INTRODUCTION
The far eastern catfish Silurus asotus is a member of the freshwater Siluridae family. In Korea, its annual production is about 8,000 metric tons (Kim et al., 1988; Yang et al., 2015). Although the demand for this species has increased in recent years, two factors limit the profitability of culturing far eastern catfish. First, the growth rate shows significant sexual dimorphism because females grow much faster than males (Kim et al., 2001). This hinders stock management and often results in cannibalism of young fish in farms. Second, precocious maturation before the fish reach marketable sizes necessitates prolonged cultivation beyond sexual maturity. After reaching sexual maturity, catfish exhibit reduced growth and decreased feeding efficiency (Choi et al., 1992).
Cryopreservation of spermatozoa could be useful for artificial propagation, which may improve the genetic characteristics of this species. Cyropreservation techniques have already been reported for spermatozoa from several fish species (Linhart et al., 1993; Conget et al., 1996; Linhart et al., 2004). Cryopreserved spermatozoa may be used in research and in aquaculture by enabling the introduction of genes from wild or native populations into hatchery stocks if spawning times do not overlap, and increases the availability of spermatozoa for use in selective breeding programs, especially during seasonal shortages of males and/or females. Furthermore, cryopreserved spermatozoa can be exchanged between countries located in both hemispheres, instead of exchanging fish, a particular advantage when the importation of live animals is prohibited (Conget et al., 1996). Marian & Krasznai (1987) were the first to successfully cryopreserve spermatozoa from European catfish, S. glanis. Their fertilization rates using frozen testicular spermatozoa were 40%-95%. They used testicular spermatozoa for freezing, because artificially stripped spermatozoa were polluted with urine, which activated and destroyed the fertilization capacity within a few minutes (Linhart et al., 1987). This problem was overcome by storing spermatozoa in diluents, and the cryopreservation techniques were refined in later studies, in which the optimization of the diluents extended the viability and freezing rates of spermatozoa (Linhart et al., 1993, 2004). Cryopreservation procedures have been successfully developed for five other catfish species: African catfish, Clarias gariepinus; channel catfish, Ictalurus punctatus; blue catfish, I. furcatus; Pseudoplatystoma corruscans; and striped catfish, Pangasius hypophthalmus, but only using testicular spermatozoa (Steyn et al., 1985; Christensen & Tiersch, 1997; Urbanyi et al., 1999; Horvath & Urbanyi, 2000; Rurangwa et al., 2001; Carolsfeldt et al., 2003; Kwantong & Bart, 2003; Lang et al., 2003).
The aim of the present study was to optimize cryopreservation methods for ex situ cryopreservation of spermatozoa from far eastern catfish. The success of the cryopreservation procedure was evaluated in terms of the spermatozoa activity index (SAI) and hatching rates of eggs fertilized with thawed cryopreserved sperm.
MATERIALS AND METHODS
Far eastern catfish, Silurus asotus were cultured according to the methods of Kim et al. (2001). Mature females were induced to spawn by a single intraperitoneal (ip) injection of human chorionic gonadotropin (hCG; Sigma, St. Louis, MO, USA) administered at 1,000 IU/kg body weight (BW). Spermatozoa were obtained by incision of testes from mature males that had also been given an ip injection of hCG at 500 IU/kg BW. The eggs were fertilized with spermatozoa diluted in saline using the wet method (Kim et al., 2001). The eggs were left to fertilize for 5 min and then rinsed to remove excess spermatozoa. The hatched fry were reared in 450 L tanks under the same conditions. The mean±standard deviation (SD) water temperature was 24±1.5℃ and the oxygen concentration was kept close to saturation (mean±SD: 9.4±0.3 mg/L). The experimental fish were fed twice daily at 2% of the mean body weight throughout the experimental period (2 years). Specimens were obtained from 2-year-old hatchlings at a mean±SD BW and standard length of 404.1±47.36 g and 42.9±5.11 cm, respectively.
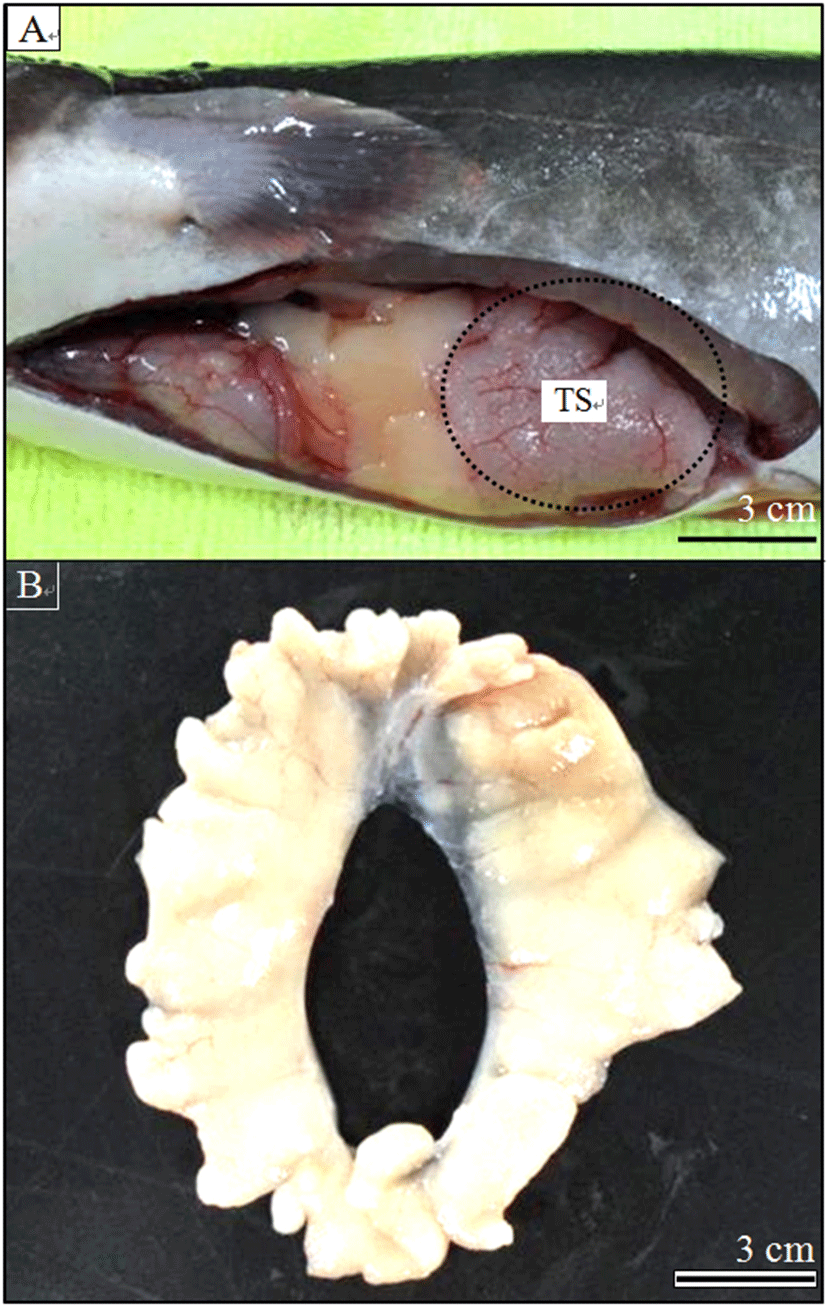
On 22 April 2010, 50 fishes were randomly captured and anesthetized with 200 ppm lidocaine-HCl/1,000 ppm NaHCO3 to remove the testes. Spermatozoa were obtained from 8 males by incision of the surgically removed testes after an ip injection of hCG at 1,000 IU/kg BW (Fig. 1). Spermatozoa were pooled from four fish with a spermatozoa motility index of >80%, and were used for cryopreservation. The spermatozoa concentration was determined using a Thoma hemocytometer (depth: 0.0025 mm2; Neubauer, Germany) under a microscope (1,000×; CH 20; Olympus, Tokyo, Japan) and the value is expressed as the mean number of spermatozoa in 20 squares of the Thoma cell. The diluents used in this study were Alserver’s solution (2.05 g glucose, 0.4 g sodium chloride, and 0.8 g sodium citrate/100 mL distilled water) and 0.3 M glucose solution (54.3 g glucose/1,000 mL distilled water). A maximum of 8 mL of sperm was added to 40 mL of diluent to maintain a dilution rate of 1:5 (sperm/diluent) to prevent spontaneous initiation of spermatozoa activity (Linhart et al., 2004). After collection, the containers were stored under aerobic conditions on ice at 0℃ for 1 hour. Sperm activity was assessed (Table 1) and SAI was assessed under a microscope and using video records (n = 100). SAI was assessed as described by Strüssmann et al. (1994).
* SAI: (Score × % motile sperm)/100 (After Strüssmann et al., 1994).
We compared the effect of two cryoprotectants (dimethyl sulfoxide [DMSO] and ethylene glycol) at different concentrations and in various combinations, as well as five different storage times. The final concentrations of DMSO and ethylene glycol were 5%, 10%, 15%, and 20%. One milliliter of mixture was transferred into a 1.8 mL cryotube and immediately placed in a controlled rate cryofreezer (PLANER Kryo 10 series III; Planer, Sunbury-on-Thames, UK) that was set at +4℃ and programmed to cool from +4 to –9℃ at a rate of 4℃/min, and from –9 to –80℃ at a rate of 11℃/min. The temperature was then held at –80℃ for 6 min. The tube was then transferred into liquid nitrogen and stored for 1, 180, 360, 540, or 720 days. After storage for the indicated time, frozen spermatozoa were thawed in a water bath at 15, 25, 35, or 45℃ for 90 sec.
For fertilization, 5 g of eggs (145 eggs per 1 g) was placed in a 50 mL dish. Then, 104,000 thawed or unfrozen spermatozoa per egg were dropped onto the eggs using a micropipette. After shaking the dish at 200 rpm with a 10 mm deflection, 5 mL of activation solution (17 mM NaCl, 5 mM Tris-HCl, pH 8) at 22℃ was added. After 3 min, about 100 eggs were placed into a special incubator cage containing 200 mL of ultraviolet-sterilized recirculated tap water at 23℃ (9 mg/L O2). The dead eggs were counted and removed from each cage 1 day after fertilization and after hatching (approximately 2.5 days after fertilization). The number of larvae in each cage was also counted. The hatching rate (Hr) was calculated for each cage using the following formula (Linhart et al., 2005):
where, Hl = number of hatched larvae and Et = total number of eggs placed in the cage.
To determine the proportion of immobile spermatozoa and for scanning electron microscopy (SEM) analysis, 100 µL of stored spermatozoa, diluted 1:100 in PBS, was placed in the center of a coverslip coated with 50 µg/mL poly-L-lysine. Cells were left to adhere for 30 min and fixed in 1% glutaraldehyde and 1% sucrose in PBS. They were then dehydrated in ethanol, subjected to critical point freezing, and sprayed with gold. We observed 100 spermatozoa per group at 720 days after storage, and counted the number of normal and abnormal spermatozoa in each group.
Each experiment was repeated three times for statistical analyses. One-way analysis of variance was used to test the statistical significance (P<0.05) of differences among experimental groups. Multiple comparisons were performed with Duncan’s multiple range test. SPSS software version 12.0 was used for all analyses (SPSS Inc., Chicago, IL, USA).
RESULTS
The spermatozoa from far eastern catfish, S. asotus showed good quality (Table 2). The quality of spermatozoa changed depending on the treatment. The SAI was greater for spermatozoa diluted in Alserver’s solution than for those diluted in glucose. The SAI of spermatozoa diluted sperm in both solutions decreased with increasing duration of storage; therefore, better results were obtained when spermatozoa were stored for 1 hour than when stored for 24 hours (Table 2).
| Diluent | SAI* | |
|---|---|---|
| Activation (1 hour) | Storage (24 hours) | |
| Alsever's solution | 1.6±0.18a | 0.5±0.07b |
| 0.3 M glucose | 1.4±0.22a | 0.3±0.09b |
* SAI: (Score × % motile sperm)/100 (After Strüssmann et al., 1994). The temperature of storage is 0℃. Values are means±SD. Values in same row having the different superscripts are significantly different (n = 100, P<0.05). Experiments are triplicate.
The SAI values of far eastern catfish spermatozoa stored in two diluents and with different concentrations of the cryoprotectants are shown in Table 3. The SAI gradually decreased with increasing storage duration in all of the experimental groups. The SAI of spermatozoa diluted in Alserver’s solution and DMSO gradually increased with increasing DMSO concentration (P<0.05). The optimal concentration of DMSO in Alserver’s solution was 20% for all storage durations. Spermatozoa stored in 5%, 10%, or 15% DMSO were immobilized after storage for 360 days, while those stored in 20% DMSO were immobilized after 720 days. The SAI of spermatozoa stored in Alserver’s solution and ethylene glycol gradually increased as the ethylene glycol concentration increased from 5% to 15%, but decreased at 20% ethylene glycol (P<0.05). The optimal concentration of ethylene glycol in Alserver’s solution was 15% for all storage durations. The spermatozoa were not immo bilized by storage in ethylene glycol for any storage duration.
*SAI: (Score × % motile sperm)/100 (After Strüssmann et al., 1994). The temperature of storage is –80℃. Mean±SD (n=100) values are shown. Means sharing the same letter superscript on given dates are not significantly different (P>0.05). Experiments are triplicate.
As shown in Table 3, the effects of dilution in glucose solution containing DMSO or ethylene glycol were similar to those of Alserver’s solution. The SAI of spermatozoa diluted in glucose solution and DMSO gradually increased with increasing concentration of DMSO (P<0.05). The SAI was lower in spermatozoa diluted in glucose solution plus DMSO than in spermatozoa diluted in Alserver’s solution plus DMSO. The optimal concentration of DMSO in glucose solution was 20% for all storage durations. Spermatozoa stored in 5%, 10%, and 15% DMSO were immobilized after 360 days, while those stored in 20% DMSO were immobilized after 720 days. The SAI of spermatozoa stored in glucose solution plus ethylene glycol gradually increased as the ethylene glycol concentration increased from 5% to 15%, but decreased when stored in 20% ethylene glycol (P<0.05). The SAI of spermatozoa stored in glucose solution plus ethylene glycol was lower than that of spermatozoa stored in Alserver’s solution plus ethylene glycol. The optimal concentration of ethylene glycol in glucose solution was 15% for all storage durations. Spermatozoa stored in glucose solution plus 5% or 10% ethylene glycol were immobilized after 540 days. By contrast, spermatozoa stored in 15% or 20% ethylene glycol were not immobilized at any storage duration.
The trends in hatching rates were similar to those for SAI and are shown in Table 4. The hatching rate in all of the experimental groups gradually decreased with increasing spermatozoa storage duration. The hatching rates of spermatozoa stored in Alserver’s solution and DMSO gradually increased with increasing concentration of DMSO (P<0.05). At all storage durations, the hatching rate was higher for spermatozoa stored in 20% DMSO than for those stored in other concentrations of DMSO. None of the eggs hatched after fertilization with spermatozoa stored in 5%, 10%, or 15% DMSO for 360 days. The hatching rates for eggs fertilized with spermatozoa stored in Alserver’s solution and ethylene glycol gradually increased as the ethylene glycol concentration increased from 5% to 15%, but decreased at 20% ethylene glycol (P<0.05). The hatching rate was higher for eggs fertilized with spermatozoa stored in 15% ethylene glycol than in other concentrations of ethylene glycol. All of the eggs hatched after fertilization with spermatozoa stored in ethylene glycol for any duration.
* The temperature of storage is –80℃. Mean±SD (n=100) values are shown. Means sharing the same letter superscript on given dates are not significantly different (P>0.05). The hatching rate (Hr) was calculated for each cage from the number of hatched larvae (Hl) and from the total number of eggs placed in the cage (Et) as follows: Hr = (Hl/Et) × 100 (after Linhart et al., 2005). Experiments are triplicate.
As shown in Table 4, the hatching rates of eggs fertilized with spermatozoa stored in glucose and DMSO gradually increased with increasing concentration of DMSO. At all storage durations, the hatching rate was highest for spermatozoa stored in 20% DMSO than in other concentrations of DMSO (P<0.05). None of the eggs hatched after fertilization with spermatozoa stored in 5%, 10%, or 15% DMSO for 360 days, whereas all the eggs hatched after fertilization with spermatozoa stored in 20% DMSO. The hatching rate of eggs fertilized with glucose solution and ethylene glycol gradually increased as the ethylene glycol concentration increased from 5% to 15%, but decreased at 20% ethylene glycol (P<0.05). The hatching rate was higher for eggs fertilized with spermatozoa stored in 15% ethylene glycol than in other concentrations of ethylene glycol. None of the eggs hatched after fertilization with spermatozoa stored in 5% or 10% ethylene glycol for 720 days. By contrast, the eggs hatched after fertilized with spermatozoa stored in 15% or 20% ethylene glycol for all of the storage durations.
The morphology of spermatozoa is shown in Fig. 2. Normal morphology was defined as the presence of a head and a flagellum (Fig. 2A, B), and the absence of a midpiece sleeve on SEM images. Immobile spermatozoa were classified into two types. The first type consisted of spermatozoa with a double-stranded flagellum (type A; Fig. 2C), and these originate from abnormal spermatogenesis before cryopreservation. In the second type, the flagellum is cut by ice crystals or damaged during thawing (type B; Fig. 2D). Therefore, type B spermatozoa are immobilized by external factors. The proportion of immobile spermatozoa after storage for 720 days is shown in Table 5. The proportions of type A spermatozoa were not significantly different among the experimental groups. The proportion of type B spermatozoa stored in Alserver’s solution was lower at 5% DMSO than at other concentrations of DMSO. The proportions of type B spermatozoa were not significantly different among 10%, 15%, and 20% DMSO. The proportion of type B spermatozoa stored in Alserver’s solution was lower at 5% ethylene glycol than at other concentrations. The trends in the proportions of type B spermatozoa were similar between ethylene glycol and DMSO.
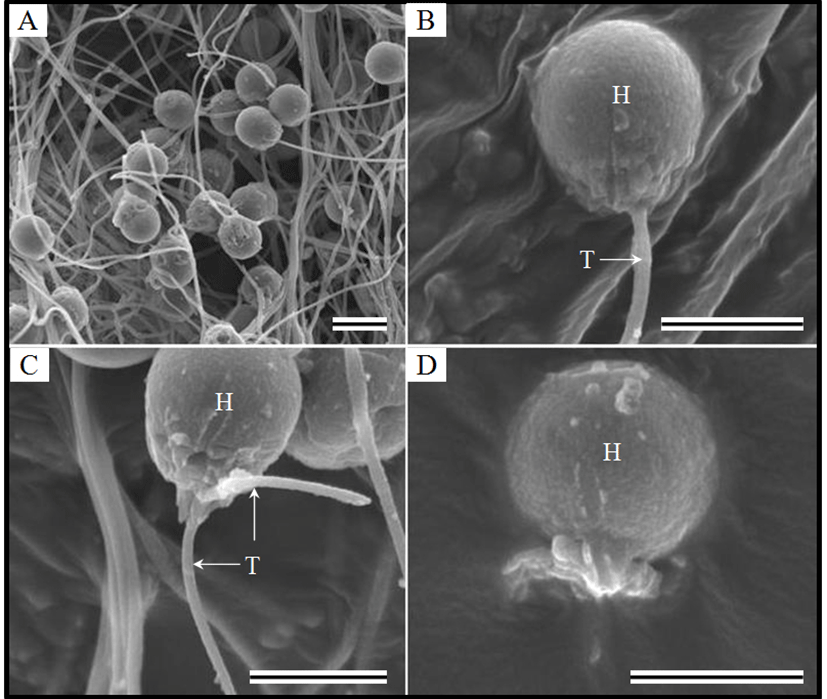
The proportions of type B spermatozoa were similar between those stored in glucose solution and Alserver’s solution when using DMSO. The proportions of type B spermatozoa were also similar between the two diluents when using ethylene glycol. However, the proportions of type B spermatozoa stored in Alserver’s solution or glucose solution were significantly lower in ethylene glycol than in DMSO. The proportion of immobile spermatozoa in the absence of a cryoprotectant was not significantly different between spermatozoa stored in Alserver’s solution or glucose solution. The proportion of immobile spermatozoa was significantly higher when stored without a diluent or a cryoprotectant compared with other groups. The proportion of immobile spermatozoa was lower for fresh sperm than for the other groups.
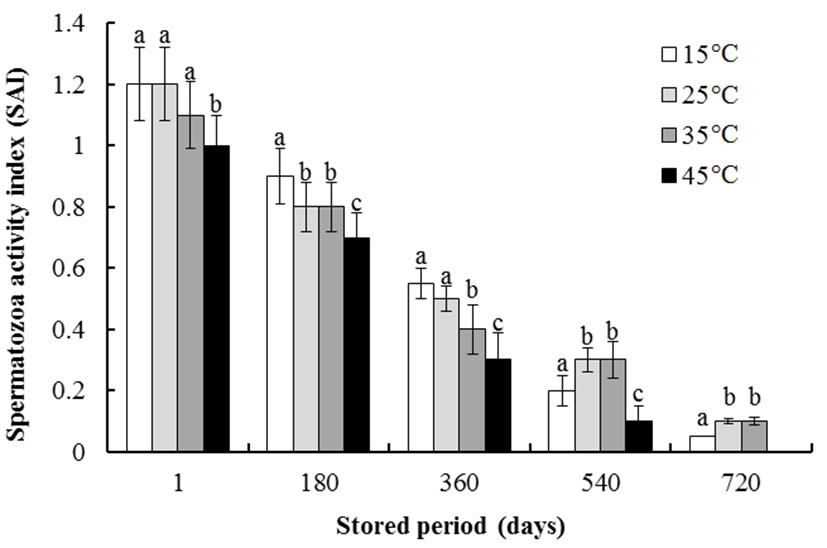
The effects of storage duration and thawing temperature on the SAI of spermatozoa are shown in Fig. 3. As thawing temperature increased, SAI gradually decreased as storage increased from 1 day to 360 days. However, SAI after storage for 540 days and 720 days increased as the thawing temperature increased from 15 to 35℃ (P<0.05). The spermatozoa stored for 720 days were not immobilized by thawing at 45℃. The SAI after storage for 540 days decreased as the thawing temperature increased from 35 to 45℃ (P<0.05). The mean SAI after storage for 540 days and thawing at 35 and 45℃ was 0.3±0.07 and 0.1± 0.07, respectively.
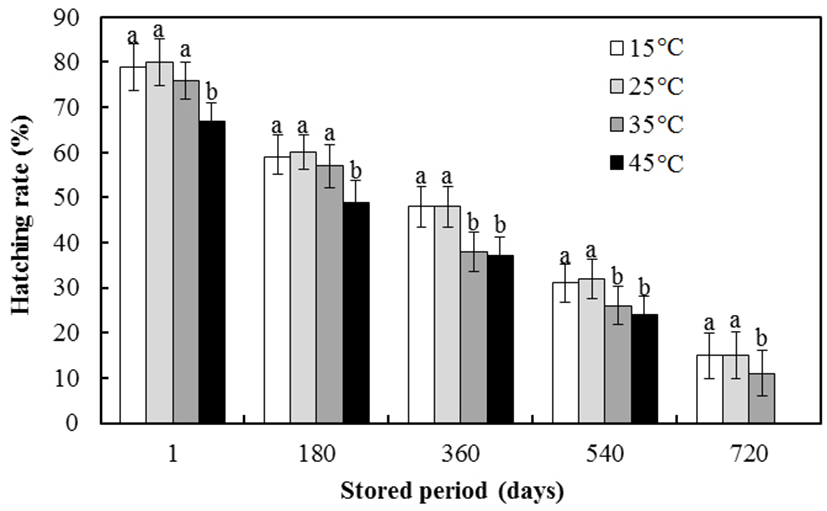
The effects of storage duration and thawing temperature on the hatching rate are shown in Fig. 4. The hatching rates gradually decreased with increasing thawing temperature for all storage durations (P<0.05). The hatching rates after storage for 1 or 180 days were not significantly affected by thawing temperatures of 15-35℃. Similarly, the hatching rates at 360, 540, and 720 days were not significantly affected by thawing temperatures of 15-25℃. At all storage durations, the hatching rates were higher when fertilizing eggs with spermatozoa thawed at 15 or 25℃ compared with the other thawing temperatures. Based on the SAI and hatching rates, our results suggest that the optimal thawing temperature of far eastern catfish spermatozoa is 25℃.
DISCUSSION
In our study of far eastern catfish, 104,000 spermatozoa per egg were dropped onto unfertilized eggs using a micropipette. When optimizing the freezing protocol of cryopreserved spermatozoa, it is necessary to use a small or moderate number of spermatozoa per egg. For European catfish, Silurus glanis, 80,00-80,000 fresh spermatozoa per egg are needed for artificial insemination, but hatcheries frequently use 100,000-200,000 spermatozoa per egg (Linhart et al., 2004). The use of a large number of spermatozoa per egg may mask the low viability or poor quality of thawed spermatozoa. Moreover, it may not be practical to use large numbers of spermatozoa in artificial propagation because male European catfish generally produce a small volume of sperm. Therefore, it is important to minimize sperm wastage. The dilution of sperm and eggs in water is another factor that must be adjusted. One part sperm to 25 parts water to 25 parts eggs was optimal for artificial propagation of European catfish in a previous study (Linhart et al., 2004).
The freezing procedure applied in our study was based on cryopreservation studies of tiger puffer, Takifugu rubripes, and European catfish, although the procedure was also successfully applied in far eastern catfish (Chang et al., 1997; Linhart et al., 2005). The cooling procedure was similar to that used for African catfish, Clarias gariepinus, where a three-step program achieved better survival of thawed spermatozoa compared with a two-step program (Steyn et al., 1993; Viveiros et al., 2001). The first step of the freezing program involved slow cooling from +4 to –40℃ at a rate of 2-10℃/min. While 5 min, the chamber temperature was maintained to –40℃, and the samples were immediately plunged into liquid nitrogen (from –40 to –200℃). As mentioned Steyn et al. (1993) and Viveiros et al. (2001), cooling rate of 5-11℃/min was considered to be optimal, and hatching rate was 81.7±5.0% in cooling rate of 5℃/min. In our study, cool rate was lower than that of previous study, however, hatching rate was similar to that of previous study. All previous studies have reported that the final temperature and its duration (i.e. holding period) just before plunging the frozen sperm into liquid nitrogen are very important. During the holding period, the temperature in the freezer remains constant, but the temperature of the freezing sperm continues to decrease until it reaches an equilibrium with the diluent. Most of the freezing protocols for catfish cool the spermatozoa to a low temperature before transferring them into liquid nitrogen. For African catfish, Steyn et al. (1985) used a holding temperature of –65℃ and Horvath & Urbanyi (2000) used a temperature of –80℃. For European catfish, Linhart et al. (1993) used a holding temperature of –85℃. A holding temperature of –80℃ was used for striped catfish, Pangasius hypophthalmus, by Kwantong & Bart (2003) and for channel catfish, Ictalurus punctatus, by Tiersch et al. (1994) and Christensen & Tiersch (1997). Frozen spermatozoa were thawed at 40℃ in most of those studies, while we used a temperature range of 25-40℃, consistent with earlier studies.
The use of ethylene glycol as the cryoprotectant resulted in significantly greater hatching rates and percentage of live spermatozoa compared with DMSO. To our knowledge, no prior studies have optimized the cryoprotectant composition for cryopreservation of catfish spermatozoa. Although many cryoprotectants have been tested for fish spermatozoa, those most commonly used for cryoprotection of catfish spermatozoa include DMSO, glycerol, methanol, dimethyl acetamide, ethylene glycol, and propylene glycol (Christensen & Tiersch, 1997; Linhart et al., 2005). DMSO was originally recommended for striped catfish, blue catfish (I. furcatus), African catfish, and channel catfish (Guest et al., 1976; Bart et al., 1998; Horvath & Urbanyi, 2000; Kwantong & Bart, 2003). According to the present results, 15% ethylene glycol is suitable for far eastern catfish. Recent studies of African catfish and channel catfish showed greater sperm motility and fertility of thawed sperm using methanol as a cryoprotectant (Tiersch et al., 1994; Christensen & Tiersch, 1997; Horvath & Urbanyi, 2000).
Taddei et al. (2001) reported that osmotic shock derived from high concentrations of the cryoprotectant (15% vs. 10%) increased the proportion of abnormal spermatozoa, as did cold shock during pre-freezing (Muchlisin & Siti Azizah, 2009). Osmotic shock injury of freeze-thawed spermatozoa is characterized by coiling and swelling of the distal end of the tail (Muchlisin & Siti Azizah, 2009). Lahnsteiner et al. (1992) reported that the morphological changes occurred before freezing and immediately after dilution with the cryosolution, and that the frequency of morphological changes increased with longer time in cryosolution. For freeze-thawed spermatozoa, osmotic shock refers to the cellular changes that occur after transferring spermatozoa from hypertonic conditions in a cryoprotectant, such as glycerol to isotonic conditions after thawing (Correa & Zavos, 1996). However, abnormalities were found in cryopreserved spermatozoa as well as fresh spermatozoa. In our study, 14.0% of fresh spermatozoa of far eastern catfish exhibited morphological abnormalities, which was comparable with that of baung, Mystus nemurus, for which 13.96% of spermatozoa exhibited abnormalities (Muchlisin & Siti Azizah, 2009). We suspect that these abnormalities occurred during spermatogenesis.
The rate of thawing may be as important as the cooling rate in terms of the viability of cryopreserved spermatozoa (Christensen & Tiersch, 2005). In previous studies of blue catfish, the spermatozoa were thawed as 0.5 mL samples in French straws at 40℃ for 7 sec (Lang et al., 2003), a method that was originally developed for mammalian spermatozoa (Pickett & Berndtson, 1974). In the present study, it took 90 sec for the samples to completely thaw at 25℃ in a water bath. It may be more convenient and economical to use tap water rather than a water bath to thaw spermatozoa. We found that thawing at 15 or 25℃ did not affect spermatozoa mobility, but, if time is limited, 25℃ should be considered, especially when large numbers of samples need to be processed or if there are time constraints in the hatchery. Cryopreservation of sperm is a useful and reliable technique for conservation of gene resources of far eastern catfish and for artificial propagation.

