INTRODUCTION
High-fructose corn syrup (HFCS) is a sweetener made from corn starch that has been processed by glucose isomerase to convert glucose into fructose (Ma et al., 2017). HFCS has been widely used as caloric beverages, and is also a primary ingredient in baked goods, cereals, breads, canned fruits, ketchups, jams and jellies, desserts, and fruit juices (Hanover and White, 1993). There is growing apprehension that the overconsumption of added sugar, including HFCS, is a major health problem, especially for onset of obesity and type 2 diabetes mellitus (T2DM) (Bray et al., 2004; Lakhan & Kirchgessner, 2013; Toop et al., 2015). Furthermore, several lines of evidence indicate that obesity is associated with multiple adverse reproduc-tive outcomes in female (Broughton & Moley, 2017).
Meanwhile, recent studies have shown that the high fat diet (HFD) exposure can induce puberty onset and estrous cycle irregularity in rodent models (Li et al., 2012; Ullah et al., 2017). We also demonstrated that advanced onset of puberty in HFD-fed immature female rats, and this phenomenon is closely related to the activation of KiSS-1 and GnRH expression in the Hypothalamus (Lee et al., 2009). Taken together, these findings strongly support the idea that excessive caloric intake is associated with disturbances of female reproductive function. Since HFCS overconsumption is believed to induce childhood obesity, it could be postulated that HFCS might have similar impact as HFD feeding on the female reproductive system. However, this aspect has not been addressed so far. In the present study, we used adult female rats and applied a 28 days HFCS feeding model to monitor the estrous cycle and changes in tissue weights and histology.
MATERIALS AND METHODS
Sprague-Dawley rats were obtained from DBL (Chungcheongbuk-do, Korea) and reared in our animal facility under conditions of 12-h light/dark cycle (lights on at 07:00 h) and constant temperature of 22±1℃. All procedures used were approved by the Animal Care and Use Committee at Sangmyung University in accordance with guidelines established by the Korea Food and Drug Administration (KFDA).
Age-matched female rats (3 months old, 250-275 g, n= 10/ group) were divided into three groups. Animals were fed with ad libitum normal chow (AIN-93G, DBL) and (1) 24 hours tap water (Control group), (2) 12 hours HFCS access during dark period and 12 hours tap water (12H group), and (3) 24 hours HFCS only access (24H group). Total feeding period was 28 days. HFCS was purchased commercially available one (CJ Cheiljedang, Formula 55, v/v dissolved in tap water, 0.24 Kcal/ml), and the working solution was diluted finally to 8%. The stage of the estrous cycle was determined through daily microscopic examination of vaginal smear samples. After 28 days of feeding, the animals were sacrificed by decapitation, and the tissues were collected and weighed.
Tissues were fixed in 4% paraformaldehyde overnight at 4℃ for 24 h. Fixed tissue were dehydrated in ethanol (70%, 80%, 90%, 95%, 100%) and embedded in paraffin block. The tissues blocks were cut at 4-5 μm using microtome (HM350S, MICROM, Germany). Sections were stained with hematoxylin-eosin and observed using a light microscope (BX51, Olympus, Japan).
RESULTS
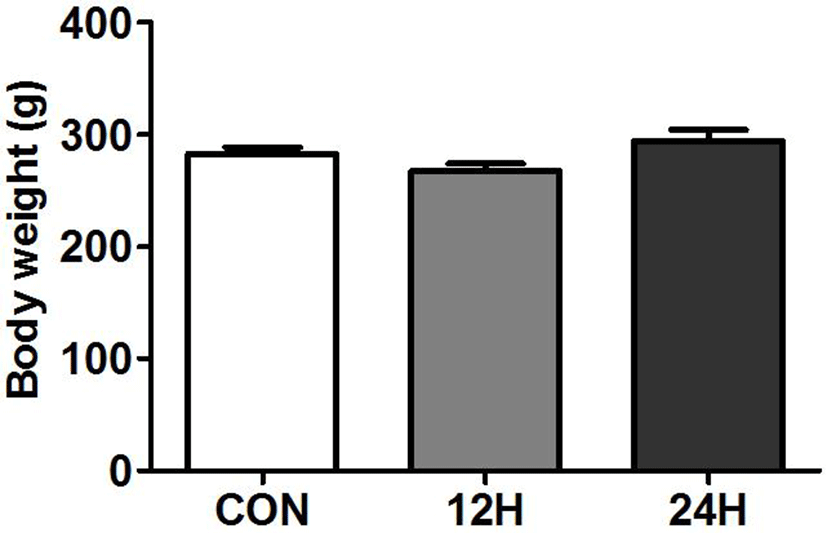
Fig. 1 depicts the final body weights of normal chow-fed (CON) and HFCS-fed(12H and 24H) female rats after 28 days feeding. There is no significant change in body weightbetween control and HFCS-fed animals (control vs 12H vs 24H = 282.5±5.0 vs 267.2±6.9 vs 294.2±9.9).
Daily vaginal smear test showed that the estrous cycles of the whole day HFCS feeding animals (24H) were significantly longer than control and half day HFCS feeding animals (12H) (control vs 12H vs 24H = 4.7±0.3 vs 4.8± 0.2 vs 5.6±0.2. P<0.05; Fig. 2 The lengths of estrous and diestrous stages were prolonged in 24H group animals (data not shown).
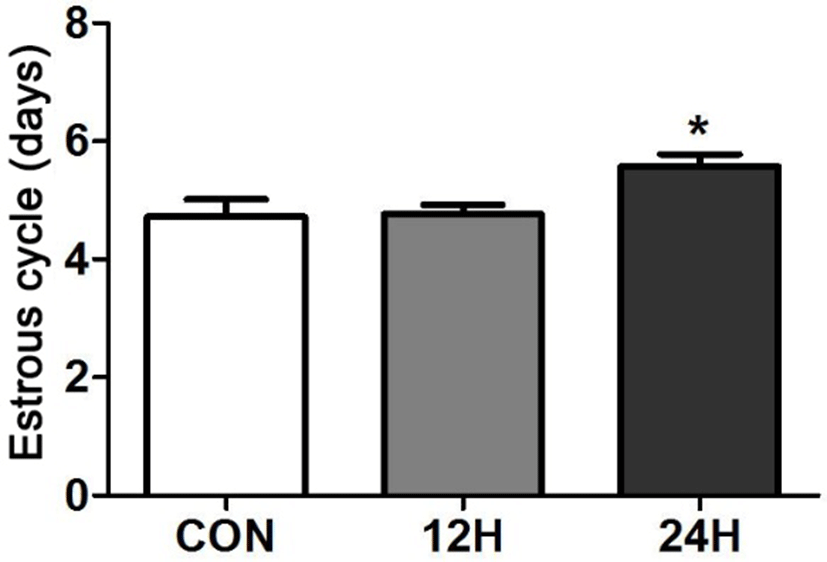
Table 1 presents the effects of HFCS feeding on changes in tissues weights. Both absolute and relative weights of ovary in 24H animals were significantly heavier than those in control or 12H animals (absolute weight : control vs 24H = 38.0±2.0 vs 47.5±2.8, p<0.05; relative weights : 13.4±0.6 vs 16.1±0.8, p<0.05). Among the non-reproductive tissues, the absolute and relative weights of the kidney in HFCS-fed groups were signifi-cantly heavier than those in control animals (absolute weights : control vs 24H = 794.9±13.3 vs 898.9±20.4, p<0.001; relative weights : control vs 12H = 281.8±0.5 vs 303.5±0.6, p<0.05; control vs 24H = 281.8±0.5 vs 306.5±0.7, p<0.01). Likewise, the absolute and relative weights of the liver in HFCS-fed groups were significantly heavier than those in control animals (absolute weights : control vs 24H = 7.46±0.13 vs 8.38±0.33, p< 0.05; relative weights : 2.64±0.62 vs 2.85± 0.34*, p<0.05). In other tissues (uterus, spleen and pancreas), no significant change in the tissue weights was found.
CON, control group. 12H, 12 hr HFCS accessed animals; 24H, 24 hr HFCS accessed animals; Rel, Relative tissue weights, and is tissue weight (mg) / body weight(g), except liver which is tissue weight (g) / body weight (g).
Values are expressed as mean ±S.E. Groups marked with an asterisk (*) are significantly different from control group, p<0.05 (** ; p<0.01, ***; p<0.001).
Fig. 3 represents the cross sections of the ovary, uterus, kidney and liver tissues used in the present study. The overall size and shape of the 24H ovaries were relatively bigger than control ovaries. More corpus lutea were found in 24H ovaries, while secondary follicles were more abun-
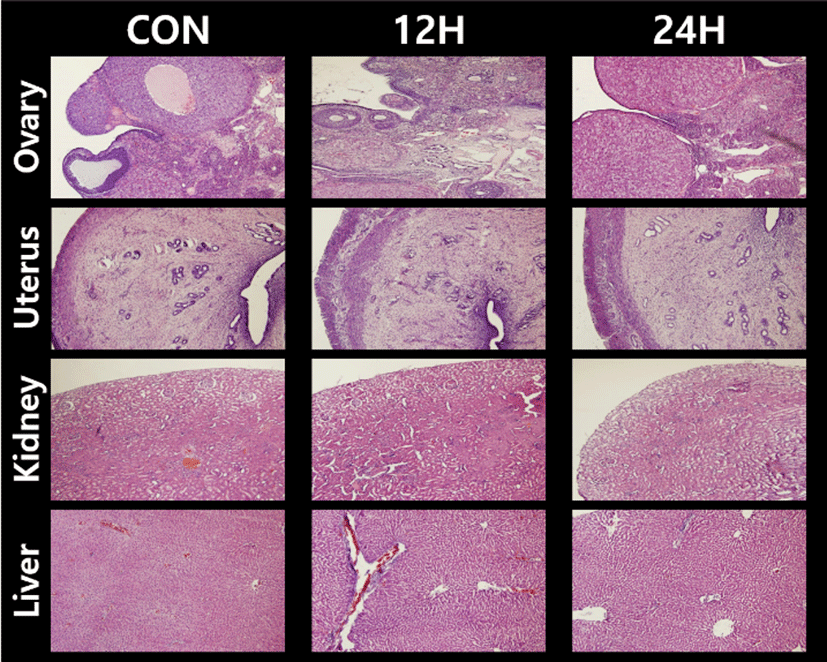
dant in control ovaries. The thickened epithelia covering the tissue surface were found in 12H ovaries. Uterine sections of 12H and 24H animals showed a well-developed stratum vasculare between prominent inner and outer myometrial layers. The numbers of endometrial glands were decreased in 12H uteri, and recovered in 24H uteri compared to control. Kidney sections revealed that the numbers of convoluted tubule in distal region considerably increased in both 12H and 24H samples. Liver specimens of 12H and 24H animals showed that the numbers of fat containing vacuoles increased compared to control samples.
DISCUSSION
Numerous agencies in the United States, such as American Heart Association and the Departments of Agriculture and Health and Human Services, recommend reducing the consumption of all sugars including HFCS. However, there is ongoing debate about the role of HFCS in the spreading obesity epidemic. There is an evidence for a detrimental effect of soft drink consumption on body weight in childhood (Libuda & Kersting, 2009; Morgan, 2013). Another study also suggests that the increase in consumption of HFCS has a temporal relation to the epidemic of obesity, and the overconsumption of HFCS in calorically sweetened beverages may play a crucial role in the epidemic of obesity (Bray et al., 2004). More specifically, HFCS may contribute to the pathogenesis of nonalcoholic fatty liver disease (NAFLD) which is associated with obesity and insulin resistance (Collison et al., 2009). On the contrary, there is a review study argued that only insufficient evidence are available to ban or restrict use of HFCS in the food supply or to require warning labels on products containing HFCS (Moeller et al., 2009). More recent study also indicates that the available evidence is not sufficiently robust to draw conclusions regarding effects of fructose, HFCS, or sucrose consumption on NAFLD (Chung et al., 2014). In the present study, we found that the HFCS feeding failed to change the body weight, while HFCS intake induced renal and liver lipid accumulation. The total period of HFCS treatment in our study was for 28 days, which was much shorter than those used in similar studies (Bocarsly et al., 2010; Merino-Aguilar et al., 2014). Treatment for 28 days could be not enough to induce body weight change when compare to over 8 weeks treatments used in above studies. However, our studies on the kidney and liver his-tology assure the adverse effect of HFCS consumption on these tissues. The precise measurements of daily total ca-lorie intake and the ratio of sugar molecules (i.e., fructose, sucrose and glucose) seem to be important factors which can determine the metabolic pathway and the ultimate effect of HFCS consumption.
Concerning the reproductive health, previous study demonstrated that the rats drinking HFCS-55 for 8 weeks showed lengthened estrous cycles due to prolonged estrus (Light et al., 2009). Our result that HFCS access for 4 weeks could induce extended length of estrous cycle is in good agreement with that. Our histological analyses also demonstrated that the changes in ovarian and uterine histology suggest female reproductive function could be altered by HFCS intake. It is well known that female obesity is closely associated with multiple adverse reproductive out-comes in female (Broughton & Moley, 2017). For example, HFD induced obesity is a trigger of premature puberty onset in immature female rats (Lee et al., 2009), and obesity impairs ovulation but has also been observed to detrimen-tally affect endometrial development and implantation (Brewer & Balen, 2010). Special attention should be paid to polycystic ovarian syndrome (PCOS) due to its high inci- dence in fertile female population. The expression of PCOS is regulated, in part, by weight, and so obese women with PCOS often have a more severe phenotype and experience more subfertility (Brewer & Balen, 2010). It will be intri-guing to elucidate whether HFCS overconsumption or high calorie intake from HFCS induces PCOS or aggravates the symptoms.
In conclusion, our study demonstrates that relatively short-term HFCS treatment could induce (1) changes in length of estrous cycle with extended estrous and diestrous stages, (2) altered ovarian and uterine histology, and (3) liver and renal lipid accumulation. As the estrous cycle is lengthened, the rats will become acyclic, a status of reproductive senescence (Westwood, 2008). Despite of the short experimental period, these findings reveal the adverse effects of HFCS drinking on the reproductive function and lipid metabolism of female rats. Further researches in this field will be helpful for understanding the relationship between unhealthy life style (such as overconsumption of HFCS) and obesity, and for maintaining sound reproductive function.

