enabled a systematic assessment of trends in stem cell–related IP, providing a foundation for subsequent correlation analyses with scientific publications and clinical trial activity.
Second, from the analyzed dataset, ASC–related patents, which exhibited the most active linkage to technology transfer, commercialization, and clinical research, were selected for further investigation. Considering that a minimum of five years is generally required for the initiation of clinical research, the development of stem cell therapeutics, and subsequent technology commercialization, the analysis was restricted to patents registered by 2015. For these selected patents, detailed patent-related variables were evaluated to identify the factors most significantly associated with successful commercialization of stem cell therapeutics.
SCI-indexed publications were retrieved from PubMed, a database maintained by the U.S. NIH. Searches were performed using key terms including ESCs, iPSCs, and ASCs, with supplementary keywords reflecting core stem cell properties, namely self-renewal and differentiation. The analysis encompassed publications issued over a 20-year period (2000–2020), and only articles published within this timeframe were included in the study.
Clinical trial data were obtained from ClinicalTrials.gov, a global registry maintained by the NIH (https://www.clinicaltrials.gov). The search encompassed trials conducted worldwide, covering all clinical indications. Trials were identified using the key terms embryo-derived stem cells, iPSCs, and ASCs. Studies involving precursor or progenitor cells, such as endothelial precursor cells (EPCs) and neural precursor cells, were excluded from the analysis to focus specifically on stem cell–based therapeutic interventions.
RESULTS
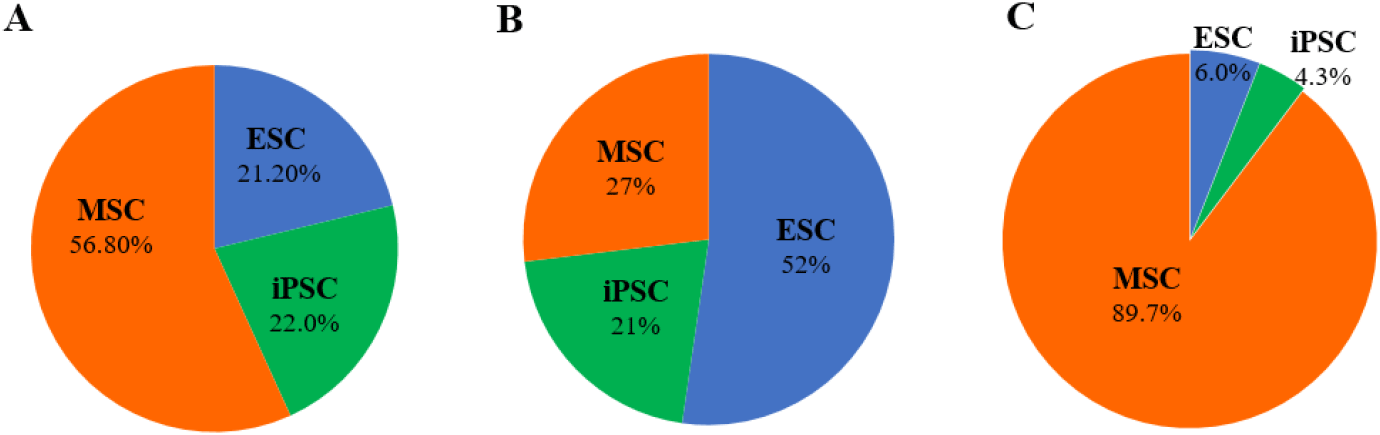
To evaluate research trends and translational progression among major stem cell types, we analyzed the relative distribution of MSC, ES, and iPS across publications, patents, and investigational new drug (IND) applications (Fig. 2). In scientific publications (×103), MSC accounted for the largest proportion (56.8%), indicating that MSC-based research constitutes the dominant area of academic investigation. iPS represented 22.0% of publications, while ES comprised 21.2%, demonstrating a relatively comparable but smaller research output for pluripotent stem cell types (Fig. 2A). In contrast to publication trends, patent filings (×102) were primarily concentrated on ES technologies (52.3%). MSC-related patents represented 26.9%, whereas iPS accounted for 20.8%. This shift suggests that, although MSC dominate academic research, ES technologies have generated comparatively stronger IP activity (Fig. 2B). Analysis of IND applications revealed a markedly different distribution pattern. MSC overwhelmingly dominated clinical translation, representing 89.7% of total IND submissions. ES and iPS accounted for only 6.0% and 4.3%, respectively. These findings indicate that MSC-based therapies have progressed most actively toward clinical application, while pluripotent stem cell technologies remain comparatively limited in clinical-stage development. Collectively, these results demonstrate a divergence between academic research output, patent activity, and clinical translation among stem cell types, with MSC leading translational advancement despite ES dominance in patent filings.
Since the seminal report by James Thomson in 1998, which characterized human ESC and highlighted their potential in drug discovery and regenerative medicine, research output in the stem cell field has expanded markedly. From 2000 onward, the number of SCI-indexed publications included 663 articles on ESC and 108 articles on ASCs. Over the next two decades, through 2020, annual publication counts increased substantially for ESCs and iPSC. Notably, publications on ESC exhibited a relative plateau around 2015, whereas iPSC-related publications continued to grow steadily through 2020. In contrast, research on ASCs demonstrated an exponential increase after 2013, reflecting heightened interest in translational and clinical applications (Fig. 3A).
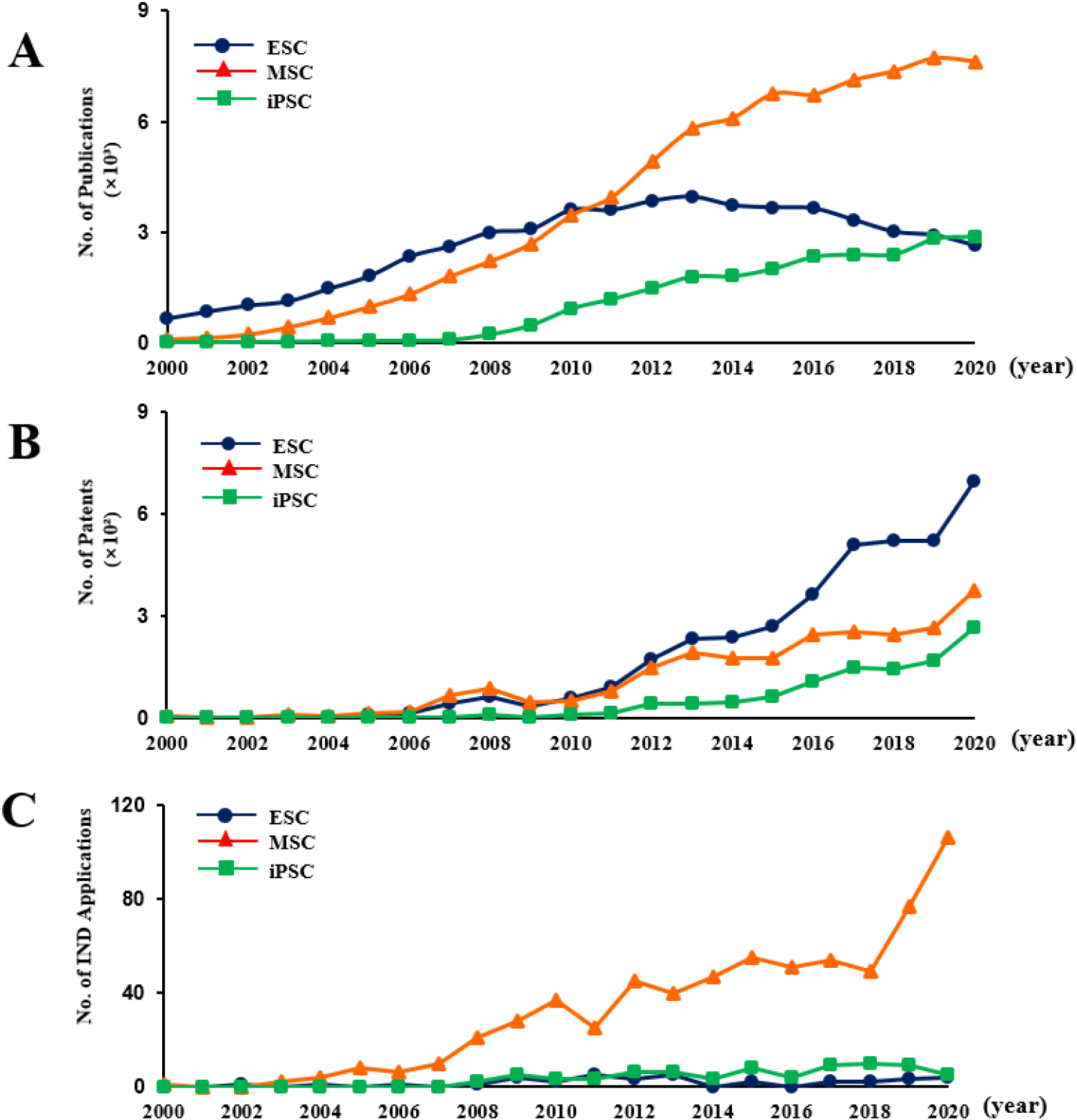
In Korea, one of the earliest stem cell–related patents was registered in 2004 by Corestem Inc. Although this patent has since expired, it pertained to umbilical cord blood–derived ASCs containing HSCs and covered therapeutic applications for cartilage defects, nerve injury, muscle and cardiac disorders, and leukemia. From 2005 onward, patents related to ESC and ASCs were actively registered. For iPSC the first domestic patent was filed in in 2007 to Geron, addressing drug screening using hepatocyte-lineage cells derived from pluripotent stem cells, as well as their clinical applications for liver function protection and treatment of liver diseases.
Beginning in 2012, the registration of patents for ESC, iPSC, and ASC accelerated substantially. ESC-related patents primarily focused on 1) the production of feeder cells essential for maintaining pluripotency, 2) culture medium formulations for stem cell maintenance, 3) technologies enhancing differentiation into specific cell types under controlled in vitro conditions, and 4) markers for isolating and characterizing differentiated cells. Also, ASC patents emphasized on characterization of stem cells derived from various tissues, methods to enhance frequency and expression of specific surface markers and use of factors regulating differentiation potential. Otherwise, iPSC patents largely addressed improvements to reprogramming technologies originally developed by Shinya Yamanaka—including Oct4, Sox2, c-Myc, and Klf4—focusing on 1) optimization of viral vector systems, 2) enhanced delivery and expression of reprogramming factors, and 3) overall increases in reprogramming efficiency. These trends reflect the maturation of stem cell technologies in Korea and underscore the growing focus on commercially relevant, clinically translatable innovations (Fig. 3B).
The clinical development of stem cell therapeutics follows a conventional phased approach, beginning with preclinical validation of safety and efficacy in vitro and in vivo, followed by phased human clinical trials. All stem cell products must be manufactured under certified Good Manufacturing Practice (GMP) standards. Based on chemistry, manufacturing, and controls data, sponsors are required to obtain IND approval from regulatory authorities before initiating human studies. Clinical trial registrations, including protocols and results, are publicly available through ClinicalTrials database (https://www.clinicaltrials.gov). This study analyzed the registration trends of clinical trials for ESC, iPSC, and ASC.
For ESCs, the first Phase I clinical trial was registered in 2002 by Hadassah Medical Organization (PI: Benjamin E. Reubinoff, M.D., Ph.D.; ClinicalTrials.gov ID: NCT0033197) to evaluate safety using surplus embryos donated from in vitro fertilization procedures. As of 2020, only 36 ESC-based clinical trials had been registered, highlighting limited clinical progress in this area. Similarly, iPSC-based trials remain limited. The first human study was registered in 2008 by the same institution (ClinicalTrials.gov ID: NCT00801333) to assess clinical-grade production and safety of iPSC derived from normal somatic cells. By 2020, a total of 73 iPSC trials had been registered, reflecting the early stage of clinical translation despite their pluripotent potential. In contrast, ASCs—including HSCs and MSCs—exhibited substantial clinical activity.
From 2010 onward, the number of registered clinical trials involving ASCs increased rapidly, with 106 trials registered in 2020 alone. Over the 20-year period from 2000 to 2020, a total of 666 ASC–based clinical trials were identified (Fig. 3C). These studies targeted a wide range of indications, including autoimmune disorders, acute myocardial infarction, neurological and cardiovascular diseases, liver cirrhosis, and other degenerative conditions. Among the 666 trials, 169 were Phase I studies primarily designed to evaluate safety. Following safety verification, 507 trials progressed beyond Phase I, advancing into Phase II or later stages of clinical assessment. Notably, 46 trials reached Phase III, where long-term efficacy and safety were evaluated through comparisons with placebo controls.
Collectively, these findings indicate that ASC–based therapies have achieved substantial clinical advancement over the past two decades. The progression of a considerable proportion of trials into late-stage development suggests a relatively mature stage of clinical translation. Provided that long-term follow-up studies confirm safety and sustained efficacy, these therapies may be positioned to advance toward regulatory approval, broader clinical adoption, and eventual commercialization.
To assess the commercialization potential of stem cell therapeutics, correlation analyses were conducted between patent activity and both SCI-indexed publications and registered clinical trials for each stem cell type (Fig. 4). When examining the relationship between patent counts and publication output, embryo-derived stem cells demonstrated a moderate positive correlation (R2=0.461), indicating a limited concordance between academic research activity and IP generation. In contrast, iPSC exhibited a strong positive correlation (R2=0.879), suggesting that increases in scientific publications were closely aligned with growth in patent filings. Adult stem cells showed the highest correlation coefficient (R2=0.945), reflecting a very strong association between research productivity and patent activity. These results indicate that ASCs and iPSC technologies display a tighter linkage between scientific output and commercialization-related patent development, whereas embryo-derived stem cells exhibit comparatively weaker translational coupling. This pattern suggests differences in the efficiency with which research activity is converted into IP across stem cell types. Among these, ASCs exhibited a very strong positive correlation (R2=0.945) between patent filings and published articles, indicating a tight link between technological development and scientific output (Fig. 4A).
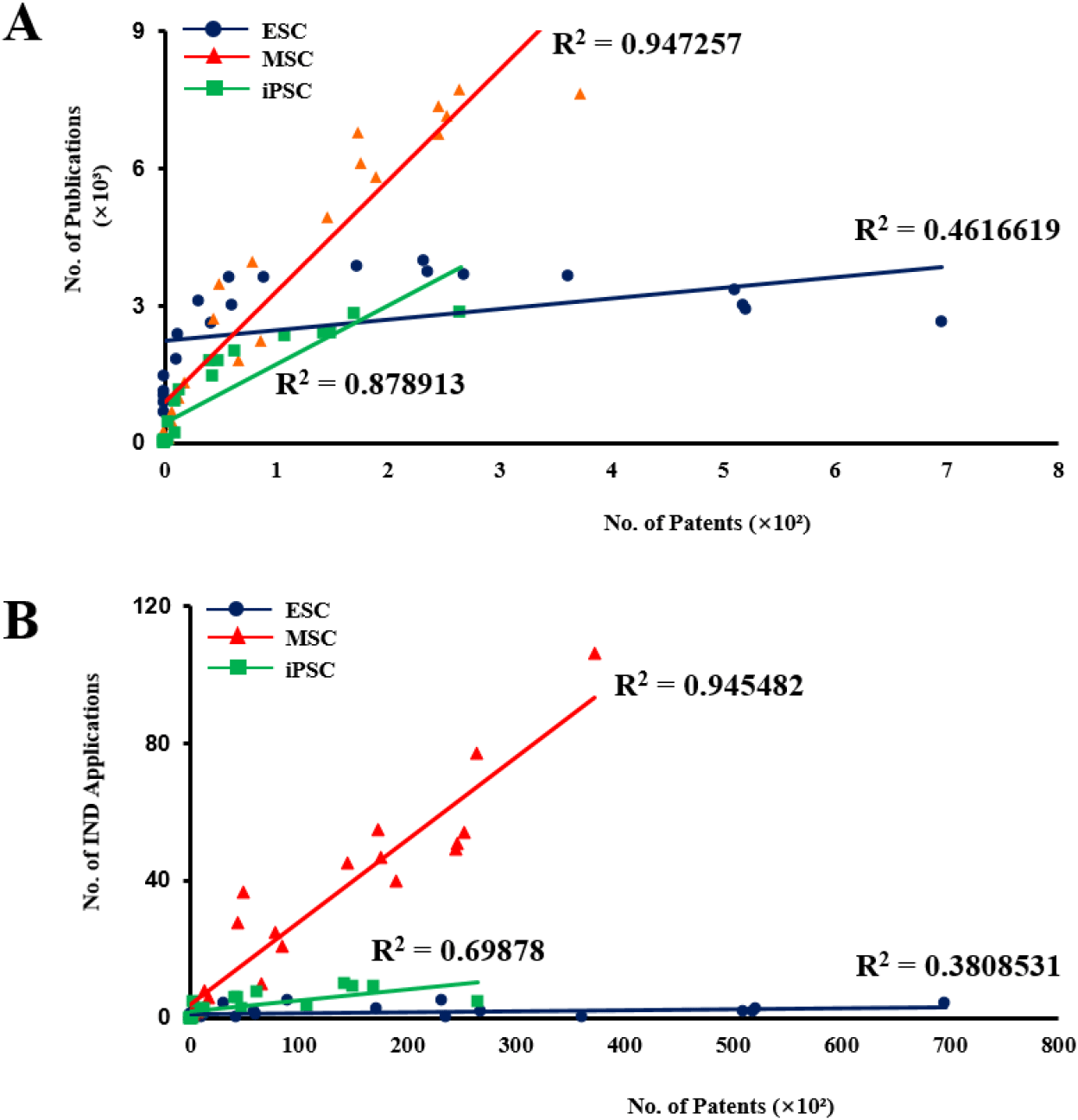
Next, the relationship between patent activity and registered clinical trials further highlighted differences in translational coupling. Embryo-derived stem cells showed a weak to moderate correlation (R2=0.381), while iPSC demonstrated a moderate association (R2=0.699), indicating partial linkage between technological development and clinical progression. Notably, ASCs again presented a very strong positive correlation (R2=0.945), demonstrating a robust connection between patent generation and advancement to clinical-stage development (Fig. 4B).
Taken together, these findings reveal clear disparities among stem cell types in the alignment of scientific productivity, IP development, and clinical translation. Adult stem cells consistently showed the strongest correlations across all analytical dimensions, suggesting a tightly integrated research-to-commercialization pathway. In contrast, embryo-derived stem cells exhibited comparatively weaker coupling, indicating potential translational or regulatory barriers limiting the conversion of research activity into clinical and commercial outcomes. Notably, for ASCs, increased patent activity appears closely associated with both higher numbers of publications and clinical trials, suggesting a greater likelihood of successful technology commercialization in this sector.
DISCUSSION
In the development of stem cell therapeutics, patents play a pivotal role in securing technological protection, enabling commercialization, and establishing exclusive rights associated with clinical research. The scope of patents in this field encompasses treatment methods utilizing stem cells, culture conditions, and genetic modification technologies. Through such IP, developers can differentiate their technologies and attain a competitive, potentially exclusive, position in the regenerative medicine and stem cell therapy market (Takahashi et al., 2023; Adedokun et al., 2024). Building a comprehensive patent portfolio covering key stem cell–related technologies—including ESC, iPSC, and mechanisms of cellular regeneration and differentiation—is critical for establishing technological leadership across diverse application areas. A strategically structured portfolio allows organizations to anticipate emerging therapeutic trends and secure a first-mover advantage in novel regenerative approaches. Moreover, patents provide a foundation for commercialization, facilitating collaboration with industry partners and research institutions through joint development and clinical research initiatives. Technology transfer agreements and licensing strategies not only generate revenue streams for patent holders but also accelerate the translation of stem cell innovations from the laboratory to clinical and market applications (Hernández-Melchor et al., 2023).
Stem cell–related patent technologies represent a critical foundation for commercialization, offering IP protection, technological differentiation, and exclusive rights for clinical research. Patents cover diverse areas, including cell-based treatment methods, culture and differentiation conditions, and genetic modification strategies. Building a robust patent portfolio encompassing ESC, iPSC, adult MSC, and associated differentiation or regeneration processes is essential for securing technological leadership and preemptively establishing competitive advantages in emerging therapeutic fields. Patents also facilitate commercialization through partnerships with industry and research institutions. Technology transfer and licensing agreements enable the translation of patented technologies into clinical applications while generating revenue streams and accelerating market entry (Wu et al., 2015; Ferguson & Kaundinya, 2020). Following clinical validation, stem cell therapeutics have demonstrated significant commercialization potential across multiple disease areas. HSC transplantation has been successfully commercialized for hematologic malignancies such as leukemia. In the MSC field, therapeutic development is advancing for cardiovascular diseases, osteoarthritis, and neurological disorders, with several products progressing through late-stage clinical trials. iPSC-based therapies, particularly in ophthalmology, have entered early clinical research in Japan, targeting retinal diseases under frameworks established by Shinya Yamanaka’s team, positioning the country at the forefront of iPSC translational research.
Despite these successes, challenges remain, including high manufacturing costs, regulatory requirements, and safety concerns such as tumorigenicity and immune rejection. Continued innovation in production methods, genome editing, and differentiation control, coupled with strategic IP management, is expected to accelerate the clinical translation and commercialization of stem cell therapeutics worldwide. In particular, ESC-based therapies face additional constraints. The process of deriving stem cells from embryos raises substantial ethical concerns in certain jurisdictions (Plomer et al., 2008). Moreover, ESC carry a relatively high risk of tumorigenicity due to their pluripotent nature, leading to safety concerns. As a result, regulatory restrictions and ethical debates significantly limit research activity and commercialization efforts for ESC-based therapies in several countries (Kirkeby et al., 2025b). These challenges have contributed to delays in regulatory approval and broader market adoption. Therefore, the development of stem cell–based therapeutics continues to face substantial technical challenges and regulatory hurdles, both of which critically influence clinical translation and successful commercialization. Overcoming these barriers will be essential to enable broader adoption of stem cell therapies in clinical practice.
Stem cell–related patent technologies are expected to serve as a key foundation for commercialization in emerging areas such as personalized medicine, cell regeneration, and combined stem cell–gene therapy approaches. Patent-driven therapeutic development holds significant promise, as advances in cell culture systems, genome editing, and controlled differentiation methods provide innovative strategies for treating a wide range of diseases (Bergman et al., 2007). Nevertheless, major obstacles remain, including high manufacturing costs, safety concerns such as tumorigenicity and immune rejection, and ethical considerations, particularly depending on the stem cell source. Addressing these challenges through continued technological innovation, coupled with strategically structured patent portfolios, will be crucial for achieving successful commercialization. Globally, the stem cell therapeutics sector is highly competitive (Hernández-Melchor et al., 2023). Leading countries are advancing distinct technological platforms, while biotechnology and pharmaceutical companies are actively leveraging IP strategies to strengthen research and development efforts, secure competitive advantages, and drive market entry (Malik, 2021). In this context, patent-based innovation not only underpins scientific progress but also shapes the translational pathway from laboratory discoveries to clinically approved stem cell therapies.
In addition, stem cell patents and clinical trials are closely interconnected in the development of stem cell therapeutics. Patents provide technological protection for stem cell–based therapies and serve as a key mechanism for enabling commercialization, whereas clinical trials represent the critical stage in which the safety and efficacy of the patented technology are validated in humans. In this way, patents and clinical trials function in a complementary and mutually reinforcing manner throughout the therapeutic development process. The primary role of patents in stem cell therapeutic development is to secure technological protection and exclusive rights, thereby facilitating commercialization. Patented technologies can form the basis for licensing agreements or strategic partnerships, allowing companies to recover research and development investments, offset manufacturing costs, and establish a competitive, potentially exclusive market position. Moreover, during the clinical trial phase, patents provide legal protection for investments made in research and development. Technologies safeguarded by patents can advance through clinical trials to demonstrate safety, efficacy, and long-term therapeutic benefit, ultimately supporting regulatory approval and market entry (Hernández-Melchor et al., 2023). In this context, clinical validation not only fulfills regulatory requirements but also enhances the legal and commercial value of patented technologies, ensuring that stem cell therapeutics can effectively transition from innovation to successful commercialization.
Within the context of patent-based stem cell therapeutic development, clinical trials represent the stage at which the safety and efficacy of therapies are rigorously evaluated in patients (Enosawa, 2022). They provide critical evidence to determine whether patented technologies possess genuine therapeutic potential. Through clinical testing, treatment effects, adverse events, and unanticipated outcomes are systematically assessed, enabling patented technologies to establish their viability as clinically applicable products. Phase I trials primarily focus on safety, assessing whether a stem cell therapy can be administered to humans—including patients and, in select cases, healthy volunteers—without causing unacceptable risks. At this stage, the stability of the patented technology and consistency of the manufacturing process are closely monitored. Phase II trials evaluate therapeutic efficacy, requiring that the patented stem cell technology demonstrates measurable clinical benefit for targeted indications. Phase III trials, which are large-scale confirmatory studies, follow successful safety and efficacy validation. These trials examine broader patient populations, assess long-term safety, and compare outcomes against placebo controls. The data generated during Phase III are essential for regulatory submissions and market authorization. Consequently, the interplay between patents and clinical trials directly shapes trial design, regulatory strategy, and commercialization pathways. The validation of a technology’s novelty and therapeutic efficacy through clinical trials reinforces its patent-protected value and facilitates advancement toward commercialization. Regulatory approval from agencies such as the FDA and the European Medicines Agency (EMA) is contingent upon these clinical outcomes, underscoring the pivotal role of clinical trials in transforming patented stem cell technologies into approved therapeutics (Riaz et al., 2024).
The relationship between patents and clinical trial outcomes also encompasses the protection and strategic utilization of clinical evidence. When a stem cell therapy demonstrates statistically and clinically meaningful results in human trials, patent protection for the underlying technology serves to prevent competitors from developing or commercializing similar therapies using the same technological foundation (Takahashi et al., 2023; Wang et al., 2024). In this manner, patents establish legal barriers to entry and provide economic protection for investments in research and development. Positive clinical trial outcomes further enhance the value of patents. Demonstrated therapeutic efficacy and safety increase the market potential and feasibility of commercialization, which in turn strengthens the commercial significance of the associated patent portfolio. This reinforces the strategic importance of IP in stem cell therapeutic development. Consequently, aligning patent strategy with clinical development confers tangible benefits for biotechnology companies and research institutions. Clinical validation of safety and efficacy not only supports regulatory approval but also facilitates investor confidence, attracts funding, and enhances corporate valuation. In this context, patent-protected stem cell technologies function as critical intangible assets, serving simultaneously as legal safeguards and as key drivers of enterprise value, market positioning, and long-term competitive advantage.
Clinical trial outcomes play a decisive role in regulatory approval for stem cell therapeutics (Musyuni et al., 2025). Regulatory authorities rely on clinical data to assess safety and efficacy, forming the foundation for market authorization decisions. When a stem cell therapy is developed from patented technology and demonstrates favorable clinical results, the regulatory review process may proceed more efficiently due to the clear demonstration of technological ownership, controlled manufacturing processes, and documented innovation (Hussen et al., 2024). Conversely, the occurrence of unexpected adverse events or insufficient therapeutic efficacy during clinical trials can substantially limit the commercialization potential of the underlying patented technology. Challenges in demonstrating safety and efficacy may delay or prevent regulatory approval, directly impacting the market viability of patent-based therapies. Regulatory standards for stem cell therapeutics are particularly stringent. While patents are essential for protecting commercialization rights and securing legal exclusivity, they do not guarantee regulatory approval. Failure to meet regulatory requirements can impede not only clinical research but also subsequent commercialization efforts. Moreover, stem cell therapeutic development requires significant financial investment, with clinical trials representing a major cost driver. Weak, uncertain, or commercially ambiguous patent protection can increase the risk of failure during clinical development. In such cases, uncertainty regarding patent scope and commercialization prospects may deter investment, thereby reducing the likelihood of successful translation from patented technology to approved therapy. Furthermore, the limitations of the present study is that the R&D cycle for biotherapeutics including stem cells is widely recognized to require a substantial period, typically ranging from 10 to 20 years. Moreover, a comprehensive analysis of commercialization pathways leading to clinical trials for individual patents would necessitate an extensive evaluation of patent-based technology transfer outcomes. Given these analytical constraints, we examined the correlation between patents and clinical trials according to stem cell type, restricting the analysis to an limited periods containing five-year period, which represents the minimum estimated timeframe for transition from patent filing to clinical trial initiation. To better understand the factors influencing the clinical translation and commercialization of stem cell-related patents, comprehensive analyses incorporating temporal trends and other relevant variables will be required.
CONCLUSION
Stem cell therapeutics represent a highly promising area in regenerative medicine, with significant potential for tissue repair and treatment of intractable diseases. Despite this potential, commercialization faces persistent challenges, including high manufacturing costs, safety concerns, ethical debates, and complex regulatory requirements. Nevertheless, research publications and technology-based patent filings aimed at addressing these barriers have continued to grow. In this study, we analyzed the characteristics of representative stem cell–related patents across major stem cell types. We further evaluated the correlations among patent activity, scientific publications, and clinical trial numbers, considering factors that influence therapeutic development feasibility. This approach enabled the assessment of commercialization potential in each stem cell category. Our findings reveal distinct differences among stem cell types in the alignment between technological innovation, scientific output, and clinical translation, highlighting key indicators for predicting successful commercialization. Specifically, ASCs demonstrated strong correlations between patent filings, publications, and clinical trial activity, suggesting a higher likelihood of market translation, whereas embryonic and iPSCs exhibited more modest correlations, reflecting ongoing technical and regulatory hurdles. These results indicate that overcoming limitations—particularly through scalable production methods, cost reduction strategies, and long-term safety validation—will be critical to accelerating the clinical and commercial adoption of stem cell therapeutics. Overall, the study provides insights into strategic patent and research planning for advancing stem cell–based therapies from innovation to clinical application.

