INTRODUCTION
Buenos aires tetra (Hyphessobrycon anisitsi) is fresh water teleost of Characiformes Characidae, and is distributed throughout Brazil, Argentina, and Paraguay in South America. Its body length is 5~7 cm, and its lifespan is about 5~7 years. Characteristically the entire body is silver and the caudal fins are reddish.
Studies which is realated to made on Characidae in Korea include those on the fertilized eggs of Hemigrammus ocellifer, Gymnocorymbus ternetzi, and H. caudovittatus (Kim et al., 1996) and of H. serpae (Kim et al., 2005), and on the microstructure of egg membrane (Lee et al., 2008).
In other countries, antioxidant defense capacity against ammonia stress (Pan et al., 2011), the early development of skull (Vandewalle et al., 2005), embryo development (Nakaghi et al., 2013; Weber et al., 2012), etc. have been studied.
Characidae fish like Buenos aires tetra is easy for mass seed production, and because it is cheaper than other tropical fish, it is sold actively as feed for large carnivorous fish. Most of its demand from Korean consumers is supplied through import, and there are only a few producers in Korea. In this situation, more research and technology for seed production are required. Thus, this study investigated the egg development of Buenos aires tetra and the mor-phological development of larvae and juveniles in order to provide basic information for the seed production of Characidae fish.
MATERIALS AND METHODS
In May 2012, 50 mature mothers 5.55~7.32 cm in full length (mean 6.44±1.25 cm; Fig. 1) were purchased from Bizidduck Aquarium in Yeosu, Jollanam‐do, and put in a rectangular glass water tank (50×50×30 cm). In the water, pH was maintained at 6.00~6.50 (mean 6.25±0.02), and dissolved oxygen concentration at 5.94~7.10 mg/L (mean 6.73±0.29 mg/L). Breeding water temperature was maintained at 27.5~28.5°C (mean 28.0±0.05°C) using an electric heater (Warmton 50w, Korea), and clean water was maintained by removing waste and feed leftover accumulated on the floor of the tank using sponge filter (SF‐2822, Korea). The fish were fed twice a day (AM 8:00; FM 17:00), and frozen blood worms (Hikari, Japan) and formula feed (Love Larva, Japan) were used.

Fertilized eggs were put in a 500 mL glass beaker, and breeding water temperature was maintained within the range of 26.5~27.5°C (mean 27.0±0.05°C) until hatching. Half of the breeding water was changed three times a day, and egg size was measured up to 0.01 mm using a profile projector (Nikon JP V‐12B, Japan) with 20 individuals sampled at random, and the process of egg development was observed and photographed under a stereoscopic microscope (Nikon JP SMZ800, Japan).
Larvae just after hatching were put in a rectangular glass water tank (50×50×30 cm), and bred there in stagnant water, and air was supplied weakly through aeration. As feed for larvae and juveniles, rotifer (Branchionus rotundiformis) was supplied until 7 days after hatching yolk absorption and the feeding rate was 5~10 individuals/ mL. Then, Brine shrimp (nauplius of Artemia sp.; Westwood premium gold) was supplied at a rate of 10~15/ mL from Day 7 to 20, and formula feed (200~310 μm; Love Larva, Japan) was supplied together from Day 20 to 30.
For the morphological obsrevation of larvae and juveniles, 5 individuals were anesthetized with anesthetic (MS‐222, Ethyl 3‐aminobenzoate methanesulfonate, Sigma Aldrich Co., St. Louis, USA) each day after hatching, and each region of the body was measured up to 0.01 mm and observed using a profile projector and a stereoscopic microscope. The morphological stages of larvae and juveniles were divided according to Rusell (1976).
RESULTS
The form of fertilized eggs was globular without a quarter, and the color of the yolk was light red. Fertilized eggs were adhesive demersal eggs, and egg diameter was 0.63~0.91 mm (mean 0.74±0.07 mm, n=20).
The developmental stage when the fertilized eggs were collected was the mid‐gastrula stage as the germinal disk covered down up to 2/3 of the yolk (Fig. 2A), and the embryoid body began to be formed along the yolk in an hour 30 minutes after the gastrula stage (Fig. 2B).
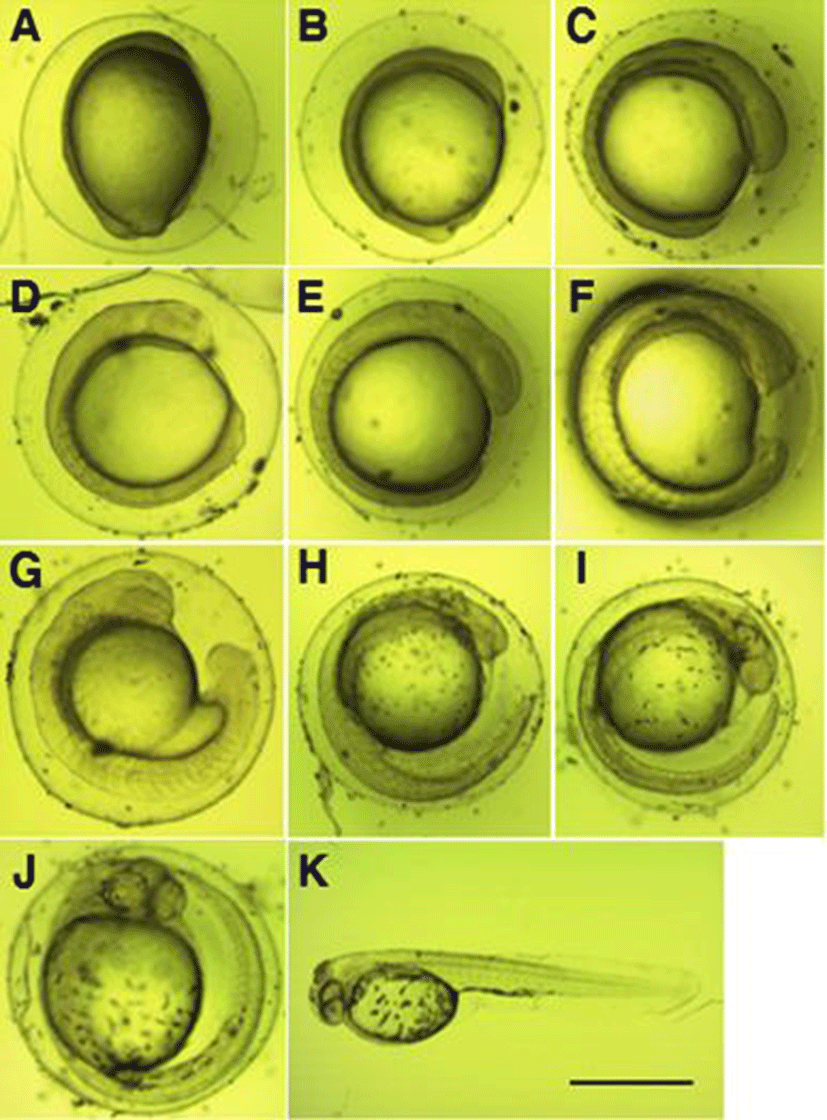
In 3 hours 53 minutes after the gastrula stage, 6 myotomes were formed on the embryoid body, and the eye vesicle began to be formed (Fig. 2C). In 4 hours 53 minutes after the gastrula stage, the number of myotomes on the embryoid body increased to 7, and the tail began to develop (Fig. 2D). In 5 hours 23 minutes after the gastrula stage, the number of myotomes on the embryoid body increased to 9, and the eyeball began to be formed (Fig. 2E).
In 6 hours 23 minutes after the gastrula stage, the number of myotomes on the embryoid body increased to 15, and Kuffer’s vesicle was formed on the base of the tail (Fig. 2F). In 7 hours 23 minutes after the gastrula stage, the number of myotomes on the embryoid body increased to 19, the tail began to be separated from the yolk, and Kuffer’s vesicle formed on the base disappeared (Fig. 2G). In 9 hours 23 minutes after the gastrula stage, speckle‐like black vesicles were deposited on the upper part of the yolk, and membrane was formed on the caudal fins and deve-loped long (Fig. 2H).
In 10 hours 53 minutes after the gastrula stage, the caudal fin extended up to the tip of the head, and the otolith developed (Fig. 2I). In 12 hours 13 minutes after the gastrula stage, black vesicles were deposited around the eyes and on the base of the tail (Fig. 2J), and in 16 hours 13 minutes after the gastrula stage, hatching through the egg membrane was started, beginning with the tail (Fig. 2K).
Fig. 4 shows the growth of larvae and juveniles. Pre‐larvae just after hatching were 3.78~3.88 mm in full length (mean 3.84±0.04 mm, n=5) and had membranous fins from the back at the center of the body to the end of the yolk, and the mouth and the anal were not opened. Black vesicles were deposited around the eyes, on the upper part of the yolk, and on the bottom of the trunk (Fig. 3A). Pre‐larvae on the 2 days after hatching were 4.01~4.24 mm in full length (mean 4.11±0.09 mm), and branch‐like black vesicles were deposited on the upper part of the head, on the back, and on the base of the tail (Fig. 3B). Post‐larvae on the 7 days after hatching were 4.11~4.58 mm in full length (mean 4.28±0.23 mm), their mouth and anal were opened, and the yolk was absorbed completely. As the swim bladder was formed inside, they began to swim, and fan‐shaped caudal fins began to develop (Fig. 3C). Post‐larvae on the 11 days after hatching were 5.30~6.13 mm in full length (mean 5.74±0.31 mm), and as their jaw developed, they began to eat feed actively. In addition, black vesicles were deposited on the upper part of the gill openings. Rays began to develop at the end of the caudal fins, and round pectoral fins developed (Fig. 3D). Post‐larvae on the 20 days after hatching were 5.94~7.15 mm in full length (mean 6.45±0.61 mm), and the number of fin rays formed was 5 in the dorsal fin, 11 in the anal fin, and 17 in the caudal fin. Speckle‐like black vesicles were deposited at the center of the caudal fins (Fig. 3E). Post‐larvae on the 24 days after hatching were 6.15~7.25 mm in full length (mean 6.76±0.52 mm), and their dorsal, anal, and caudal fins connected through membrane were separated. The ventral fins were separated but were still membranous. At this stage, the number of fin rays increased to 10 in the dorsal fin, to 14 in the anal fin, and to 20 in the caudal fin. Speckle‐like black vesicles were deposited also on the dorsal and anal fins (Fig. 3F). Juveniles on the 28 days after hatching were 6.53~8.42 mm in full length (mean 7.77±1.59 mm), and the number of fin rays increased to 11 in the dorsal fin, to 25 in the anal fin, and to 22 in the caudal fin. In addition, the caudal fins developed into the form of homocercal fin. The black vesicles deposited at the center of the caudal fins developed into large speckles (Fig. 3G).
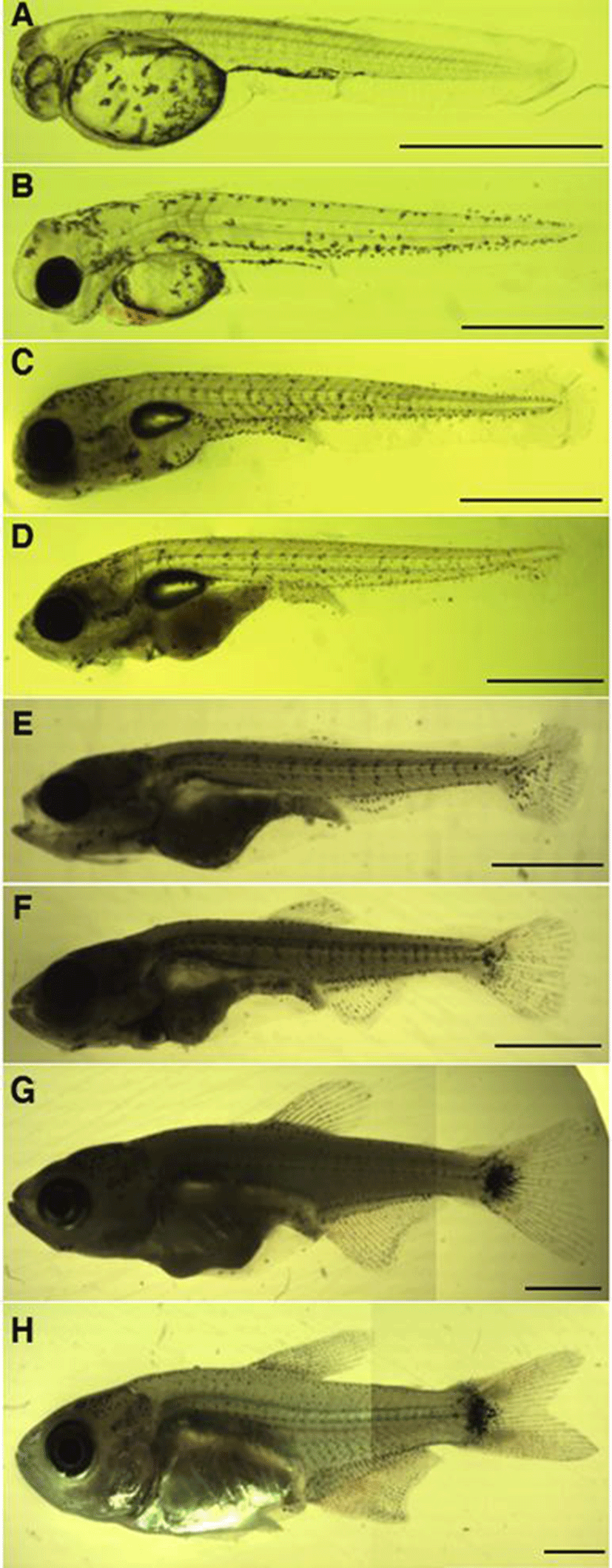
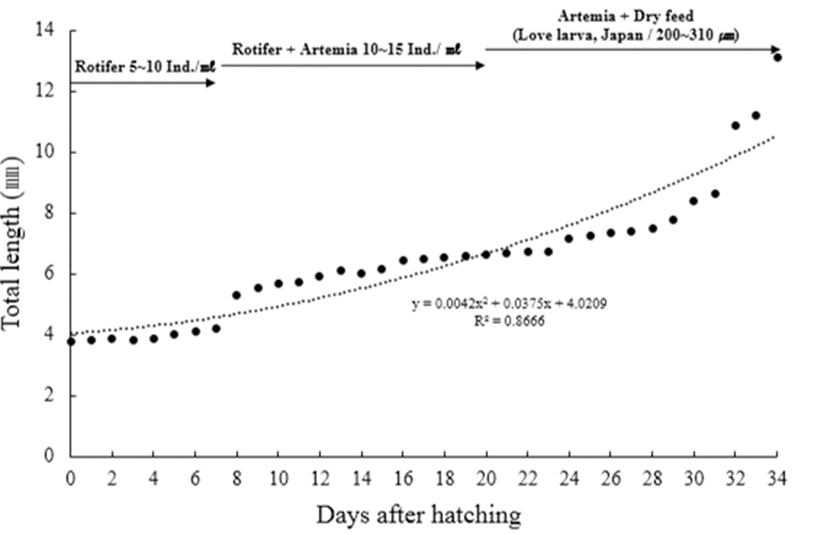
Juveniles on the 34 days after hatching were 8.63~13.1 mm in full length (mean 10.9±1.66 mm). Rays were formed in the membranous ventral fins, and the adipose fin was formed on the upper part of the caudal peduncle. The lower jaw developed longer than the upper jaw, and as the color turned silver their overall shape became similar to the mother’s (Fig. 3H).
DISCUSSION
The Amazon River in South America inhabited by Buenos aires tetra has several tributaries along with the main stream and therefore it teems with a large diversity of tropical fish species. However, tropical fish resources are shrinking rapidly due to recent development of primary forest areas, droughts caused by extreme weather events, indiscreet over fishing, etc. As environmental conditions for producing various kinds of tropical fish are growing worse, it is urgently necessary to make research on the reproduction and preservation of species.
Buenos aires tetra has one adipose fin on the upper part of the caudal peduncle, and such a fin is also observed in Serpae tetra, Hyphessobrycon eques (Park et al., 2014), Black skirt tetra, Gymnocorymbus ternetzi (Ihsan et al., 2012), etc.
Buenos aires tetra swims along the current of water, but it repeats staying in stagnant water and then swimming. At that time, the adipose fin is believed to play an important role in balancing the body while swimming in the water.
The fertilized eggs of Buenos aires tetra were globular without a quarter, and were adhesive demersal eggs. Egg diameter was 0.63~0.91 mm (mean 0.74±0.07 mm), larger than that of Tiger fish and Hydrocynus vittatus (Steyn et al., 1996) and smaller than 0.92 mm of Serpae tetra (Park et al., 2014), 1.13 mm of Brycon gouldingi (Faustino et al., 2011), and 1.49~3.09 mm of B. orthotaenia (Gomes et al., 2011) among other fish of the same family.
In the results of comparing time taken until hatching, it took 16 hours 13 minutes after the gastrula stage at mean water temperature 27.0±0.05°C in Buenos aires tetra, 12 hours at 28.0±0.05°C in Serpae tetra, 22 hours 30 minutes at 28.0±1°C in Tiger fish (Steyn et al., 1996), 13 hours at 26.4±1.12°C in B. gouldingi (Faustino et al., 2011), 28 hours 20 minutes at 21°C in Bryconamericus caucanus (Montoya et al., 2010), 21 hours 30 minutes at 24°C in B. orthotaenia (Gomes et al., 2011), and 20~21 hours at 24.0±0.5°C in Black skirt tetra (Ihsan et al., 2012). Characidae fish tended to develop slow at low breeding water temperature, and fast at high breeding water temperature (Byun et al., 2009).
As to the size of larvae just after hatching, the full length of hatched larvae was 3.78~3.88 mm (mean 3.84± 0.04 mm) in Buenos aires tetra, 2.76~3.05 mm (mean 2.89±0.16 mm) in Serpae tetra (Park et al., 2014), 2.87± 0.15 mm in B. orthotaenia (Gomes et al., 2011), 2.90 mm in Tiger fish (Steyn et al., 1996), and 1.44±14.3 mm in Black skirt tetra (Ihsan et al., 2012), so Buenos aires tetra showed the largest larva size.
As to the time for transition to the juvenile stage and size at that time, it was the 34th day from hatching and the full length was 8.63~13.1 mm (mean 10.9±1.66 mm) in Buenos aires tetra, and the 45 days and 11.9~14.1 mm (mean 12.5±1.60 mm) in Serpae tetra (Park et al., 2014) (Table 1). The process of early development and the morphological development of larvae and juveniles are very important data not only for the identification of species but also for the preservation of endangered species (Seo et al., 2010). In order to preserve the number of Characidae fish against overfishing, we must find good solutions with ecological research on broodstock rearing, life, environment and so on.
| Species | Egg diameter (mm) | Time of hatching (*WT) | Hatching larvae size (mm) | Juvenile (mm) | Reference |
|---|---|---|---|---|---|
| Hyphessobrycon anisitsi | 0.74±0.07 (0.63 0.91) | 16 hrs 13 mins (27.0±0.05°C) | 3.84±0.04 (3.78 3.88) | 10.9±1.66 (8.63 13.1) | Present study |
| Hyphessobrycon eques | 0.92±0.01 (0.91 0.93) | 12 hrs (28.0±0.05°C) | 2.89±0.16 (2.76 3.05) | 12.5±1.60 (11.9 14.1) | Park et al., 2014 |
| Hydrocynus vittatus | 0.65 | 22 hrs (28.0±1°C) | 2.90 | - | Steyn et al., 1996 |
| Brycon gouldingi | 1.13 | 13 hrs (26.4±1.12°C) | - | Faustino et al., 2011 | |
| Brycon orthotaenia | 1.49 3.09 | 21 hrs 30 mins (24.0°C) | 2.87±0.15 | - | Gomes et al., 2011 |
| Bryconamericus caucanus | - | 28 hrs 20 mins (21.0°C) | - | Montoya et al., 2010 | |
| Gymnocorymbus ternetzi | - | 20-21 hrs (24.0±0.5°C) | 1.44±14.3 | - | Ihsan et al., 2012 |

