INTRODUCTION
Carp and salmon gonadotropic hormones, human chorionic gonadotropin, and synthetic gonadotropin releasing hormone agonists (GnRHa) are used to artificially accelerate final oocyte maturation (FOM) and induce ovulation in fish species that have high failure rates of natural ovulation. Steroid hormones have also been used to induce FOM. However, the influence of protein hormones varies depending on the type, administration time, and fish species, whereas excess administration of steroid hormones can have a suppressive effect on FOM. Therefore, it is necessary to obtain endocrinological data about FOM and ovulation of the target fish species to successfully induce ovulation using gonadotropins. In addition, the appropriate concentration and administration time of the gonadotropic hormones must be determined. However, it is desirable to obtain fertilized eggs from natural spawning, as the quality of artificially induced eggs may not be as good as naturally spawned eggs. Thus, basic studies on environmental factors need to be conducted.
Early studies explored the effects of environmental factors on FOM and were based on similar studies on the effects of photoperiod on the mating seasons of birds, sheep, and deer. Experiments have been conducted to identify the impact of environmental factors on FOM and spawning of salmon and trout (An, 1995). The common methods to control fish spawning include control of water temperature and photoperiod as well as use of hormone treatments. One study assessed changes in plasma steroid hormones in rockfish, Sebastes inermis by controlling water temperature and photoperiod (Chang et al., 2001). Another study was conducted on the effects of photoperiod and water temperature on the reproductive cycle of Korean rose bitterling, Rhodeus uyekii, and another was conducted on the impact of water temperature and photoperiod on gonadal development in the banded catfish, Pseudobagrus fulvidraco (Lim & Han, 2012). However, studies on red marbled rockfish, Sebastiscus tertius have only been based on the reproductive cycle and not on the effect of water temperature and photoperiod in relation to FOM. Therefore, we assessed the influences of water temperature and photoperiod on FOM in S. tertius to provide basic data for future studies related to the fish’s reproductive cycle.
MATERIALS AND METHODS
Fifty-five adult S. tertius, which were bred in a natural habitat, were purchased in January–February 1999 and acclimated for 25 days in an indoor circular plastic water tank (500 L) together with 180 farmed fish. Then, 15 females (total length, 299.9±6.2 cm; weight, 593.7±417.5 g) and five males (total length, 29.2±5.3 cm; weight, 314.6± 315.5 g) adults were placed in a single tank.
The experiment was conducted with various combinations of water temperature (15 and 19°C) and artificial photoperiods (9L:15 D, 12L:12D, and 15L:9D) using natural light. The experiments were 15 females and 5 males each tank during 50 days and repeated twice. We used florescent lamps of 25 W as the artificial light source, which were controlled by timers. Water temperature was controlled by thermostated heaters (500 W). Breeding water depth was approximately 45 cm. Live feed including krill shrimp was provided to satiation at 10 AM every day. Water quality was checked every 5 days by measuring dissolved oxygen, pH, and ammoniumnitrogen. Ovary development stage was perinucleolus and granules accumulated in cytoplasm at the started experiment.
RESULTS
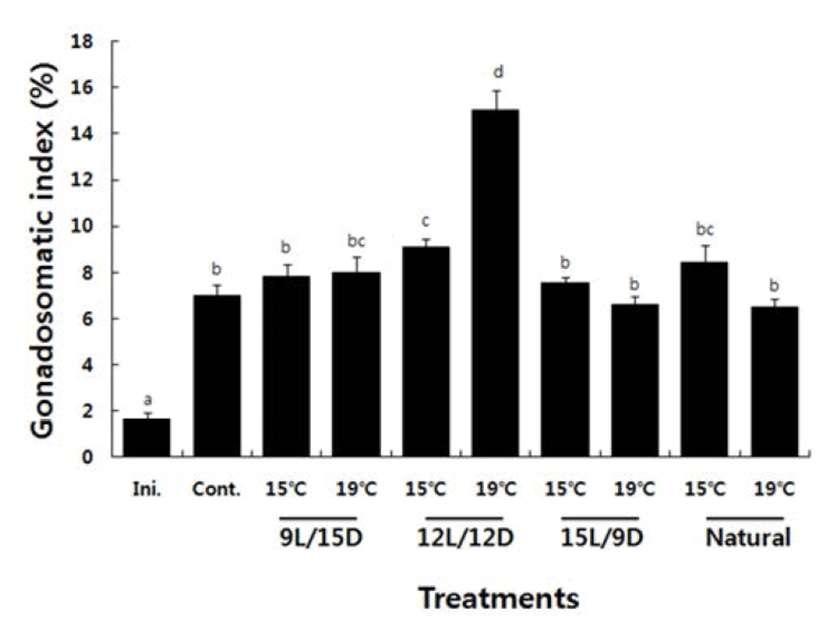
The GSI at the beginning of the experiment in females was 1.62±0.32, which increased to 7.01±0.46 on day 50 at the end of the experiment. The GSI in the 15°C groups increased to 7.80±0.54 at the end of the experiment under a photoperiod regimen of 9L:15D to 9.10±0.35 under a photoperiod of 12L:12D, to 7.54±0.25 under a photoperiod of 15L:9D, and 8.44±0.74% under natural light. The photo-period regimen of 12L:12D resulted in the highest GSI among the 15°C water temperature groups. The GSI in the 19°C groups increased to 8.01±0.65 at the end of the experiment under a 9L:15D photoperiod, to 15.03±0.86 under a 12L:12D photoperiod, to 6.75±0.36 under a 15L: 9D photoperiod, and to 6.75±0.36% under natural light. The photoperiod regimen of 12L:12D resulted in the highest GSI among the 19°C water temperature groups (Fig. 1).
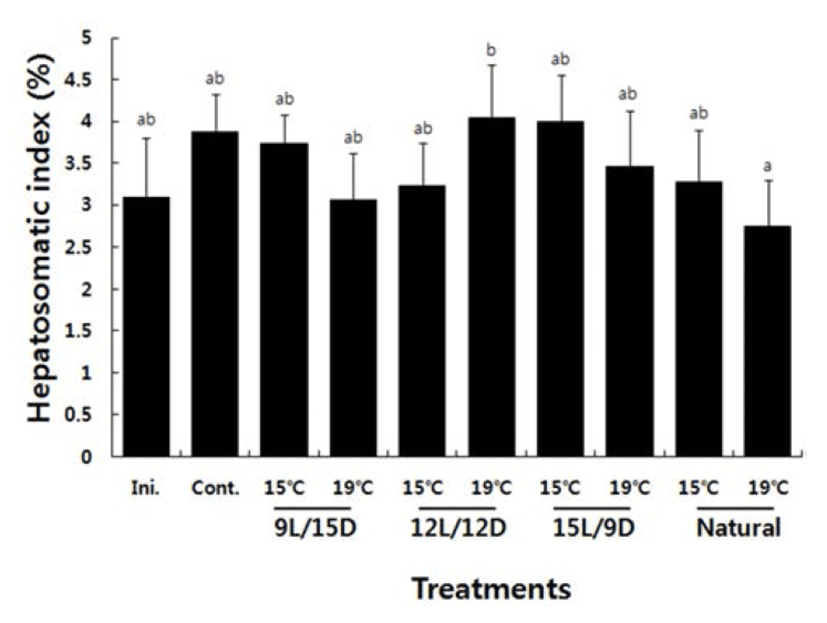
The HSI in the control group was 3.09±0.72 at the beginning of the experiment, which increased to 3.88± 0.45 on day 50 at the end of the experiment. The HSI in the 15°C water temperature groups increased to 3.73±0.35 at the end of the experiment under a 9L:15D photoperiod, to 3.23±0.52 under a 12L:12D photoperiod, to 4.00±0.56 under a 15L:9D photoperiod, and to 3.27±0.62% under natural light. The 15L:9D photoperiod resulted in the highest HSI among the 15°C water temperature groups. The HSI in the 19°C water temperature groups increased to 3.06± 0.52 at the end of the experiment under a 9L:15D photoperiod, to 4.05±0.63 under a 12L:12D photoperiod, to 4.05 ±0.63 under a 15L:9D photoperiod, and to 2.75±0.54% under natural light. The 12L:12D photoperiod resulted in the highest HSI among the 19°C experimental water temperature groups (Fig. 2).
DISCUSSION
We conducted a series of experiments during the spawning season in March to identify the effects of environmental factors such as water temperature and photoperiod on the reproductive cycle of S. tertius. Environmental factors such as water temperature and photoperiod play a significant role in the reproductive cycle of fish (De Vlaming, 1975; Scott, 1979; Asahina & Hanyu, 1983; Aida, 1991). The spawning seasons of fish can be classified into six categories: spring spawning, spring-summer, summer, springfall, fall, and winter spawning types (Aida, 1991). The most freshwater fish were induced gonadal maturation and gonadal active as water temperatures rise in the spring, along with photophase increases facilitate compensation effect (Asahina & Hanyu, 1983; Lim & Han, 1995), but, bluegill, Lepomis macrochirus was effected two factors rising water temperature and long photophase (Lee & Kim, 1987). In seawater fish, maturation of spring-summer type small filefish, Rudarius ercodes began by long photophase, along with water temperature compensation, and effect of water temperature in red sea bream, Pagrus major was reported for ovary development (Yoo et al., 1998). Also, spawning induction reported in olive flounder, Paralichthys olivaceus, rock bream, Oplegnathus fasciatus and red sea bream according to water temperature and photoperiod (Kim & Hur, 1991; Kim & Kim, 1990; Jeong et al., 1998; Ko et al., 1998). According to Lim et al. (2011), the spawning season of S. tertius spans from March to May. Thus, this species is classified into the spring spawning type, based on the yolk globule stage when oocytes appear at early maturation. This study was conducted during early February under different water temperature (15 and 19°C) and photoperiod (combinations of 9L, 5L, 12L, and 12D) conditions. The GSI of females at the beginning of the experiment was 1.62±0.32, which increased to 7.01±0.46 at the end of the experiment. The GSI of females in the 15°C water temperature groups increased the most to 9.10 ±0.35 under a 12L:12D photoperiod, whereas the groups under the other photoperiods showed less of an increase in GSI than that that under natural light (8.44±0.74%). The GSI in females in the 19°C water temperature groups increased the most to 15.03±0.86% under the 12L:12D photoperiod, and groups under the other photoperiods showed a significantly greater increase in GSI than that under natural light. The GSI under the 15L:9D photoperiod was 6.75±0.36, whereas it was 6.75±0.36% under natural light. The 12L:12D photoperiod resulted in the greatest increase in GSI. We found that the HSI in the control group was 3.09 ± 0.72 at the beginning of the experiment, which increased to 3.88 ± 0.45 on day 50 at the end of the experiment. The HSI of females in the 15°C water temperature groups increased the most to 4.00±0.56% under a 15L:9D photoperiod, whereas the 19°C water temperature groups showed the highest HSI under the photoperiod of 12L:12D. Similar to the GSI results, the 19°C water temperature groups exhibited a higher HSI than that of fish under natural light. We conclude that the spawning season of S. tertius is more influenced by photoperiod than water temperature. The water temperature of the natural habit of S. tertius ranges from 13.5 to 14.0°C, which is lower than the 15°C experimental condition, and the 12L:12D photoperiod resulted in higher GSI and HSI compared to those under natural light. These results seem to water temperature and photoperiod promoted sexual maturation in S. tertius similary rock bream and red bream (Jeong et al., 1998; Kim & Kim, 1990).