INTRODUCTION
Epinephelinae contains 159 species in 15 genera and generally inhabits rocky or coral subtropical and tropical regions (FAO, 1993). There are 11 species of grouper inhabit the Southern Sea near Jeju Island in Korea, including Epinephelus bruneus, E. septemfasciatus, E. akaara and E. fasciatus (Kim et al., 2001). Groupers are commercially important species in Southeast Asian countries including Korea. Recently, grouper resources decreased rapidly due to overfishing in Jeju coastal waters. Thus, there are many studies on artificial seed production of groupers in several Asian countries including Korea. Previous research on the E. bruneus has been conducted as follows: spawning behavior and ontogeny development in indoor rearing tank (Manabe & Kasuga, 1988; Yang et al., 2007), larvae's environmental tolerance, growth, and morphological development (Okada et al., 1996; Sawada et al., 1999), the effectiveness of water temperature on the growth, feeding of juvenile (Inoue, 2001), seed production (Lee et al., 2008) and pubertal characteristics (Ryu et al., 2013).
Epinephelinae has sex characterization of protogynous hermaphrodism in that all fish sex differentiate and function as a female, and then some fish reverse sex to male as they age and grow. The grouper’s natural sex reversal has been found in an E. tauvina (Chao & Lim, 1991) showing it took seven years to have natural sex reversal after hatching. Another study on E. costae showed 11.0 kg in body weight fish have natural sex reversal (Glamuzina et al., 2000). As for the E. akaara (Hamamoto et al., 1986), the fish longer than 25.0 cm in total length and heavier than 500.0 g in body weight has an increased ratio of males. Due to their sex characteristics in nature, capture of mature male is difficult and it is the most significant constraint to artificial seed production of grouper. Thus, the sex reversal technique is being used to produce functional males by injecting androgen. Previous experiments attempted masculization in groupers through hormonal manipulation. In E. tauvina (Chen et al., 1977; Chao & Chow, 1990), E. septemfasciatus (Tukashima & Kitajima, 1983; Tsuchihashi et al., 2003; Song, 2004), and E. akaara (Hwang et al., 1998), functional males were obtained when exogenous androgen or 17α-methyltestosterone (MT) was administered. In E. merra (Alam et al., 2006), an aromatize inhibitor, or anendogenous hormone, was used to induce masculinization. However, more research is needed whether the functional males remained male after the maturation season or if they revert back to be females.
In this study, we investigated induced sex reversal and sex revert of E. bruneus reared in indoor artificial rearing condition to obtain functional males for fertilization egg production and seed production via observation of the treatment of 17α-methyltestosterone (MT).
MATERIALS AND METHOD
From September 2002 to December 2005, 64 wild caught E. bruneus (total length 65.6±13.1 cm, body weight 4.7±4.0 kg) were used in this study. Specimens were transported to the Ocean and Fisheries Research Institute in Korea’s Jeju Province and maintained in 6 inner concrete rearing tanks (two tanks were 5.0×5.0×3.0 m; 62 m3, and four tanks were 4.0×4.0×2.5 m; 36 m3). Specimens were individually marked via insertion of micro ID tag into the dorsal skin (ID tag, Trovan Ltd. UK). Specimens were fed frozen horse mackerel twice a week and were fed a mixture of frozen horse mackerel and squid 2 to 3 months before the maturation season.
We examined the sex ratio depending on the body weight and sex reversal of E. bruneus with ID tags reared in indoor rearing tanks. The sex division was judged by cannulation between June and August. Female E. bruneus were identified when the oocyte diameter in the gonad is larger than 200 μm, males showing spermiation when pressed on the abdomen, and unidentified when showing neither ovulation nor spermiation. When spermiation occurred to specimens identified as female the previous year, we decided those were naturally sex reversed in the rearing tank.
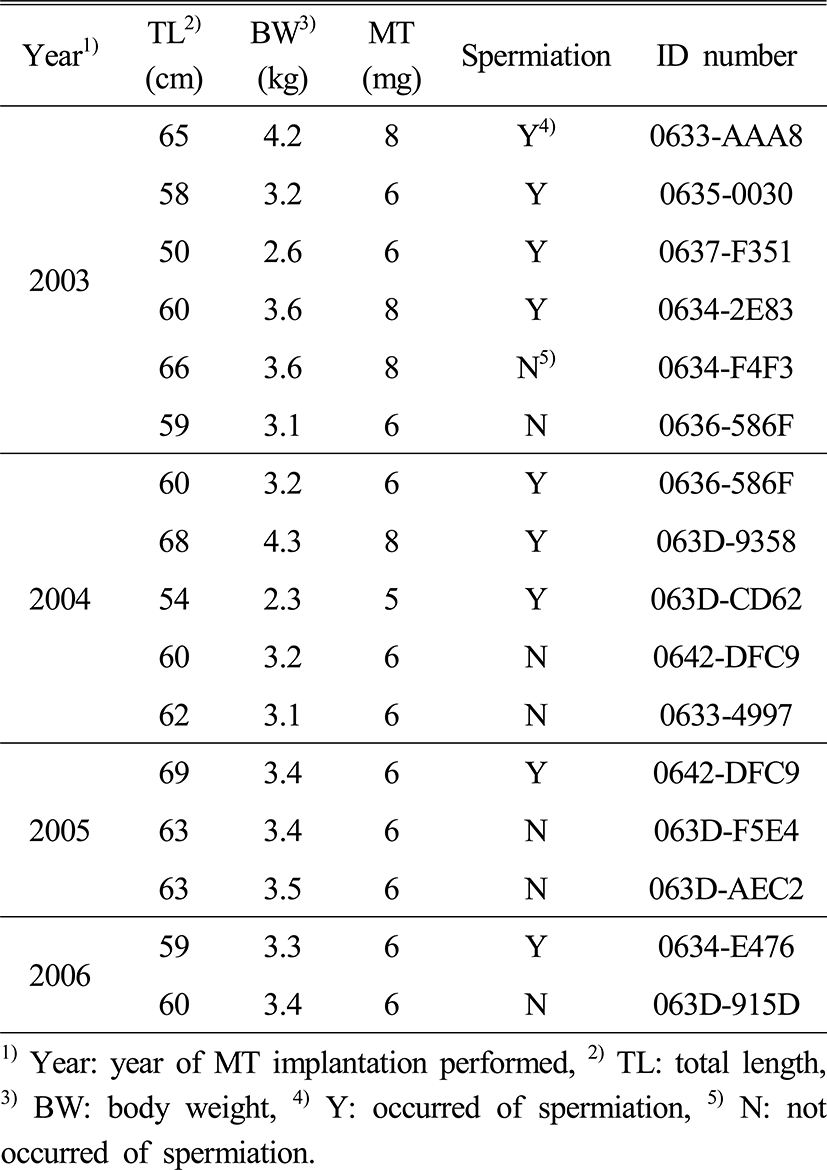
Sexually immatured E. bruneus (n=14) between 50 to 69 cm in total length (61.5±4.5 cm) and a body weight between 2.3 to 4.3 kg (3.4±0.5 kg) were used to experiment induced masculinization 4 times from 2003 to 2006. To induce masculinization via the method of Tsuchihashi et al. (2003), a silastic capsule with androgen hormone (MT, 17α-methyltestosterone, Sigma Co. Ltd, USA) was implanted into the abdomonal cavity. MT (1 mg MT/10 μl) in the silastic capsule was produced by adding castor oil (Junsei Chemical Co. Ltd., Japan) 800 μl to 100 mg of MT dissolved in 200 μl of 95% ethanol. The lower abdominal underneath the pectoral fin was cut about 0.5 cm to inject the silastic capsule after anesthetizing with 2-phenoxyethanol (Sigma Co. Ltd, USA) 300 ppm, and the wound sealed after-wards. We examined the success of artificial sex reversal with a histological analysis and external observation by a method of seeing if spermiation occurs when pressing the abdomen in 2003. From 2004 to 2006, we judged sex reversal with external observation described above. For the histological analysis, gonads were dissected from abdominal cavity, and fixed in Bouin’s solution before the silastic capsule implantand 8 weeks after the implant. The samples were dehydrated in a graded series of ethanol, embedded in paraffin and then cut into 5 μm cross section. Slides were stained with Hansen's hematoxylin and 0.5% eosin then observed using a light microscope (HBO 50, Carl Zeiss). For external observation, we judged if masculinization was successful through observing spermiation by pressing the abdomen between 8 and 14 weeks after the silastic implant.
Once specimens were identified as functional males, they were tested a year after the MT treatment. The spermiation for specimen was observed between June and August in the maturation season by the abdomen pressing method and the cannulation method. Revert to females among masculinized specimen was determined that oocytes could be obtained by cannulation or ovulation occurred after HCG treatment.
RESULTS
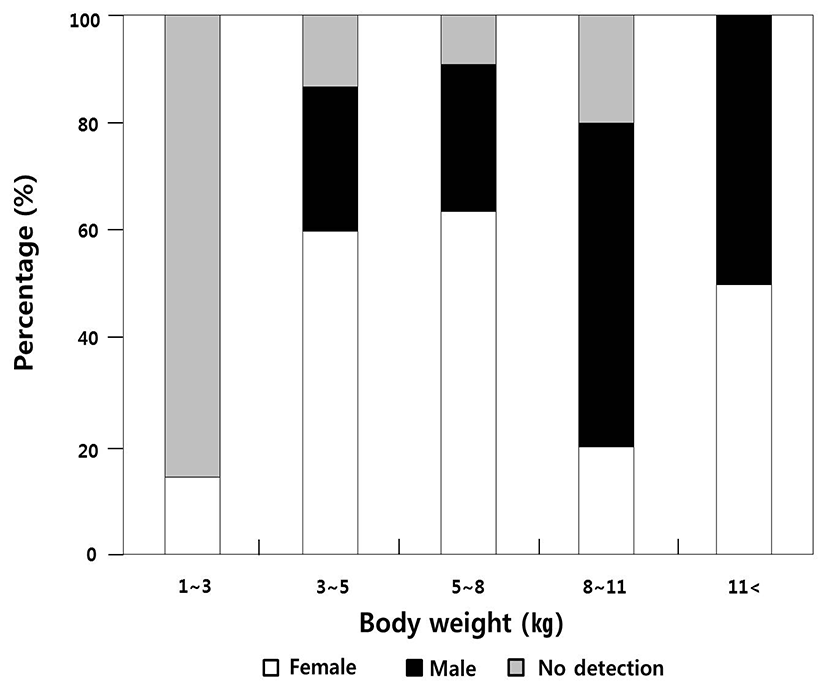
Body weight between 1.0 to 3.0 kg, had 1 female (14.2%), 0 male (0%), and 6 indiscernible (85.7%) from a sample of 7 fish. Body weight between 3.0 to 5.0 kg, had 9 females (60.0%), 4 males (26.6%) and 2 indiscernible (13.3%) from a sample of 15 fish. Body weight between 5.0 to 8.0 kg, had 7 females (63.6%), 3 males (27.2%), 1 indiscernible (9.0%) from a sample of 11 fish. Body weight between 8.0 to 11.0 kg, had 1 female (20.0%), 3 males (60.0%), 1 indiscernible (20.0%) from a sample of 5 fish. Body weight more than 11.0 kg, had 1 female (50.0%), 1 male (50.0%) from a sample of 2 fish (Fig. 1, Table 1).
|
For the sex reversal in indoor rearing condition, we obtained 1 sex-reversed male (total length 99.0 cm, body weight 13.2 kg) from a sample of 5 females bought in 2002. We obtained 5 sex-changed males out of 23 females bought in 2003. It occurred in a different time period, 1 female (total length 63.0 cm, body weight 4.4 kg) in 2004, 1 female (total length 72.0 cm, body weight 5.0 kg) in 2005, 3 females (total length 77.0 cm, body weight 6.7 kg; total length 77.0 cm, body weight 7.0 kg; total length 78.0 cm, body weight 7.3 kg) in 2006. In 2005, we obtained 1 sex-reversed male (total length of 70.0 cm, body weight of 7.2 kg) from a sample of 22 females that were bought in 2004. Sex reversal was observed when fish were 63.0 to 99.0 cm total length and 4.4 to 13.2 kg body weight, but most of sex reversal occurred in 5.0 to 8.0 kg body weight.
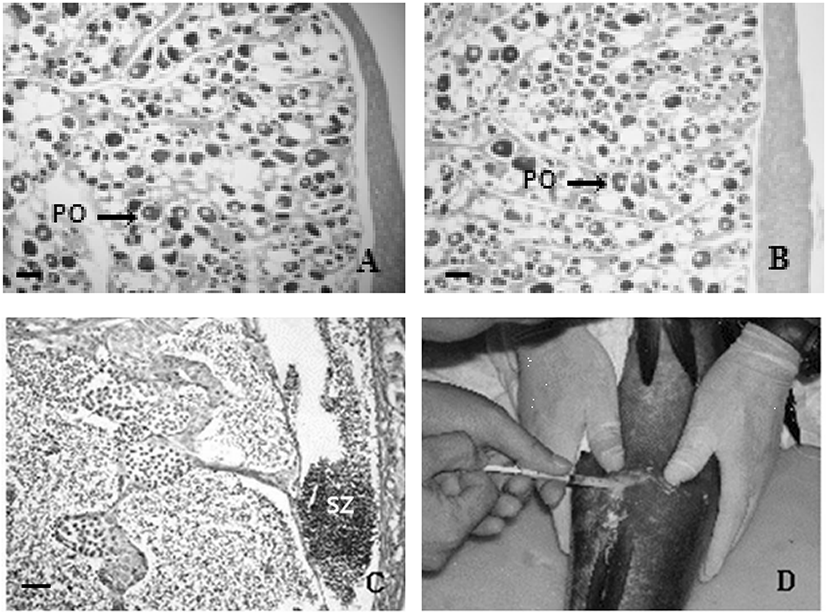
At the onset to the experiment in March 2003, the gonad of E. bruneus mainly contained perinucleous oocyte (Fig. 2A). 8 weeks after the experiment, the control group had gonad with perinucleolus oocyte (Fig. 2B) but the gonad of MT treated had developed into testes, which the lumen of lobule and efferent duct was filled with sperms (Fig. 2C). 14 weeks after implant silastic capsule, we collected sperm by pressing the abdomen and were able to collect again 17 days later (Fig. 2D). The following is the yearly amount of induced functional males by silastic capsule that showed spermiation: 4 out of 6 in 2003, 3 out of 5 in 2004, 1 out of 3 in 2005, 1 out of 2 in 2006 (Table 2).
|
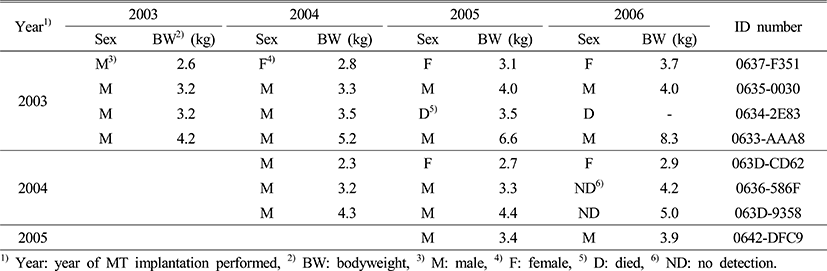
Two (2.3 and 2.6 kg) out of 8 sex-reversed males (2.3 to 4.3 kg) with artificial masculinization during 2003 to 2005 reverted back to being female and ovulated. One out of 4 sex-reversed males that had artificial masculinization in 2003 has reverted to female in 2004, and ovulated with HCG treatment in 2006. One out of 5 sex-reversed males that had artificial masculinization in 2004 reverted back to being female in 2005, and ovulated with HCG treatment in 2006 (Table 3).
|
DISCUSSION
Most groupers are protogynous hermaphrodites, which change sex depending on the rearing condition such as age, size and social control. In the different size E. coioides of the same rearing tank, the bigger fish changed into males, while the smaller ones remained female (Quinitio et al., 1997). Each species had a different period for the sex reversal. E. morio had sex reversal when 9 years old and weighing 11.0 kg, E. marginatus when 9 to 16 years old and weighing 6.0 to 10.0 kg (Chauvet, 1988; Randall & Heemstra, 1991), E. septemfasciatus when weighing more than 6.0 kg (Tshchihashi et al., 2003), and E. malabaricus when 5 years after hatching and weighing more than 6.0 kg (body length 60.0 to 70.0 cm) (Tang et al., 1979; Tschihashi et al., 2003). From this study, inner tank reared E. bruneus reversed sex from female to male when their body length was 63.0 to 99.0 cm and their weight was 4.4 to 13.2 kg. In the male population according to the body weight, 5 out of 32 (less than 5.0 kg body weight) were male and 10 out of 18 (5.0 kg or more body weight) were male.
Since groupers, such as E. septemfasciatus and E. marginatus reverse their sex at a large size, it is difficult to obtain sex-reversed males in the rearing tank for those fish. To solve this problem, many studies are being done to induce small sized females to have sex reversal with androgen treatment (Chen et al., 1977; Tukashima & Kitajima, 1983; Chao & Chow, 1990; Tsuchihashi et al., 2003). Sex reversal in groupers induced by androgen administration is more common, but various dose concentrations and methods of hormone administration. In E. tauvina, sexreversed males were obtained when a dose of 145.0 mg MT for a year and MT 120.0 mg/kg • BW for seven months was given orally, and in E. septemfasciatus males sex-reversed when a dose of MT 1.0 mg/kg • BW once a day for 73 days was given orally (Chen et al., 1977; Tukashima & Kitajima, 1983; Chao & Chow, 1990). For the abdominal cavity or muscle injection methods, sex reversal occurred when MT 30.0 mg/kg • BW was injected 6 times every other week to E. coioides (Chao & Lim, 1991). E. suillus were induced to functional males when injected with a MT 0.5 mg/kg • BW silastic capsule over 4 months (Tan-Fermin et al., 1994). E. septemfasciatus had spermiation 2 months after a MT 1.0 mg/kg • BW and 4.0 mg/kg • BW silastic capsule was implanted and was induced to have sex reversal when injected with MT 0.5 to 2.0 mg/kg • BW and a MT 1.0 to 2.0 mg/kg • BW silastic capsule implanted into the abdomen (Tsuchihashi et al., 2003; Song, 2004). E. merra were induced to functional males after 24 days injecting an aromatase inhibitor fadrozole 1.0 mg/kg • BW with a silastic capsule during the maturation season (Alam et al., 2006). For E. bruneus, we obtained functional males from the 12 weeks of implant a MT 2.0 mg/kg • BW silastic capsule into the abdomen and was able to identify spermiation in 9 out 14 fish. In some specimen, we did not produce sex-reversed males due to the silastic capsules slipping out of the fish.
Two sex-reversed males reverted back to female 2 to 3 years after the hormone treatment. It was due to the aspect of hormone treatment for immature fish (body weight 2.3 to 2.6 kg). We suggested that immature fish had androgen with MT silastic capsule for a while, and they reverted back to female because there is no effect or low concentration level of MT treatment. This result corresponds to the result of an experiment where MT treated sex-reversed males did not remain as males and reverted back to being females during the next maturation period (Marino et al., 2000; Sarter et al., 2006). These results show that masculinization in E. bruneus with MT treatment is more effective for fish weighing more than 3.0 kg to have sex-reversed males remain as functional males.

