INTRODUCTION
Ovarian follicular fate (i.e. development/ovulation versus atresia) in the mammals is determined by the fate of the cells within the follicles and is regulated by complex interactions of endocrine (gonadotropin and steroid hormones) and locally produced (growth factors and cytokines) factors (Hirshfield, 1991). Although follicular atresia can occur at all the stages of follicular development, a high incidence of granulosa cell apoptosis, a well recognized cellular basis for this degenerative process, is predominantly observed at the early antral stage of follicular development during the ovarian cycle (Kim et al., 1998).
Fas antigen (Fas), a member of the TNF receptor family, and its ligand Fas ligand (FasL), have been identified as cell death mediator and factor, respectively in a variety of cell types (WatanabeFukunaga et al., 1992; Suda et al., 1993). Their involvement in the induction of granulosa and luteal cell apoptosis has been demonstrated in several mammalian species (Quirk et al., 1995; Hakuno et al., 1996; Kondo et al., 1996; Kim et al., 1998). Fas and FasL expression is up-regulated in granulosa cells undergoing apoptosis during early stage of follicular development (Kim et al., 1998) and p53-mediated granulosa cell apoptosis is associated with the activation of Fas/FasL system during follicular atresia (Kim et al., 1999). In addition, follicle stimulating hormone (FSH) is an important negative regulator of the Fas/FasL system and a key cell survival factor during the follicular development and the role of the Fas/FasL system in granulosa cell apoptosis appears more prominent at early than at late stages of follicular maturation.
Interferon gamma (IFN-γ) is a member of a family of heterogeneous regulatory proteins having antiviral, immunomodulatory, antiproliferative, and cytodifferentiation regulatory activities (Pestka et al., 1987). It increases adrenal steroidogenesis (Blalock and Harp, 1981), increases iodide uptake by thyroid cells (Friedman et al., 1982), and suppresses hCG-induced testosterone production by Leydig cells (Orava et al., 1989). At physiological concentrations, IFN-γ inhibits granulosa cell differentiation and steroidogenesis (Gorospe et al., 1988; Xiao and Findlay, 1992). In addition, IFN-γ increases Fas mRNA expression in mouse lymphoma cells and BAM3 cells (WatanabeFukunaga et al., 1992) and Fas content in human granulosa / luteal cells (Quirk et al., 1995). Apoptosis in ovarian surface epithelial cells is mediated by increased Fas expression induced by IFN-γ (Quirk et al., 1997). Moreover, IFN-γ induces Fas-mediated apoptosis in differrentiated mouse granulosa cells which could be potentiated by the presence of TNFα (Quirk et al., 1998). Although the importance of IFN-γ in undifferentiated or poorly differentiated granulosa cells is well established, the presence of this cytokine in the ovary throughout follicular development has not been investigated. In addition, the regulation of Fas system in undifferentiated granulosa cells by IFN-γ, subsequent Fas-mediated apoptotic response as well as their modulation by gonadotropin in vivo remain unclear.
The purpose of the present study was to examine the role of IFN-γ in regulation of Fas expression and apoptosis in granulosa cells at different stages of follicular development and granulosa cell differentiation in vitro. In addition, we assessed the presence of IFN-γ in the ovary during follicular maturation and studied the relationship between the presence of IFN-γ, Fas/FasL system and apoptosis in vivo.
MATERIALS AND METHODS
[α32P]-ddATP and dNTPs were obtained from Amersham Pharmacia Biotech (Piscataway, NJ). Horse radish peroxidase (HRP)-conjugated goat anti-rabbit secondary antibody was purchased from Bio-Rad Laboratories (Hercule, CA). Rabbit anti-rat interferon-γ (IFN-γ) was obtained from Biosource International (Camarillo, CA). Biotinylated 16-dUTP, In situ cell death detection (Fluorescein) kit, proteinase-K and terminal deoxynucleotidyl transferase (TdT) were purchased from Boehringer-Mannheim (Indianapolis, IN). Recombinant rat IFN-γ was from Genzyme (Cambridge, MA). Fetal bovine serum (FBS), minimal essential medium (MEM), non-essential amino acids (NAA), penicillin and streptomycin were from Gibco/BRL (Burlington, ON, Canada). FITC-conjugated mouse anti-hamster IgG, hamster anti-mouse Fas monoclonal antibody (Clone Jo2) and hamster IgG were obtained from PharMingen (San Diego, CA). Avian myeloblastosis virus (AMV) reverse transcriptase, oligo(dT)15 primer, rRNasin ribonucleotide inhibitor and Taq DNA polymerase were from Promega (Madison, WI). Rabbit IgG, rabbit peroxidase kits, rabbit polyclonal anti-mouse Fas (sc-716) and anti-rat FasL (sc-834) antibodies, and neutralization peptides (Fas; sc-716P, FasL; sc-834P) were from Santa Cruz Biotechnology (Santa Cruz, CA). Agarose, bovine serum albumin (BSA; fraction V), diethylstilbesterol (DES), equine chorionic gonadotropin (eCG), methyl green, normal goat serum, phenylmethylsulfonyl fluoride (PMSF) and veronal acetate were purchased from Sigma Chemical Co. (St Louis, MO).
To obtain ovaries consisting mainly of follicles synchronized at the preantral / early antral and medium / large antral stages in immature (21-23 days old) Sprague-Dawley rats (Charles River Canada; Quebec, Canada or SamTako Bio-Korea; Osan, Korea), DES (1mg/day, sc, for 3 consecutive days and sacrificed 24 hr after last injection) and eCG (15 IU, ip and sacrificed 48 hr thereafter) were administered, respectively. The animals were fed Pro Lab RMH 4018 (Agway Inc., C.G., Syracuse, NY) and water ad libitum. A 14 h light:10 h dark photo-cycle was maintained with lights-on at 0600 h. All procedures were performed in accordance with protocols approved by the Dong-A University Animal Care and Use Committee. Following removal of connective tissues, the ovaries were washed in PBS (pH 7.4) to remove excess blood and were either immediately fixed in 10% neutral buffered formalin (pH 7.4) for histological processing or used for granulosa cell isolation by follicle puncture (Rao et al., 1991).
Granulosa cells (2.5×106) from ovaries of DES- and eCG-primed rats were plated for 6 hr (5% CO2, 37℃) in 60 mm Falcon plastic culture dishes (Becton Dickins, Lincoln Park, NJ) in MEM supplemented with 2% FBS, 1X NAA, penicillin (100 U/mL) and streptomycin (100 μg/mL). The medium was then replaced with MEM supplemented with 0.1% BSA, 1X NAA, penicillin (100 U/mL) and streptomycin (100 μg/mL) in subsequent culture. Non-viable (floating) cells were removed at this medium-replacement step.
For induction of Fas mRNA and protein expression, granulosa cells were cultured in the presence of recombinant rat IFN-γ (0-1,000 U/mL of the medium) for 18 hr. Floating and attached cells harvested by centrifugation and trypsinization respectively, were combined and washed with ice-cold PBS, pending protein and RNA extraction, and flowcytometric analysis. For immunocytochemistry and TUNEL, sterilized cover glasses (22×22 mm) were added onto the bottom of the culture dishes prior to cell plating. At the end of the culture period, the cover glasses were removed, washed briefly with ice-cold PBS and processed for immunocytochemistry.
To assess the influence of IFN-γ on Fas expression and subsequent granulosa cell apoptotic response, cells pretreated for 18 hr with / without the cytokine were incubated for an additional 6 h with either agonistic Fas monoclonal antibody (hamster anti-mouse Fas mAb, IgG; 1 μg/mL) or hamster IgG (control; 1 μg/mL). The effect of the treatment was assessed by phase-contrast microscopy, TUNEL and DNA fragmentation analysis.
Paraffin embedded whole ovarian sections (4-5 µm) were used for IFN-γ, Fas and FasL immunohistochemistry. The deparaffinized and hydrated sections were quenched in H2O2 (0.3%; 30 min) and rinsed thoroughly with PBS (3×15 min). The sections were blocked with normal goat serum in PBS [1.5%; room temperature (RT), 1 hr], and then incubated (RT, 45 min) with rabbit polyclonal anti-rat IFN-γ (0.8 μg/mL) or rabbit polyclonal anti-mouse Fas (0.3 μg/mL), or rabbit polyclonal anti-rat FasL antibodies (0.3 μg/mL) in PBS containing normal goat serum (1.5%). The sections were incubated at RT with biotin-conjugated goat anti-rabbit IgG (1:200; 1 hr), avidin-biotin-peroxidase complex (Santa Cruz rabbit peroxidase kit; 1 hr) and DAB solution (1-5 min), with nuclei lightly counterstained with hematoxylin. For negative controls, rabbit IgG (1 μg/mL) instead of the primary antibodies was added to the reaction. In this experiment, antibody specificity was confirmed by antibody preabsorption test, using recombinant rat IFN-γ (2-3 µg) and Fas and FasL neutralizing peptides (1 µg) in the respective primary antibody reaction.
For localization of Fas protein in cultured granulosa cells, the cells were fixed for 10 min in ice-cold paraformaldehyde in PBS (2%; pH 7.4). After several washes with PBS, the cells were incubated (20 min) in PBS containing normal goat serum (10%) to suppress nonspecific IgG binding, washed with PBS (3×5 min) and incubated for 1h with rabbit anti-mouse antibody [0.1 µg/mL in PBS containing normal goat serum (1.5%)]. Following a PBS wash, the cells were incubated (30 min in the dark) with fluorescein-conjugated goat anti-rabbit antibody [1 µg/mL PBS with normal goat serum (1.5%)], mounted with 90% glycerol in PBS and observed under fluorescence microscope.
The TUNEL method of Gavrieli et al. (1992) was used to localize apoptotic cells in paraffin whole ovarian sections mounted on positively charged slides (ProbeOn Plus; Fisher Scientific Ltd, Ottawa, ON). Briefly, the sections were deparaffinized, hydrated, treated with proteinase-K (10 μg/mL., 30 min.; 37℃). Endogenous peroxidase activity was removed by immersing the sections in methanol containing H2O2 (0.3%, 30 min., RT). The sections were soaked in the TdT buffer [Tris-HCl (25 mM), sodium cacodylate (200 mM), cobalt chloride (5 mM), BSA (250 μg/mL), pH 6.6; 15 min), incubated in 50 μL of TdT buffer containing TdT (10 U) and biotinylated 16-dUTP (1 nmol) [60 min., 37℃], reacted with avidin-biotin-peroxidase complex (Santa Cruz rabbit peroxidase kit; 30 min) and subsequently with DAB solution (1-5 min), as per manufacturer’s instruction. If required, the nuclei were counterstained with methyl green (5%; in 0.1 M veronal acetate, pH 4.0). TdT enzyme or biotinylated 16-dUTP in the labelling reaction were omitted in the negative control slides. For in situ detection of apoptosis in cultured cells, cells were fixed (10 min, 0℃) in fresh paraformaldehyde (4% in PBS) and an In situ cell death detection (Fluorescein) kit (Boehringer Mannheim; Quebec, Canada) was used, as per manufacturer’s instructions.
Total DNA was extracted from cultured granulosa cells (floating cells + trypsinized attached cells) according to the modified procedure of Gross-Bellard et al. (1973), as previously described (Li et al., 1998). For biochemical assessment of DNA fragmentation, DNA was radiolabeled at the 3’-ends according to an established method (Tilly et al., 1993). Briefly, one microgram of DNA was incubated (37℃; 60 min) with 50 μL of the labelling buffer [Tris-HCl (25 mM), sodium cacodylate (200 mM), cobalt chloride (5 mM), BSA (250 μg/mL), pH 6.6] containing terminal deoxynucleotidyl transferase (TdT; 25 U) and [α32P]-ddATP (5 mCi, 3,000 Ci/mmol). The labeled DNA were resolved on 1.8% agarose gel (3.5 h, 60 V) in TAE buffer. The gel was dried and subsequently exposed to X-ray film at -70℃.
Total RNA was isolated from cultured granulosa cells using the Qiagen RNeasy Mini RNA isolation kit (Valencia, CA). Briefly, the cells attached to the culture dishes were lyzed with lysis solution (buffer RLT) provided by the manufacturer, and the lysate was passed through the Qiagen QIA shredder (Valencia, CA). To ensure the extracted RNA for PCR was free of DNA, samples were repeatedly treated with DNase.
Two micrograms of total RNA was reverse-transcribed (42℃, 15 min) with oligo (dT)15 primers (0.5 µg/µg RNA) and Avian myeloblastosis virus (AMV) reverse transcriptase (15 U/µg RNA) in a 20 µL reaction mixture containing 10X reverse transcription buffer [Tris-HCl (100 mM) (pH 8.8), KCl (500 mM), Triton X-100 (1%)], MgCl2 (25 mM), dNTP mixture (10 mM) and rRNasin ribonucleotide inhibitor (2 U). The reaction was terminated by inactivating the reverse transcriptase (heating at 99℃, 5 min).
Gene-specific oligonucleotide primers (46.1% GC content) were designed based on published rat Fas cDNA sequence [22; GenBank Accession No. D26112] and synthesized using a Beckman Oligo 1000M DNA synthesizer (Fullerton, CA). Primers for rat β-actin (Nudel et al., 1983) were obtained from Clontech (Palo Alto, CA). The primers used for PCR amplification for Fas were 5'-TGCACCTCGTGTGGACTTGAAG-3'[479-500; forward primer] and 5'-CCAGTCTTCCCGTGAGATTGATAC-3' [888-865; reverse primer]. Polymerase chain reactions were carried out with a PCT-100 Programmable Thermal Controller (MJ Research, Inc., Watertown, MA). The PCR reaction mixture included the oligonucleotide primers (5 pmol each), and Taq DNA polymerase (0.5 U) for each reaction. The total volume was brought up to 50 µL with 1X PCR buffer [Tris-HCl (50 mM), KCl (20 mM), MgCl2 (3 mM), DMSO (0.5%)] and dNTP (20 µM). Amplification was carried out for 35 cycles under the annealing temperature of 60℃ (45 sec), primer extention at 72℃ (1 min) and denaturation at 94℃ (30 sec). The amplified DNA was electrophoresed on 2% agarose gel, and the gel was stained with ethidium bromide for visualization under UV light. At initial stage of this experiment, the amplified Fas product was confirmed by Southern hybridization (Sambrook et al., 1989), using the rat Fas cDNA probe. The intensities of the bands were densitometrically scanned and each Fas amplified product was normalized by β-actin.
Harvested granulosa cells (2×107 cells) were resuspend in ice-cold wash buffer [NaN3 (0.1%), FBS (0.5%) in PBS] and centrifuged (500 ×g; 5 min, 4℃). The cells were immediately incubated in primary antibody solution [hamster anti-mouse Fas (0.5 µg/mL) in wash buffer)] at 4℃ for 30 min., washed (500 ×g, 2×5 min in wash buffer), and incubated (30 min., 4℃ in dark), with FITC-conjugated mouse anti-hamster IgG (1.0 µg/mL). The cells were then washed twice with the wash buffer, filtered through a 35 μm nylon mesh to remove cell clumps and aliquoted for appropriate cell concentration (final concentration, 1×106 cells in 500 µL) for flow cytometric analysis (Coulter Epics Profile II, Hialeah, FL). Ten thousand cells were counted in duplicates and analyzed by FACScan. As negative controls, nonspecific binding in flowcytogram was determined in the absence of primary antibody.
PCR and flowcytometry data presented herein were expressed as the mean±SEM of three separate experiments. Statistical analyses were performed by one-way ANOVA. Signicant differences between treatment groups were determined by the Tukey test. Statistical significance was inferred at p<0.05.
RESULTS
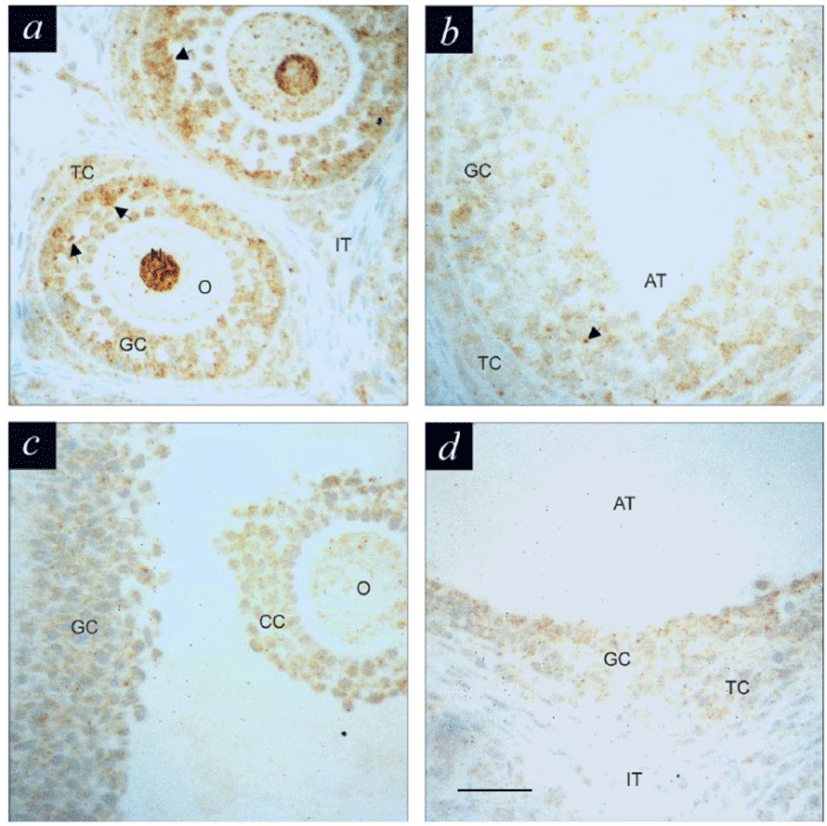
The presence and relative abundance of IFN-γ protein in the follicles during development were screened by immunohistochemistry, using a rat specific IFN-γ antibody (Fig. 1). Most of follicular cells from preantral follicles (Fig. 1a) exhibit immunoreactivity for IFN-γ, however, the intense and aggregated signals were detected in granulosa cells (arrows) as well as oocyte (including its nucleus). In contrast to its intense immunoreactivity in the cytoplasm, the nuclei of oocytes of secondary and tertiary preantral follicles showed considerably lower immunostaining. This pattern, however, was reversed with follicular matura-tion. In non-treated or DES-treated immature rats (Fig. 1b), granulosa cells in preantral follicles exhibited immunopositivity for IFN-γ, although with lesser intensity and aggregated characteristics than those in primordial and early stage of preantral follicles. The intensity of immunereactivity observed in granulosa cells of medium (Fig. 1c) and large (Fig. 1d) antral follicles was much less than that seen in the preantral follicles. Generally, the immunereactivity in theca cells was minimal in the antral follicles.
Based upon the immunohistochemical evidence for the presence of INF-γ at different stage of follicular development in vivo (Fig. 1), the potential role of IFN-γ in the regulation of Fas mRNA and protein in relatively undifferentiated (DES-primed) and differentiated (eCG-primed) granulosa cells was investigated in vitro, using RT-PCR (Fig. 2), immunocytochemistry and flowcytometry (Fig. 3). In cultured DES-primed granulosa cells, IFN-γ up-regulated Fas mRNA in a concentration-dependent manner (Fig. 2). High concentration (>100 U/mL) of IFN-γ (1,000 U/mL) resulted in the loss of effectiveness of the cytokine in inducing Fas mRNA and protein expression (Fig. 2).
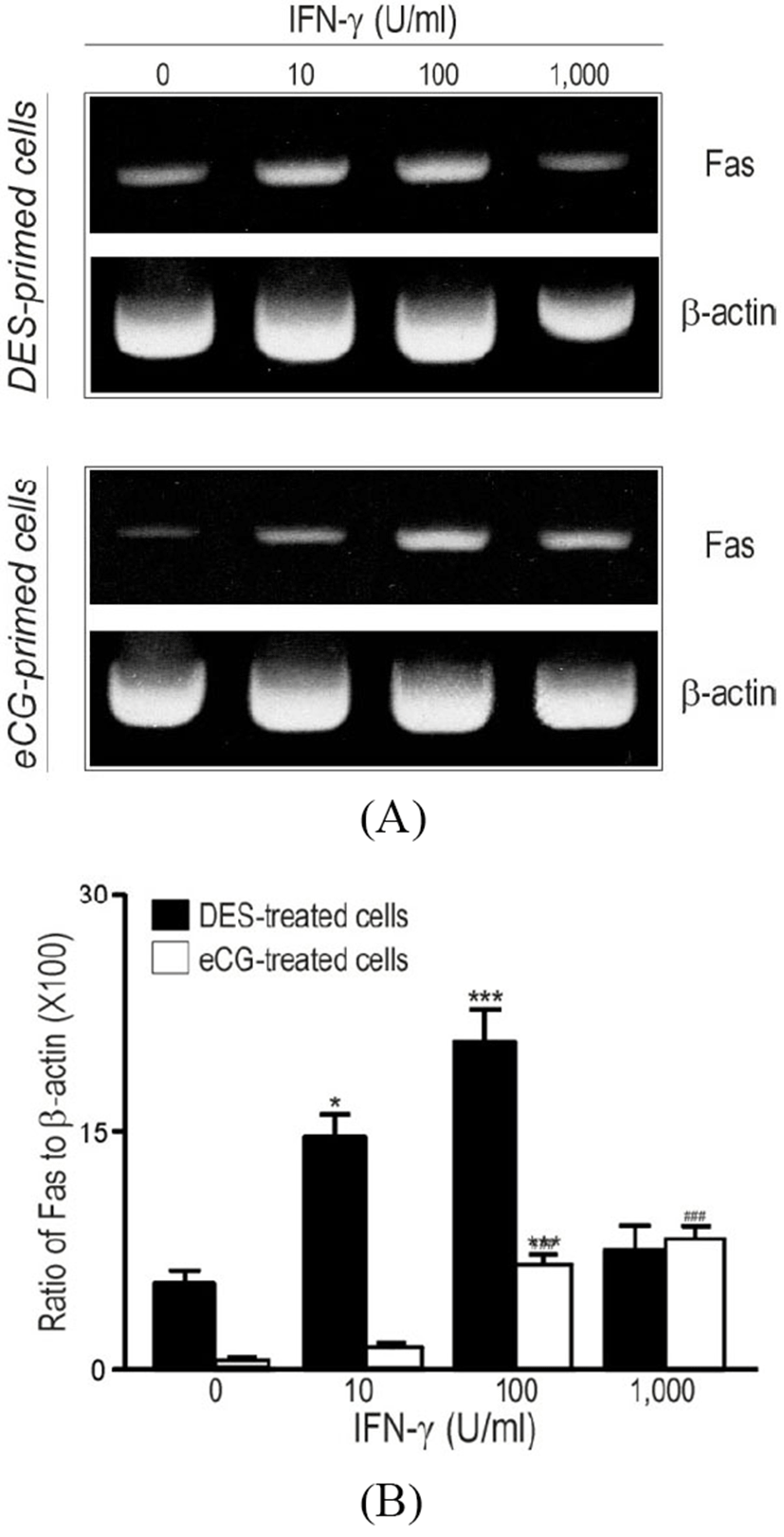
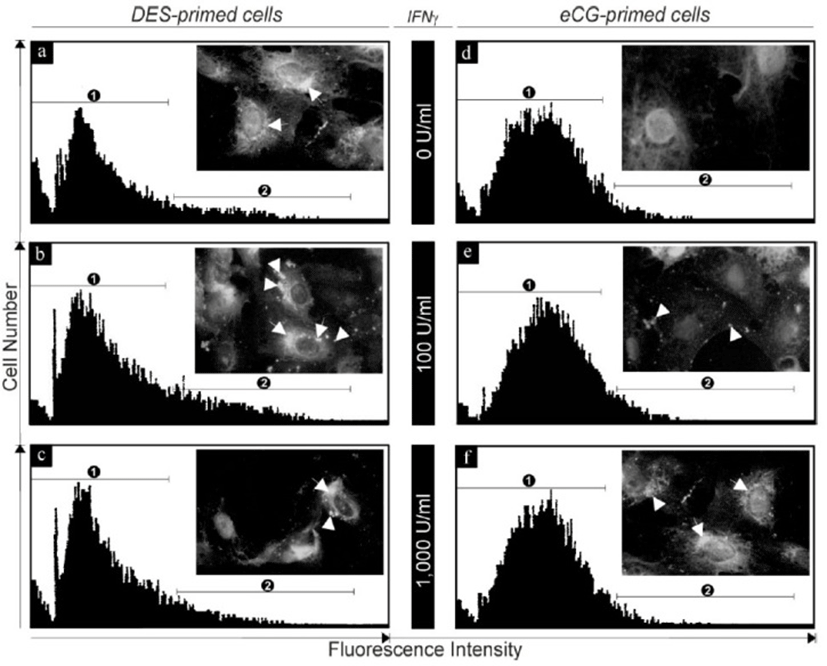
Although cultured eCG-primed granulosa cells also expressed Fas mRNA, its abundance was considerably lower than that in DES-primed cells. In addition, while Fas mRNA also increased in a concentration-dependent manner in the presence of IFN-γ, significantly higher concentrations of the cytokine (100-1,000 U/mL, p<0.01) were required to elicit a significant increase in Fas expression than those in DES-primed cells (Fig. 2). The Fas protein was localized in the DES- and eCG-primed granulosa cells after IFN-γ treatment and the expression levels were subjected to Fas antibody-conjugated immuno-flowcytometric analysis (Fig. 3). As shown in the inserts within each representative flowcytogram (Fig. 3), Fas protein was localized in both the cellular membrane (arrow head) and the cytoplasm [peri-nuclear region (arrow)]. In addition, intense and aggregated immuno-fluorescence was observed in both DES- (Fig. 3b and 3c) and eCG-primed (Fig. 3) granulosa cells after IFN-γ exposure. Whereas a significant population of the control group in the DES-primed granulosa cells exhibited the high intensity of immunofluorescence for endogenous Fas immunoreactivity [Fig. 3a, portion 2 (18±4.3%)], the eCG-primed control granulosa cells showed relatively smaller population with high immunofluorescence intensity for Fas [Fig. 3d, portion 2 (4±3.5%)]. IFN-γ treatment (100 U/mL) of DES-primed granulosa cells significantly increased high intensity (33±7.5%) portion of Fas immunofluorescene (Fig. 3b), as also confirmed by immunocytochemistry (Fig. 3b, inert). However, treatment with high concentration of IFN-γ (1,000 U/mL) resulted in a lower number of cells exhibiting immunofluorescence positivity for Fas in high intensity (25±2.3%) portion than those with 100 U/mL but higher than that of the control group. In eCG-primed cells, however, only small increases in immunofluorescence were detected with IFN-γ at concentrations of 100 and 1,000 U/mL when compared to controls (Fig. 3e and 3f).
The functional role of IFN-γ-induced Fas expression in induction of apoptosis were tested in undifferentiated (DES-primed) and differentiated (Ecg-primed) granulosa cells in vitro was determined by assessing their the ability to undergo apoptosis following a challenge with an agonistic effect of Fas mAb. While typical cellular morphology and apoptosis in DES-primed granulosa cells are illustrated in Fig. 4, differences in these features between DES- and eCG-primed groups are summarized in Table 1. In DES-primed (undifferentiated) granulosa cells, IFN-γ had minimal effects on cellular morphology (Fig. 4b) and apoptotic characteristics (Fig. 4f) compared to control (IgG treatment; Fig. 4a & 4e). However, addition of Fas mAb to the cell cultures resulted in the appearance of apoptotic cellular morphology and characteristics (Fig. 4c & 4g), which could be enhanced with pretreatment with IFN-γ (Fig. 4d & 4h). In eCG-primed (differentiated) granulosa cells, however, IFN-γ pretreatment failed to potentiate the pro-apoptotic effects of agonistic Fas mAb, which were observed prominently in DES-primed cells (Table 1).

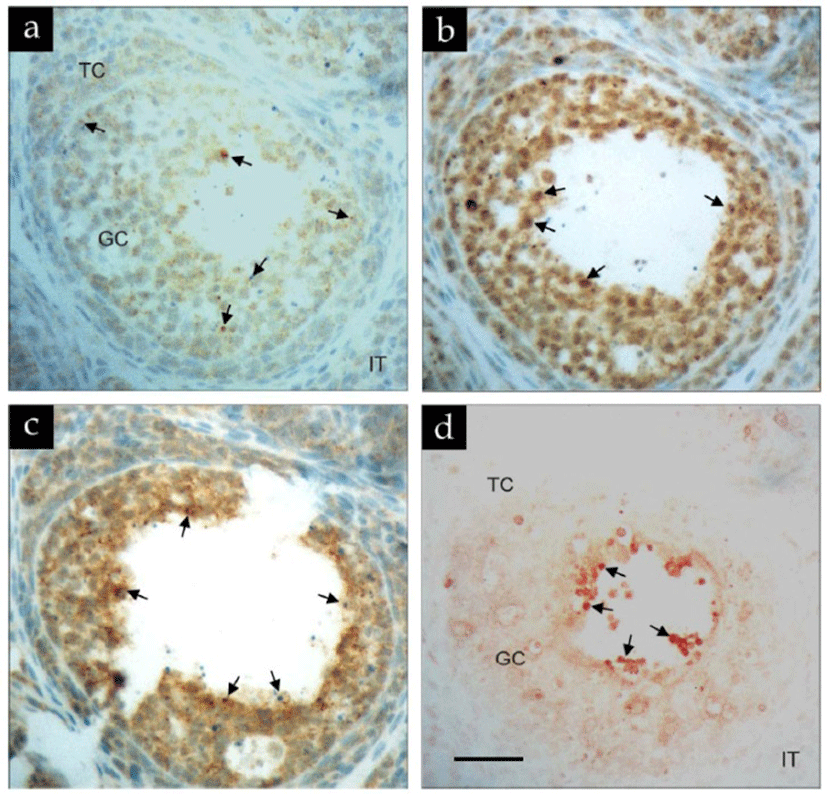
To further examine the colocalization of IFN-γ, Fas and FasL expression and occurrence of apoptosis in vivo, follicles at very early stage of antral formation (estimated between preantral and early antral stages) were analyzed serially by immunohistochemistry and in situ TUNEL method (Fig. 5). Immunoreactivity for IFN-γ was distributed in the granulosa but not the thecal layers (Fig. 5a). Likewise, the most intense and aggregated form of Fas immuno-positive signals were detected in the loosely attached granulosa cells (Fig. 5b). Although FasL immunoreactivity could be colocalized in Fas-positive cells, he most aggregated and intense (arrows) immunoreactivity was present in granulosa cells mainly lining the antrum (Fig. 5c). Similarly, apoptotic (TUNEL-positive) granulosa cells were also detected in antral granulosa cells (Fig. 5d; arrows) which also exhibited most intense immunoreactivity for FasL.
DISCUSSION
In the present study, we have localized rat IFN-γ in the rat ovary during the follicular development and tested its role on granulosa cell death via induction of the Fas/FasL system in granulosa cells at different stage of differentiation. We have found that the responsiveness of granulosa cells to IFN-γ in terms of Fas receptor expression was significantly compromised with follicular maturation. The immunoreactivity of IFN-γ was most intense in early stages of folliculogenesis, such as in preantral follicles. Up-regulation of Fas by IFN-γ and the subsequent increased responsiveness to an apoptotic death signal (e.g. Fas activation by agonistic Fas mAb) were more evident in granulosa cells from the preantral follicles than those from antral follicles previously exposed to gonadotropins.
The importance of intraovarian interaction between immune cells (lymphocytes and macrophages) and granulosa cells in ovarian physiology via the synthesis and actions of cytokines (e.g.TNFα and IL) has been studied extensively (Adashi, 1992; Terranova et al., 1993). Although the modulatory roles of IFN-γ in granulosa cell differentiation and steroidogenesis have been demonstrated in vitro (Gorospe et al., 1998; Xiao and Findlay, 1992), the cellular source of IFN-γ in the ovary is still unclear. IFN-γ is detectable in the follicular fluids (Grasso et al., 1988) and lymphocytes present in the ovary have been suggested to be an important contributor to the follicular IFN-γ (Adashi, 1992). Immune cells (lymphocytes / macrophages) are believed to be mainly present in atretic antral follicles (Best et al., 1996), inflammated post-ovulatory follicles and corpus luteum (Brannstrom et al., 1994a; Brannstrom et al., 1994b) where they remove damaged and dead cells by secreting cytotoxic cytokines and phagocytosis. The presence of immune cells during early stage of follicular development has not been reported and the possibility that immune cells are the only the source of IFN-γ in preantral follicles cannot be excluded. In this study, IFN-γ was localized predominantly in granulosa cells and oocytes throughout the follicular development but more abundantly in those at the early follicular (preantral) stage. It has been demonstrated that TNF-α and Fas ligand are expressed in granulosa cells and oocytes (Chen et al., 1998; Kim et al., 1998). Although we have not studied IFN-γ gene expression in ovarian cells in the current study, it is possible that oocytes and granulosa cells in the preantral follicles may be an important source of IFN-γ in the ovary and that this cytokine plays an autocrine and/or paracrine role in regulating the expression and actions of other cytokines involved in cell removal process in the degrading follicles.
IFN-γ is an important modulator of granulosa cell differentiation during the early stage of follicular development (Gorospe et al., 1988; Xiao and Findlay, 1992), although its functional role in differentiated granulosa cells in late follicular development (i.e., preovulatory follicles) remains to be further established. Granulosa cell apoptosis is frequently observed in early antral (penultimate) stage of follicular development, it is only assumed that up-regulation of Fas receptor by IFN-γ in granulosa cells of preantral follicles is prerequisite for FasL ligation and that the latter could be activated by intra-ovarian factor(s) following antrum formation. It has been suggested that IFN-γ facilitate apoptosis in luteinized granulosa cells by decreasing endogenous IGF activity via stimulation of IGFBPs rather than of Fas activation (Cataldo et al., 1998). However, the concentrations of IFN-γ used in above studies could have been pharmacological and cytotoxic to the cells. Quirk et al (1995) demonstrated Fas-mediated apoptosis in human granulosa-luteal cells and suggested that IFN-γ up-regulates Fas mRNA and protein expression and that pretreatment of cells with IFN-γ alone or in combination with hCG facilitates Fas mAb-induced cytotoxicity. Therefore, it is unlikely that the physiological concentration of IFN-γ could induce Fas expression in fully differentiated luteinized (or luteinizing) granulosa cells. Recently, Quirk et al (1998) showed that combined IFN-γ and TNFα treatment was more effective in inducing Fas expression and potentiating Fas mAb-induced apoptosis in cultured granulosa cells (mostly from large preovulatory follicles in eCG-primed mice) than by either IFN-γ or TNFα alone. The results suggest that the mechanism(s) of induction of apoptosis and responsiveness against apoptotic insult (e.g. IFN-γ) in differentiated granulosa cells might be much more complex and resistant than those of undifferentiated granulsoa cells, as also seen in the present study.
The possible mediatory role of the Fas/FasL system in granulosa and luteal cell apoptosis during follicular atresia and luteal regression, respectively have been investigated in several mammalian species (Quirk et al., 1995; Hakuno et al., 1996; Kondo et al., 1996; Kim et al., 1998; Roughton et al., 1999). Previous reports have demonstrated that increased granulosa cell Fas and FasL protein content is associated with apoptosis at the penultimate (early) stage of follicular development in vivo (Kim et al., 1998), and that p53-mediated granulosa cell apoptosis involves the activation of the Fas/FasL system (Kim et al., 1999). These studies also suggested that FSH is a key inhibitory factor on granulosa cell death as well as the involvement of the Fas/FasL system in follicular atresia. The nature of the regulation of Fas receptor expression throughout the follicular development, however, remains unclear. Although the regulation of Fas receptor expression by IFN-γ has been demonstrated in human granulosa-luteal cells in vitro (Quirk et al., 1995), the potential regulatory role of IFN-γ in Fas mRNA and protein expression in undifferentiated granulosa cells during early stages of folliculogenesis has not been thoroughly investigated. In the present studies, we have studied the possible role of IFN-γ in the regulation of granulsoa cell Fas expression during follicular development. While increased Fas expression in granulsoa cells from preantral or early antral follicles could be induced with low concentrations of IFN-γ (10-100 U/mL), higher concentrations (≥1,000 U/mL) were required in eCG-primed cells. Our present immuno-flowcytometric analysis also confirmed that undifferentiated (DES-primed) granulsoa cells were more responsive to IFN-γ in the induction of Fas receptor than cells pretreated with gonadotropin. These findings are not only consistent with the studies by Quirk et al (1995) which showed increased expression of Fas protein in gonadotropin-primed human granulosa cells following exposure to very high concentration (> 10,000 U/mL) of IFN-γ in vitro, but extended their observations to suggest a role for the cytokine in the ovary at physiological concentrations.
It is well established that FSH is an important cell survival factor in the mammalian ovarian follicle (Chun et al., 1996; Boone et al., 1997). Whereas stimulation of immature rats with eCG resulted in suppressed basal granulosa cell Fas/FasL levels and apoptosis, decreased atresia and enhanced follicular growth, withdrawal of gonadotropic support with the injection of a neutralizing antibody in vivo during early antral development led to increased granulosa cell Fas/FasL expression and apoptotic cell death, and eventual follicular demise (Kim et al., 1998). In the present studies, we have demonstrated that while IFN-γ effectively up-regulated Fas expression in granulosa cells from preantral and early antral follicles and sensitized them to the pro-apoptotic action of agonistic Fas antibody, cells isolated from large antral follicles previously exposed to the gonadotropin in vivo were more differentiated but less responsive to IFN-γ. These findings suggest that the anti-apoptotic role of FSH in the granulosa cells may in part involve its modulatory influence on IFN-γ action, and are consistent with the concept that ovarian follicles beyond the “penultimate” stage of development are protected physiologically from pro-apoptotic insult and destined to ovulate (Chun et al., 1996; Boone et al., 1997). The mechanism(s) by which the gonadotropin modulates IFN-γ-induced Fas expression granulosa cells is unclear. It has been demonstrated that Fas expression is regulated by the newly identified Fas-regulatory gene TDAG51, the expression of which is PKC-dependent (Wang et al., 1998). Since IFN-γ, like TNFα (Sancho Tello and Terranova, 1991) and IL-3 (Farrar et al., 1985), appears capable of activating PKC (Ostrowski et al., 1988), it is possible that the reduced responsiveness of differentiated granulosa cells to IFN-γ might be due to PKA activation by FSH. In addition, activation of PKA pathway is known to inhibit PKC-dependent mechanism(s) in a variety of biological systems including the granulosa cell (Shinohara et al., 1985). In this context, differential responsiveness of granulosa cells at different cytodifferentiation to IFN-γ may be related to possible interactions between signal transduction pathways of IFN-γ and gonadotropin. It is possible that the efficient Fas gene expression in undifferentiated granulosa cells may involve activation of PKC pathway by IFN-γ.
Fas receptor signaling is a complex process and continues to capture considerable attention and research efforts. Fas activation initiated by ligation and oligomerrization results in recruitment of FADD (Fasassociated death domain-containing protein) (Chinnaiyan et al., 1995). FADD then activates procaspase8 (Muzio et al., 1996), which appears to be the first step of a proteolytic cascade that activates other caspases such as caspases3, 6, and 7 (Hirata et al., 1998). In the present studies, IFN-γ significantly increased the population of granulosa cells exhibiting high fluorescence intensity of Fas immunoreactivity. While this increase might merely represent an increase in the cell population with enhanced Fas expression, it is tempting to speculate that the high intensity of fluorescence reflects the oligomerization of Fas receptor proteins, and that Fas oligomerization in response to IFN-γ in undifferentiated granulsoa cells may be more efficient and functional than in differentiated granulosa cells. Whether this indeed is the case requires further investigation and confirmation. In this context, intense and aggregated immunoreactive Fas protein has consistently been detected in cytoplasm as well as cellular membrane of cultured granulosa cells of the present study. These results support our previous observation suggesting the presence of aggregated Fas immunostaining in the granulosa cells of atretic early antral follicles in the ovary (Kim et al., 1998). Moreover, recent studies have indicated that Fas is also localized in the peri-nuclear region of the cytoplasm and that Fas-trafficking from Golgi apparatus to membrane is P53-dependent and required for Fas-mediated apoptosis (Bennett et al., 1998). In light of our recent demonstration of the role of p53 in the regulation of Fas expression in rat granulosa cells (Kim et al., 1999) and their intense Fas immunostaining following IFN-γ challenge, investigations into whether this cytokine plays a role in Fas trafficking in the granulosa cell would provide important clues on the cellular mechanism involved in the control of Fas-mediated granulsoa cell apoptosis during early folliculogenesis.
In summary, we have demonstrated that IFN-γ plays a pivotal role in regulating granulosa cell differentiation and apoptosis by potentiating Fas function, a process modulated by gonadotropin (i.e. FSH). The role of IFN-γ is most prominent in undifferentiated granulosa cells during early stage of follicular development.

