INTRODUCTION
The uterus is essential organ for reproduction of most mammals, and regulated by several specific steroid hormones as estrogen (E2) and progesterone (P4) in endometrium during uterus cycles (Martıín et al., 2002; Riesewijk et al., 2003). Endometrium, one of the three layers of the uterus, is the most active layer and composed with luminal epithelium (LE), glandular epithelium (GE), and stroma (Hu et al., 2004; Huang et al., 2012). Repeated implantation failure (RIF) is occurred due to dysfunction of uterine receptivity with abnormal gene expression in the endometrium. Recent studies attempt to fine the cause of fertilization failure, but it remains as yet unknown due to lack of research on the signaling pathway.
The E2 plays important roles in physiological regulations in the whole body including reproductive organ, central nerve system, skeletal tissues and vascular (Gruber et al., 2002; Hewitt et al., 2005). E2 activities are mediated by the estrogen receptor alpha (ERα) and estrogen receptor beta (ERβ). E2 and ERs are up regulated by insulin resistance, and insulin resistance-mediated inflammation might be modulator for estrogen activity (Calle & Kaaks, 2004). cyclic AMP-response element binding protein zhangfei (CREBZF) is an insulin-inducible co-repressor during gluconeogenesis in hepatocyte, and insulin resistance decreases CREBZF expression (Lee et al., 2016).
CREBZF belongs to basic region of leucine zipper (bZIP) family, and it has the ability to dimerize like other bZIP family (Akhova et al., 2005; Misra et al., 2005; Xie et al., 2008). CREBZF has been reported as a co-regulator of ATF4 and CREB3, another HCF-binding transcription factor (Misra et al., 2005; Hogan et al., 2006). Recently, CREBZF regulates bone formation and adipocyte differentiation by interacting transcription factors (Jang et al., 2014; Jang et al., 2015). Also, recent study indicated that CREBZF is induced by estrogen and closely related to implantation (Lin et al., 2013). However, this study is unclear and lacks the correlation between estrogen and CREBZF expression in endometrium of uterus. To discover the function of CREBZF during menstrual cycle, we examined the expression of CREBZF in endometrium during mouse estrous cycle.
MATERIALS AND METHODS
All animal experiments were approved by Institutional Agricultural Animal Care and Use Committee of CHA University (IACUC No. 160018). The animal studies were conducted with 3-week old and 6 week old female ICR mice from KOATECH (Korea). Mice were housed in animal care facility at the CHA University of CHA biocomplex with a temperature-controlled environment under 12 hour dark-light cycle and free access to standard rodent diet and water. Six-weeks old female ICR mice were ovariectomized (OVX) and then following a recovery period for 2 weeks.
Three-week old and OVX female ICR mice were injected subcutaneously with 17β-estradiol (E2, 10 ug/kg, Sigma-Aldrich, USA) and Sesame oil (100 uL, Acros, USA) as control group. The Uteri were collected at 0, 1, 3, 6, 12, and 24 hours after injection of hormones from sacrificed mice. To identify whether the expression of CREBZF is regulated by E2 administration, an estrogen receptor antagonist (ICI 182,780, 25 mg/kg) was co-injected to OVX mice.
To obtain a sample of epithelial cells from the mouse vagina some of the saline solution was flushed into mouse vagina using pipette tip to a depth of about 5 mm. The isolated cells were placed on glass slide and dried. The cells were fixed using methanol and stained by Hematoxylin and eosin solution. The stage of the estrous cycles were determined by vaginal smear cytology (Evans & Long, 1922).
To extract total RNA from four cycles of uterus and each of tissues, 6-week old female ICR mice were sacrificed and isolated after analysis of vaginal cytology. The tissues were placed into a vessel and homogenized in Trizol reagent (Invitrogen, USA) using homogenizer. The total RNA extraction was according to manufacturer’s protocol. cDNA synthesis was performed using SensiFAST cDNA Synthesis Kit (Bioline, UK) according to manufacturer’s protocol, and the thermal cycling conditions were as follows: reverse transcription at 45℃ for 60 minutes, and 4℃ for 10 minutes. Real-time PCR and RT-PCR analyses were performed in triplicate using Solg Taq DNA polymerase (Solgent, Korea) and iQ SYBR Green Supermix (Bio-Rad, USA). The thermal cycling conditions were as follows: initial denaturation at 95℃ for 3 minutes, followed by 40 cycles of 95℃ for 10 seconds, 60℃ for 15 seconds, and 72℃ for 20 seconds. Expression levels were normalized to those of endogenous glyceraldehyde 3-phosphate dehydrogenase (GAPDH).
Transfected 293T cells were prepared using PRO-PREP solution (iNtRON Biotechnology, Korea) according to manufacturer’s protocol. Western blot was performed using anti-CREBZF antibody (Santacruz, USA) and β-Actin (Santacruz, USA).
Uteri isolated from mice were fixed for 1 week in 4% formaldehyde at 4℃ for immunostaining. After fixation, uteri were embedded in paraffin and sectioned at 5 μm of thickness using a microtome and put on microscope slides (HistoBond, Germany). Sections were deparaffinized as follows; slides were dipped three times for 5 min in xylene (Biosesang, Korea), two times for 5 min in 95% ethanol, one time for 5 min in 90 %, 80 %, 70 %, 50% ethanol, and 10 min in distilled water. For immunofluorescence, deparaffinized slides were placed into a Retrieval Steamer (IHCworld, USA) and antigen retrieval was performed using an Epitope Retrieval Solution (IHCworld, USA) according to the manufacturer’s protocol. After washing in PBS, excess PBS was removed, and blocking buffer (4% BSA and 5% rabbit serum in PBS) was added to the slides. The slides were incubated in a humidified chamber for 4 h at room temperature (RT). Then, the slides were incubated with the following primary antibodies for 16 h at 4℃: Goat polyclonal antibody against CREBZF (1:1000, Santacruz, USA), rabbit polyclonal antibody against PR (1:500, Santacruz, USA). After three washes in PBS, the slides were incubated with Alexa 488 mouse anti-Goat and Alexa 546 mouse anti-rabbit (1:1,500, Invitrogen, UK) for 2 h at RT. A 4’,6-diamidino-2-phenylindole (DAPI, 1:20000, Life Technologies, USA) was used to stain the nuclei. Mounting medium (DAKO, USA) was applied to the tissue sections prior to covering them with glass coverslips. All images were obtained using a confocal microscope (Leica) and analyzed by the imaging software LAS lite (Leica). For immunohistochemistry, antigen-retrieved sections were washed with PBS with 0.5% Tween 20 (PBS/T) and incubated in 0.5% hydrogen peroxide in distilled water for 10 minutes (min) to quench endogenous peroxidase activity. Then, the slides were washed two times in cold PBS/T for 10 min. Blocking was conducted using blocking solution (4% BSA and 5% rabbit serum in PBS/T) for 2 h at RT. The sections were incubated in the previously described primary antibody against CREBZF (1:1,500) for 16h at 4℃, followed by incubation in diluted rabbit HRP-conjugated anti-goat antibody (1:500, Invitrogen, UK) for 1 h at RT. The slides were washed in PBS/T three times for 5 min and put into distilled water. A 3,3’-diaminobenzidine (DAB) solution (Vector Laboratories, USA) was dropped on the sections until the desired staining was obtained, according to the manufacturer’s manual. The sections were counterstained with hematoxylin and dehydrated using ethanol and xylene. The mounting was performed using Permount mounting medium (Fisher Scientific, USA), and then coverslips were added.
RESULTS AND DISCUSSION
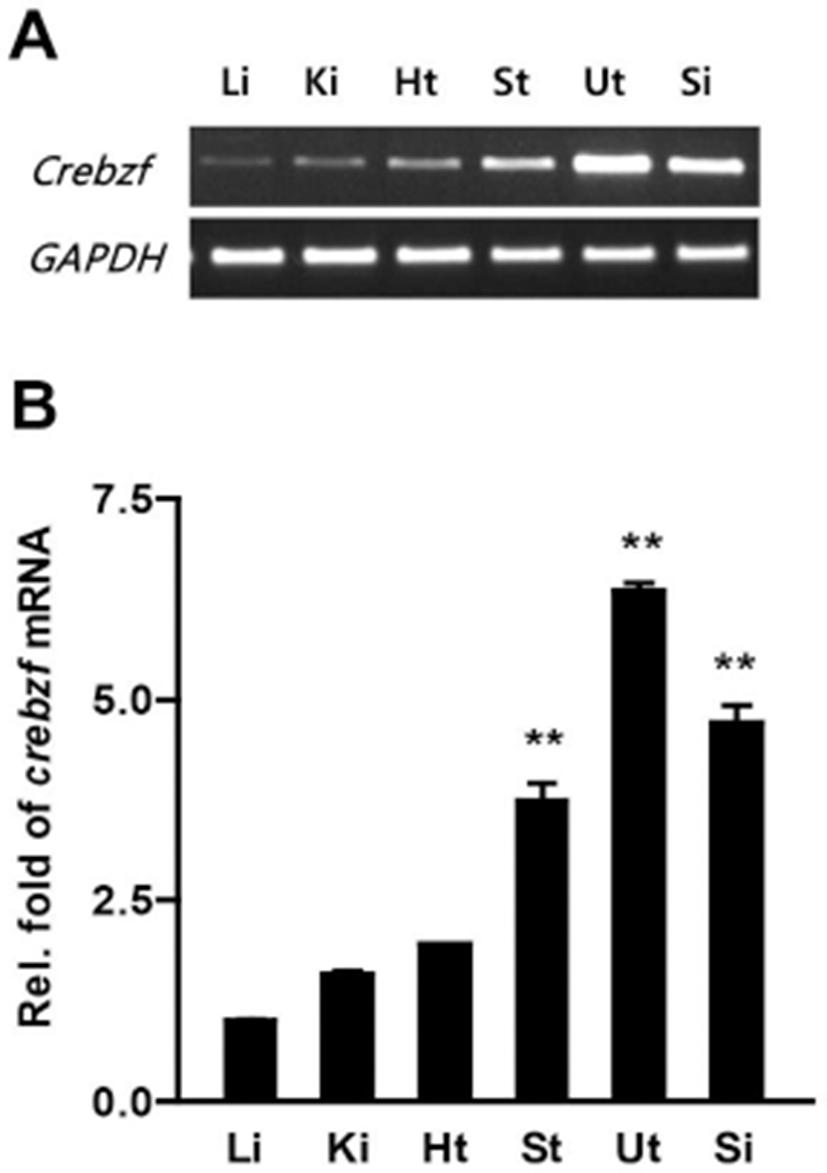
To examine expression levels of CREBZF transcript in various tissues, we performed RT-PCR and qRT-PCR using respective cDNA from 6-weeks old mice organs and designed mouse CREBZF primers (Table 1). The CREBZF transcript was detected in various tissues and was highly expressed in uterus than other tissues in adult mice (Fig. 1A-B). Recent studies showed that CREBZF expression is regulated by insulin and regulates hepatic gluconeogenesis (Kim & Ahn, 2016; Lee et al., 2016). These results suggest that CREBZF plays major roles in liver tissue and that expression is also high in liver tissue. Therefore, we examined the expression of CREBZF in several tissues of mice based on liver tissue, and found that CREBZF expression is highest in the uterus. Taken together, our results indicated that CREBZF has an important role in uterus.
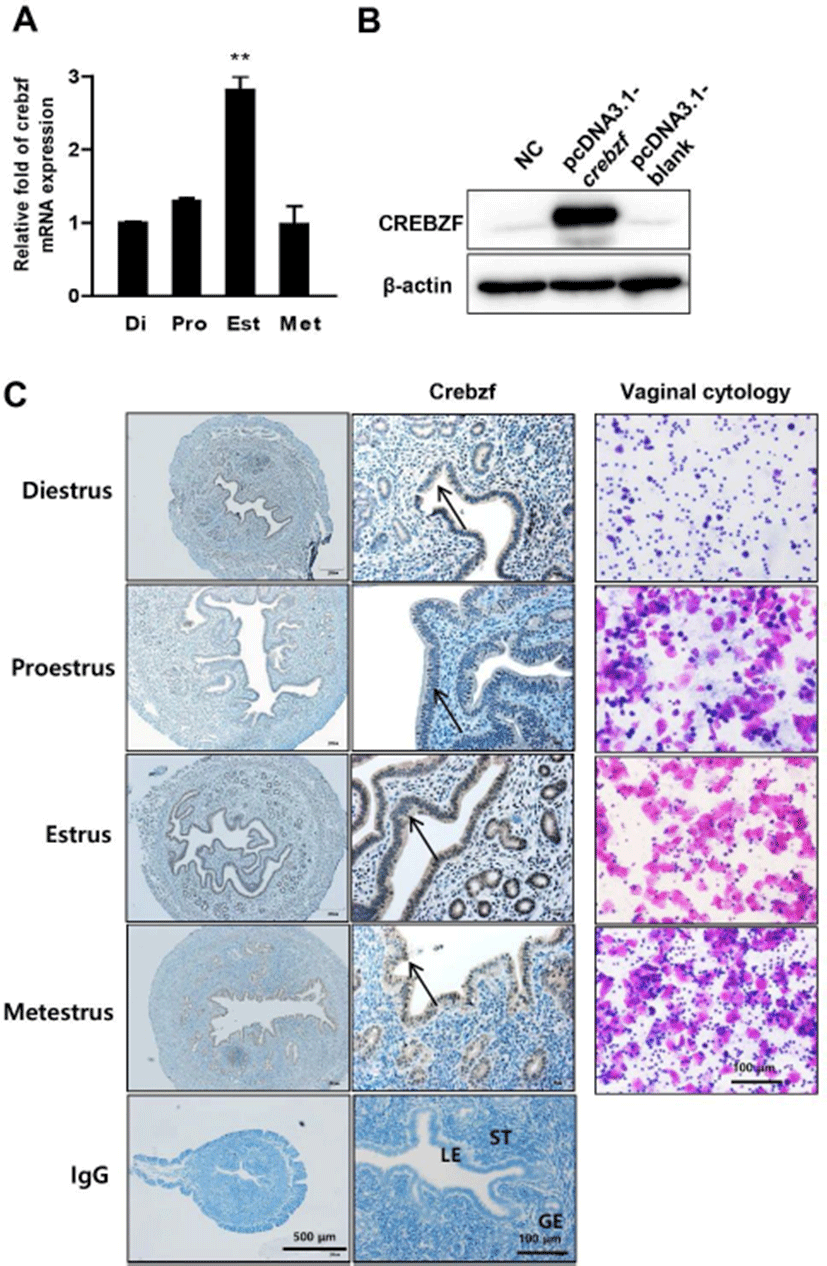
To evaluate expression patterns of CREBZF in the uterus, we identified changes in expression over the development of the estrous cycle such as diestrus, proestus, estrus, and metestrus. Semi-quantitative PCR showed that CREBZF was significantly increased at estrus stage (Fig. 2A). Because the estrogen is dramatically decreased at estrus stage (Miller & Takahashi, 2014), it suggested that CREBZF is sensitively regulated by estrogen. To obtain definite results, we performed an antibody test by western blot using 293T cells transfected with the mouse CREBZF over-expression vector and found that the antibody was well working (Fig. 2B). To evaluate the localization of CREBZF protein during estrous cycle, we conducted immunohistochemistry analysis in uterine sections collected from all stages of the estrous cycle using CREBZF antibody. CREBZF expression was more highly expressed in the LE and GE than stroma cells in the endometrium of uterus at the estrus stage (Fig. 2Cleft panel). Each stage of the estrous cycle was identified by the ratio of leukocytes and epithelial cells in the vagina using hematoxylin and eosin staining method (Fig. 2Cright panel). In the uterus, the endometrium is dynamically regulated throughout the estrous cycle and it consists of the LE, GE, and the stroma. The proliferation and differentiation of endometrium were closely controlled by two major ovarian hormones, E2 and P4. The sexual steroid hormones from ovary controlled estrous cycle which is critical for a pregnancy due to modulate uterus. Circulating E2 levels are increased at the proestrus stage and then decline during estrus stage (Walmer et al., 1992; McLean et al., 2012), whereas P4 begin to elevate after estrus and peak at diestrus (McLean et al., 2012; Miller & Takahashi, 2014). Also, E2 stimulates uterine cell proliferation and P4 stimulates uterine cell differentiation (Finn & Martin, 1974). In addition, numerous studies demonstrated that E2 and P4 regulates various signaling factors (Altmäe et al., 2010; Cha et al., 2012; Choi et al., 2016; Rosario et al., 2016). Taken together, our results suggest that CREBZF expression is closely regulated by blood E2 levels in epithelial cells of endometrium.
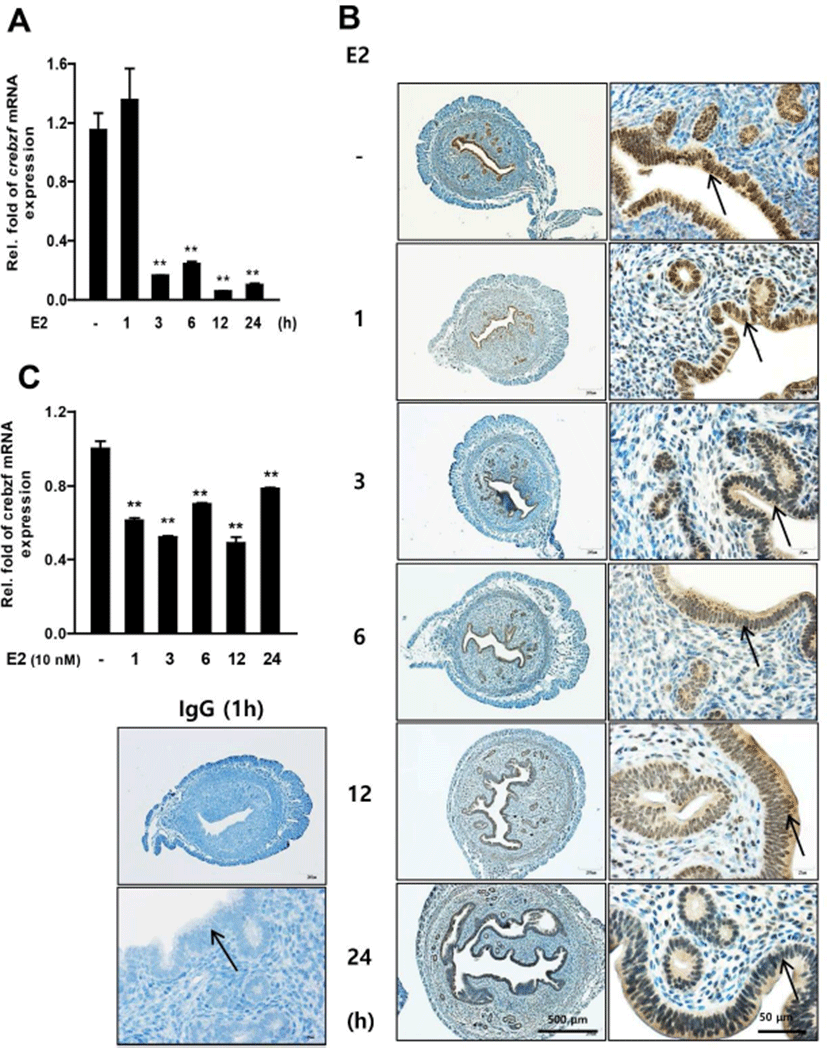
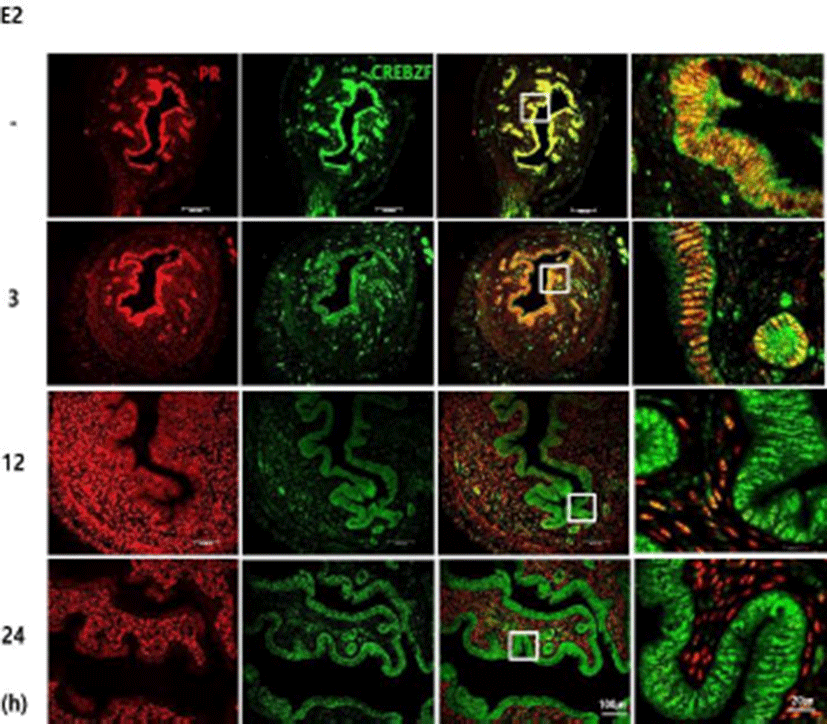
To investigate whether CREBZF is regulated by E2 treatment in the uterus of prepubertal mice, we performed E2 injection to 3-week-old female ICR mice. Semi-quantitative PCR and immunohistochemistry were performed using collected uterus at various time points. The results showed that E2 treatment dramatically reduced CREBZF mRNA expression from 3 hours after administration (Fig. 3A). CREBZF protein expression was specifically declined by E2 in only epithelium of endometrium (Fig. 3B). To further confirm that CREBZF is down-regulated by E2 in other cells, E2 was treated to Ishikawa cells, a human endometrial cell line. Similar to the previous results, E2 reduced human CREBZF mRNA expression level (Fig. 3C). Next, to further determine whether CREBZF was reduced by E2 in OVX mice, we treated the E2 to OVX mice and collected the uteri at various times. A progesterone receptor (PR), an intra-uterine E2 marker, was also identified to conform whether E2 was administrated correctly. Immunofluorescence showed that expression of CREBZF was decreased in the OVX mouse endometrium while it was expressed only in epithelium, but the expression of PR markedly decreased in epithelium and dramatically increased in stroma after 12 hours of E2 injection (Fig. 4). Previous studies have shown that the E2 response has progressed from about 2 hours after E2 injection (Seong et al., 2014). In light of that study, it is possible to interpret the result of qRT-PCR that did not affect the expression of CREBZF in uterine samples after 1 hour of E2 injection (Fig. 3A). In addition, since the reaction by E2 is rapid in cell based experiments, the expression level of CREBZF is decreased even at 1 hour (Fig. 3C). Interestingly, our results indicate that CREBZF is expressed specifically in endometrial epithelium, whereas PR is expressed in the epithelium when the E2 concentration is low, but is specifically expressed in the stroma when the E2 concentration is high Respectively (Fig. 4). These results indicate that the expression of PR is rapidly decreased when blood E2 level is elevated, thereby controlling the environment of endometrial epithelium. The CREBZF has been reported as a co-regulator without a DNA binding domain and plays a role through interactions with major regulators to perform most roles (Misra et al., 2005; Hogan et al., 2006; Jang et al., 2015). Recently, Seo et al have suggested the possibility of role of the transcription factor egr1, an important regulator of cell growth, differentiation, and apoptosis, in the uterus (Seo et al., 2014). These results suggest that CREBZF may interact with critical factors as egr1 or PR.
In order to examine whether CREBZF expression is affected by E2 in estrogen receptor dependent (ER) manner, OVX ICR mice were co-treated with an ER antagonist, ICI 182,780 (ICI), and E2. As shown in Fig. 5, immunohistochemistry presented that ICI treatment significantly restored E2-induced CREBZF suppression. Most induction of target factors by hormones occurs via hormone receptor, stimulated by extra nuclear actions. We demonstrated that the CREBZF expression is regulated by the ER signaling pathway through experiment of co-administration of ICI.
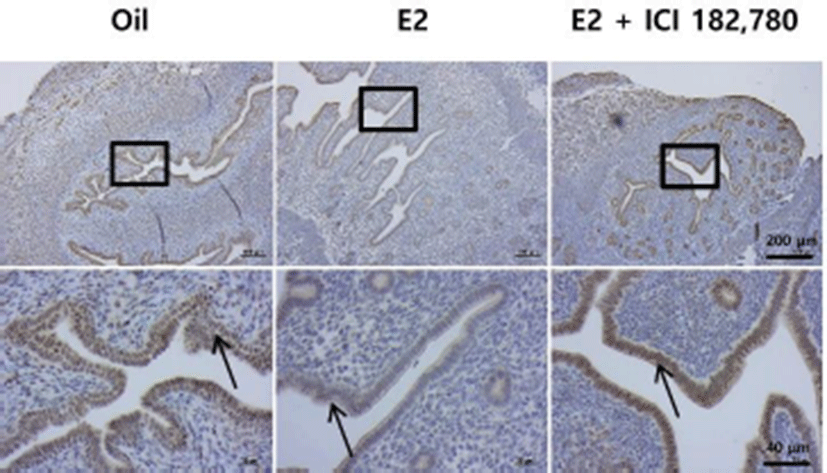
Further study will be conducted to determine what mechanism is involved in CREBZF among various ER signaling pathways in endometrium.
In conclusion, we showed that CREBZF was specifically expressed in the uterine epithelium and its expression was regulated by E2 levels, and also, the CREBZF expression is moderated by ER signaling pathway. It will give us better understanding the function and mechanism of CREBZF in dynamic cycles of uterus.

