INTRODUCTION
Recently, a vast amount of information has been accumulated regarding to the role of adult stem cells in tissue regeneration. Many researches have focused on the regenerating ability of stem cells following implantation into the body. In clinical applications, cells are either introduced into the vessels or directly to the tissues or organs (Galderisi & Giordano, 2014). Once cells are implanted, one of the important factors to guarantee the regenerating ability is a homing property to target tissues. In response to myocardial infarction, endothelial progenitor cells are mobilized from bone marrow (BM) and then recruited to the site of injury. At the injured site, they are believed to help repair by inducing vasculogenesis via secreting paracrine factors (Losordo & Vaughan, 2014). While the treatment has been applied frequently, only a few of transplanted cells showed to retain in the ischemic myocardium. Thus efficient migration of implanted cells to the targeted tissues appears to be one of most important issues in cell therapeutics (Chavakis et al., 2010).
Unlike BM-derived mesenchymal stem cells (MSCs) that require surgical operation to isolate, human adipose-derived stem cells (hADSCs) have easily been obtained from lipoaspirates during plastic surgery (Liao & Chen, 2014). Due by this convenience, hADSCs have been studied extensively for the possible clinical application. During culture in vitro, hADSCs show abilities to migrate on the plastic dish surface as revealed by wound healing assay or transwell chamber assay (Kabiri et al., 2012; Stuermer et al., 2015). Like other contact-dependent cells, hADSCs have also shown to migrate randomly in culture dishes. Once a population of cells reaches confluence in a culture well, they stop dividing and maintain a monolayer structure, as consistently seen in the culture in FBS-containing medium.
Previously we have observed that hADSCs isolated from eyelid adipose tissues could form aggregation when they were cultivated in a human serum (HS)-containing medium at higher density than confluence (Song et al., 2012). Since the aggregation occurred to accompany with the appearance of cell-free area (spacing), it was assumed to be a result from the three-dimensional migration of hADSCs from one place to another. Besides, the migrated cells could aggregate to form various types of cell mass (Yang et al., 2013).
During development, cell migration plays very important roles in immunosurveillance, wound healing, and tissue morphogenesis. In metastatic cancers, migration of the malignant cell also plays crucial roles in cancer invasion and metastasis (Friedl & Gilmour, 2009). Cancer cell migration shows two modes: single cell and collective cell migration (Kale et al., 2015). Single-cell migration can be subdivided into mesenchymal and amoeboid types which have different requirements of molecular signaling. Collective cell migration can be subdivided based on the characteristics of the cell group as solid strand, protrusive strand or clusters. However, cancer cells have also been shown to rely on plasticity to switch modes of migration depending on the availability of molecular machinery. Similar types of cell migration have also been suggested in mesenchymal cell migration (Petrie et al., 2009; Bear & Haugh, 2014). We have seen that HS-induced cell aggregation accompanied with changes of MMPs and tight junctional molecules during culture (Yang et al., 2013, Yoon et al., 2014). Since not all cells aggregate during culture in HS-containing medium, however, it is not clear whether these changes might be directly related to the cell aggregation phenomena.
In the present study, we wanted to address the question how HS could induce the formation of spacing from hADSCs monolayer during culture in vitro. For the purposes, cell adhesivity assay and selected drugs related to the cAMP metabolism and PI3K activity were used to seek out HS effect on spacing.
MATERIALS AND METHODS
hADSCs were isolated from abdominal adipose tissues which were donated by the patients undergoing liposuction with informed consent. All experiments were approved by Institutional Review Board of Seoul Women’s University. After several washings with Dulbecco’s phosphate-buffer saline (DPBS; Gibco, Grand Island, NY), lipoaspirate was mixed with 1× volume of 0.15% type I collagenase (Gibco) and incubated at 37°C for 1 h with gentle shaking. Then 1× volume of the culture medium consisting of DMEM-low glucose type (DMEM-LG; Gibco), 10% FBS (Gibco), 1× Anti-Anti (Gibco) and 3.7 mg/mL sodium bicarbonate was added to block collagenase activity and centrifuged at 500 × g for 10 min. Cell pellets were washed twice, and then plated in T25 culture flask (Nunc, Roskilde, Denmark) containing 5 mL of the culture medium. Cells were cultivated at 37°C with 5% CO2. When adherent cells were 70~80% confluent, they were detached using Hank’s balanced salt solution containing 0.125% trypsin (Gibco) and 1 mM EDTA at 37°C for 2 min and resuspended in the culture medium at a density of 4,000 cells/cm2. The culture medium was changed twice a week. At passage (p) 2 or p3, proliferated cells were cryopreserved in CryoTube vials (Nunc) at –196°C. For the experiment, cells were thawed and expanded in the culture medium for one week and then used. FBS and HS (Millipore, Temecula, CA) were heat-inactivated at 56°C for 30 min and kept frozen at –80°C until use. Unless indicated elsewhere, all reagents were supplied from Sigma (Seoul, Korea).
hADSCs were seeded in 24-well or 4-well plate (Nunc) at a density of 5-10 × 104 cells/well. Cells were cultivated in DMEM-LG containing 10% of FBS or HS. They were cultivated in duplicates and when a spacing appeared in both of two wells, the spacing was assessed as 100%. Spacing was assessed as positive when the diameter was 100 μm or larger. Forskolin 10 μM (Santa Cruz, Dallas, TX), isobutylmethyl xanthine 100 μM (IBMX; Lonza, Swiss), H89 20 μM, Y27632 10 μM and LY294002 10 μM (Cell signaling, Danvers, MA) were used.
To examine the adhesivity of cells, cells after culture were washed several times with PBS. Then cells were treated with 0.06% trypsin-EDTA (TE, Gibco) for 2 min. During this mild TE treatment, cells that loosely adhered to the plastic culture dishes were detached. After removal of detached cells by gentle washing, cells that remained tightly adhering to the dish were detached using 0.125% TE solution for 5 min. They were harvested and the cell number was counted using hemocytometer.
RESULTS
When hADSCs were cultivated in FBS-containing medium, they showed typical monolayer structure regardless of the high density (5-10 × 104 cells/well of 4- or 24-well dish) culture. Even after 8 days of culture, little structural change was observed in these cells (Fig. 1A). In contrast, cells cultivated under the same density but in HS-containing medium exhibited a remarkable change in the monolayer structure, showing appearance of cell-free areas (Fig. 1B-D). Morphology of these cell-free areas was variable including oval type or non-specific type. In most cases, the spacing accompanied with cell aggregates of various size of which diameter varied from a hundred to a dish well-diameter. Occasionally, when the spacings were observed in the vicinity of dish wells, hADSCs frequently aggregated to form a long, roll type aggregation consisting of a folded cell sheet or round cell mass with compact or loose cell arrangement (Fig. 1F).
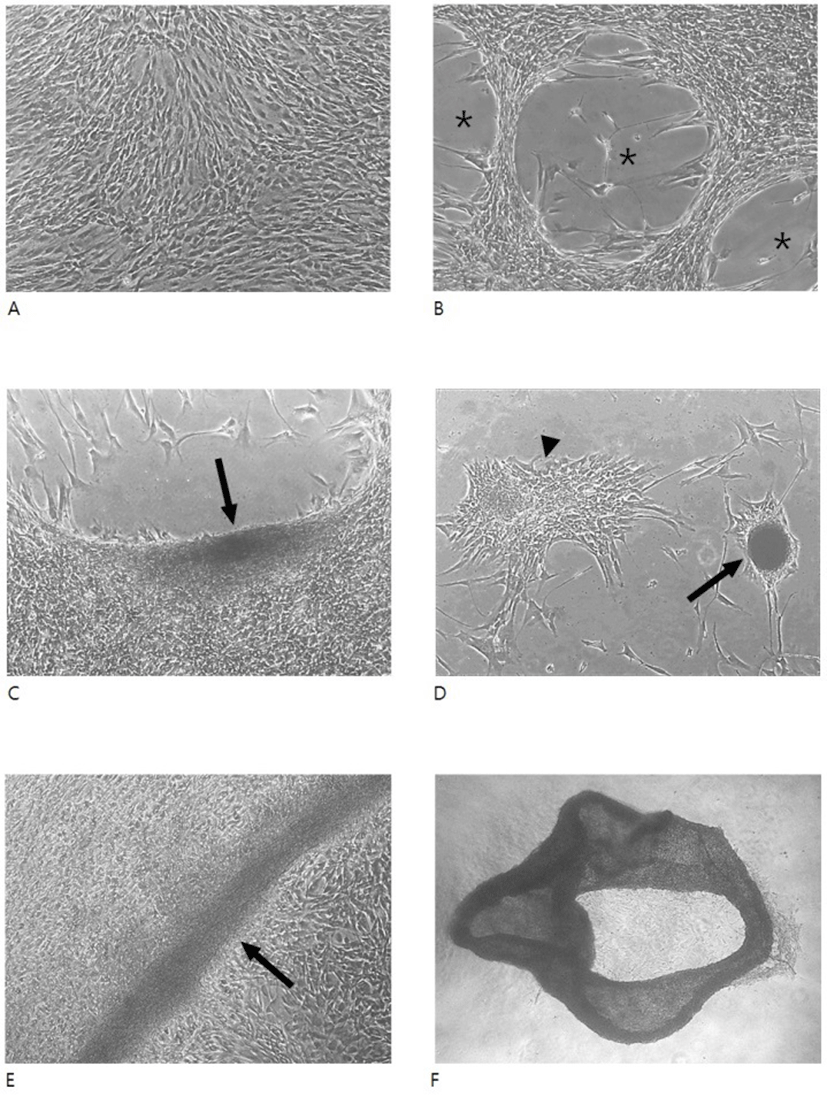
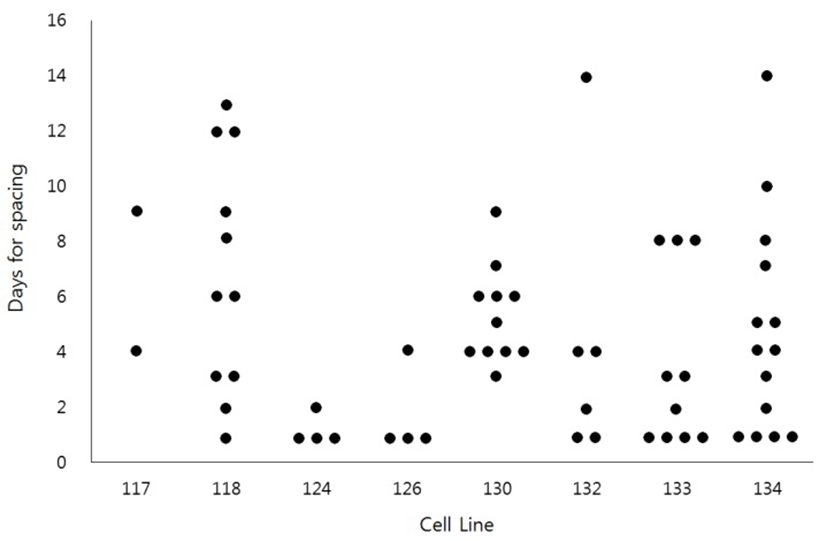
To see if the HS-induced spacing might be restricted to the specific cell lines, we have examined the HS effect sixty two times using eight different cell lines (Fig. 2). The results demonstrated that HS-induced spacing could happen in most cell lines. However, even the same cell lines showed a great variation of timing of spacing. For example, the spacing was observed as early as 1 day of culture or as late as 14 days of culture even in the same cell lines.
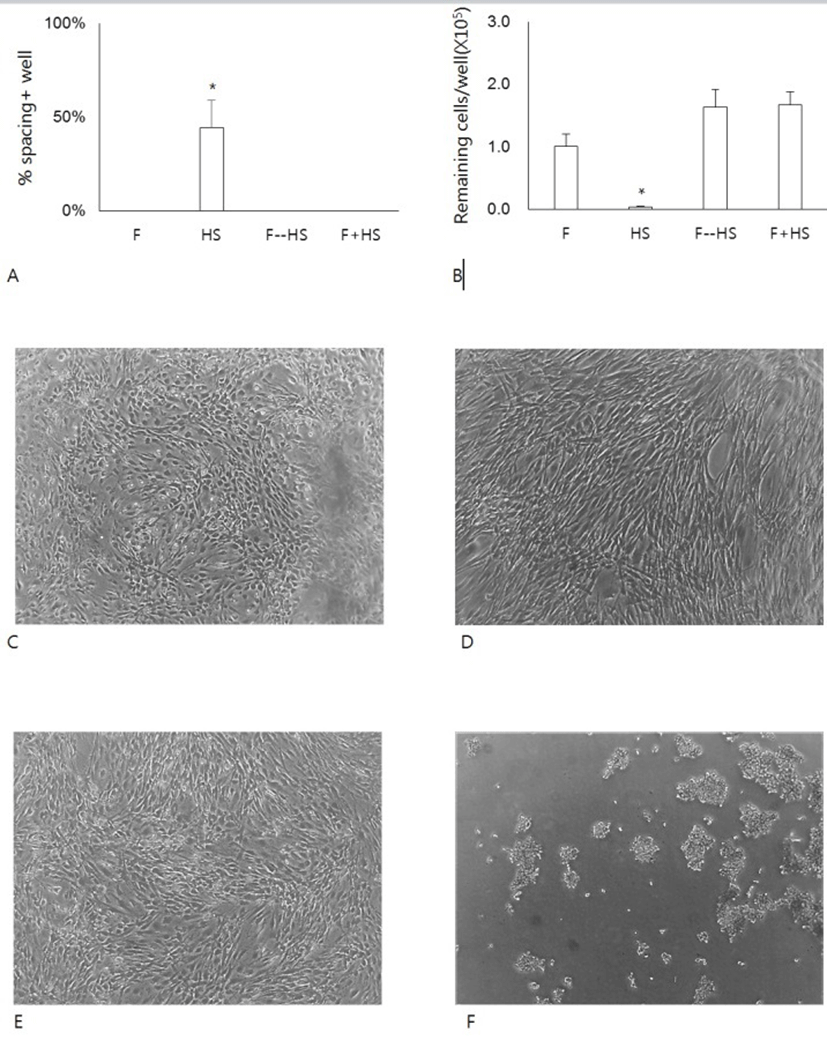
To investigate further the effect of HS on the formation of the spacing, cells were cultivated in FBS, HS or a mixture of FBS and HS. A group of cells were pretreated with FBS and then with HS (Fig. 3A). When cells were cultivated HS alone, they began to show the appearance of spacing as early as 1 day of culture. In contrast, cells cultured with FBS alone, with a mixture of FBS and HS did not show any spacing. Even cells pretreated with FBS alone for 1 day also showed little spacing during subsequent culture in the presence of HS. Since the spacing results from the migration of cells pre-existing in the area, we have questioned whether the spacing/migration somehow might be related to the changes of cell adhesivity to the culture dishes. To do this, we treated cells with 0.06% TE, which was lower concentration than 0.125% TE used for the subculture of hADSCs (Fig. 3B). At this concentration, almost cells cultured in FBS remained attached to the dishes. Similarly cells pretreated with FBS or cells cultured in FBS mixture also remained attached. In contrast, almost cells cultured in HS alone did not remain attached after the same treatment (Fig. 3C-F). These results demonstrate that either FBS might allow the strong attachment or HS might weaken the cell adhesivity.
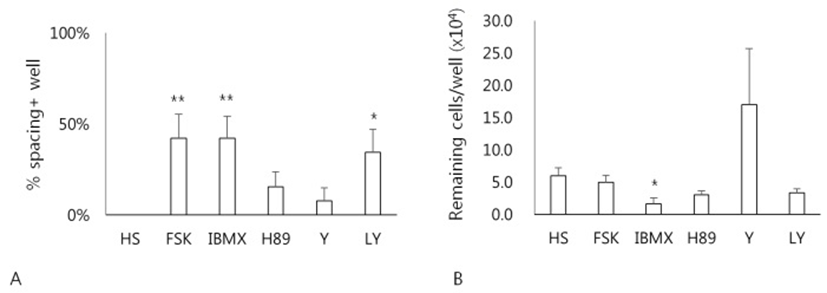
Whether a cAMP-dependent signaling pathway might be involved in the HS-induced spacing, effects of various drugs related to cAMP metabolism on the formation of spacing have been examined (Fig. 4). Treatment of cells with forskolin, an activator of adenylyl cyclase, IBMX, a cAMP phosphodiesterase inhibitor, or LY294002 significantly enhanced the formation of spacing compared to the HS alone (p<0.05) (Fig. 4A). However, neither H89, a protein kinase A inhibitor, nor Y27632, a ROCK inhibitor, showed any difference. Next, any relationship between the spacing and cell adhesivity in these cells was examined. When cells after culture were treated with 0.06% TE, only cells cultured in both IBMX and HS showed less number of remaining cells compared to the HS alone (Fig. 4B). Other groups of cells exhibited little difference of cell adhesivity compared to the cells of HS alone. These results demonstrate that cAMP-dependent signaling pathway might be involved in the HS-induced spacing and migration of hADSCs.
DISCUSSION
In the present study, we have observed that HS could induce three-dimensional migration of hADSCs overcoming contact-dependent property, and that cAMP-dependent signaling pathway might be involved in the HS-induced spacing event.
During normal development or in cancer, cells have shown to move/migrate in specific ways. Depending on the number of cells at the time and space of migration, a mode of cell migration can be classified as single cell migration or collective cell migration (Kale et al., 2015). Collective migration is that groups of cells move together via stable or transient cell–cell connections (Rørth, 2012; Theveneau & Mayor, 2012). It has been suggested that there are two different events, a collective movement of epithelial sheets and a cooperative interaction between looser mesenchymal cohorts (Scarpa & Mayor, 2016). During culture in the presence of HS, hADSCs showed several different types of cell aggregates. Some of them exhibited a folded sheet structure while others showed round type cell mass with compact or loose cell arrangement. Appearance of a folded cell sheet implies that cells on the sheet moved together. In contrast, irregular morphology of various sizes of cell mass suggest that cells on the mass might result from single cell migration. Taken together, it appears that HS-induced cell migration is heterogeneous and might take place via both modes of single cell migration and collective cell migration.
Our results showed that HS-induced cell migration was enhanced by forskolin and IBMX, which produce higher amount of cAMP in cytosol. These observations imply that the migration mechanism might involve cAMP signaling pathway. However, the facts that about half of cells did not respond and H89, a PKA inhibitor, failed to inhibit the space forming indicate that other pathway could also play a role in the event. Particularly, an observation that treatment of cells with LY294002, a PI3K inhibitor, also enhanced the space forming lead to suggest an involvement of PI3K signaling.
Single-cell migration is further subdivided into mesenchymal and amoeboid types which have different requirements of molecular signaling. Mesenchymal type migration is characterized by elongated morphology, dependency on focal adhesions, MMPs and stress fibers (Pankova et al., 2010). Amoeboid migration is characterized by ROCK dependency, round morphology, and is independent of MMP secretion (Kosla et al., 2013). In the present study, we have observed that treatment of cells with ROCK inhibitor did not show any significant change in cell migration or adhesivity. In this context, it is probable that a mode of HS-induced cell migration resembles mesenchymal type. However, further studies are needed to address the question. Opposite roles of ROCK in the cell migration have been demonstrated in different cell types. Wharton's jelly-derived MSCs exhibit active cell migration during culture in vivo. However, treatment of cells with ROCK inhibitor resulted in the inhibition of cell migration as confirmed by the scratch migration assay and time-lapse microscopy (Arora et al., 2015). Similarly, inhibition of ROCK has shown to disturb the collective cell migration of stratified TE-10 cells derived from a human esophageal cancer specimen (Mikami et al., 2015). On the contrary, ROCK inhibition in porcine aortic endothelial cells resulted in a farther collective cell migration when cells were cultured on soft substrates (Canver et al., 2016). Inhibition of ROCK in dental pulp cells significantly increased the cell migration rate while it facilitated early cell adhesion and spreading (Cheng et al., 2015). Also ROCK inhibition in C2C12 myoblasts increased migration velocity (Goetsch et al., 2014).
In some cases, cells exhibited aggregations consisting of a rolled or folded cell sheet. Together with the appearance of large cell-free space, these modes of cell migration should be regarded as a collective cell migration. In the collective cell migration, cell–cell interactions are required to mechanically link one cell to the other as well as to influence each other’s motile and protrusive behavior (Theveneau et al., 2010; Weber et al., 2012; Cai et al., 2014; Davis et al., 2015). In addition to cell–cell interactions, cells in a collective interact with their extracellular environment during migration. In particular, interactions with the ECM, with other tissues, and responses to chemotactic cues produced in the surrounding environment are believed to be essential for collective tissue guidance during embryonic development. Previously, we have observed that cells during HS-culture expressed higher level of CLDN3, -6, -7, -15, and -16 genes among the tight junction proteins, and higher level of JAM2 gene (Yoon et al., 2014). We found that HS treatment to the cells decreased the cell adhesivity to the dish surfaces, and this might help cells in the monolayer together form a roll-type cell sheet. However, as seen in the adhesivity assay results, decrease in the adhesivity did not always accompanied the migration. Therefore, while decreased adhesivity might be ensured for the HS-induced cell migration, other mechanisms are required to cell movement.
The present results suggest a presence of inhibitory activity in FBS, as seen in the observation that FBS pretreatment with 1 day inhibited the appearance of spacing during the subsequent HS culture for 6 days. If hADSCs possess self-spacing activity even in the absence of HS, then the absence of spacing in FBS suggest the presence of inhibitory activity in FBS. Similarly, if spacing is due by HS, then the absence of spacing in the presence of FBS suggest the presence of inhibitory activity of FBS. Taken together, our results suggest that FBS could possess inhibitory activity against the spacing of hADSCs in vitro.
While many cells migrated significantly during culture in response to HS, however, many cells in the same well did not show the remarkable movement. Since the size and the number of spacing in a well were largely variable, it might reflect the heterogeneity of hADSCs population. It has been shown that surface markers such as CD105 and CD166 increased during hADSCs culture, while the expression of CD34 decreased (Mitchell et al., 2006; Varma et al., 2007).
Based on the above experimental results, it is suggested that cell culture with HS could induce three-dimensional migration of hADSCs in vitro, and that cAMP signaling pathway might be involved in the event. It needs further studies to solve the question how important role the observed events might play in vivo.

