INTRODUCTION
It is evaluated that the developmental competence of oocyte has been improved by modulation of cyclic adenosine monophosphate (cAMP) levels during in vitro maturation (Gilchrist, 2011; Ezoe et al., 2015). Addition of a cAMP modulator forskolin or 3-isobutyl-1-methyl-xan-thine (IBMX) to the maturation media improves the developmental competence of vitrified-thawed GV oocytes as in natural GV oocytes (Ezoe et al., 2015). It based on the LH or FSH works through cAMP mediated signaling pathways (Dekel & Kraicer, 1978; Sela-Abramovich et al., 2006; Conti et al., 2012) and maturation promoting factor (Ezoe et al., 2015).
In meiotic arrest, spontaneous maturation is observed after releasing from follicle (Edwards, 1962; Pincus & Enzmann, 1935). So, follicular environment is important for oocyte competence. From 1970s, it is suggested that maintenance of appropriate cAMP levels is critical factor for meiotic resumption regulation (Luciano et al., 2011; Richard 2007; Cho et al., 1974). cAMP levels are under the multiple factors such as G protein-coupled receptor 3 (GPR3), GRP8, cyclases, phosphodiesterases, and growth factors such as EGF-like peptides (Richard, 2007; Conti et al., 2005; Kawamura et al., 2004). Adenosine increases FSH-stimulated accumulation of cAMP in cumulus-oocyte- complexs (COC) and markedly inhibits oocyte maturation in a dose-dependent manner (Miller & Behrman, 1986).
On the other hand, de novo synthesis of purines in somatic compartment of follicle and the timing and duration of such synthesis determines whether meiotic resumption will be suppressed or promoted in hormone mediated actions Downs & Verhoeven, 2003; Downs, 2000). The purine synthesis by de novo pathway is important in maintenance of meiotic arrest under the hormone. Purine de novo synthesis is nearly doubled in OCC by FSH treatment, and this response was completely prevented by adenosine (Downs, 2000). FSH had no effect on hypoxanthine salvage, although adenosine reduced this activity by 98% (Downs, 2000). hCG-induced maturation is blocked by adenosine as effectively as by the purine de novo synthesis inhibitor, azaserine (Downs, 2000). Cheon et al. (1997) revealed that the azaserine, de novo synthetic inhibitor, at GV intact stage increases the nuclear maturation and polar body extrusion rates. On the other hand, it inhibited the polar body extrusion rate if it is treated from GVB stage. So it is suggested that purine metabolism may concern with the maturation process with other signaling mediators. Salvage synthesis of purine is so far confused in oocyte maturation, although it is used to recover bases and nucleosides and can be converted back into nucleotides. Downs suggested that salvage pathway may be not required for meiotic induction (1997a, b) but he could not conformed (Downs, 2000).
The possible roles of purine metabolism including adenosine on germinal vesicle breakdown is mediated the cAMP. cAMP levels also showed fluctuation during maturation (Webb et al., 2008; Hegele-Hartung et al., 1999). On the other hand, it has been suggested a role as signaling mediator in oocyte maturation. However, it is largely un-known whether exogenous adenosine works as messenger during oocyte maturation or after maturation and its roles in oocyte developmental competence. In here, the possible roles of adenosine as messenger and its metabolism were evaluated with combination of purine synthesis metabolic inhibitors and adenosine.
MATERIALS AND METHODS
All animals used in this study were CD-1 mice (6-8 weeks old) and were handled according to the National Institutes of Health Standards for the use and care of animals. The animal protocols were approved by Sunshin University Institutional Animal Care and Use Committee. Animals were maintained on a 14L:10D light dark cycle under standard conditions. Food and water were provided ad libitium. For IVF sperm was obtained from same strain male mice between 8 and 12 weeks old.
Immature CD-1 mice (21-day-old) were injected with 5 IU of pregnant mare serum gonadotropin (Sigma) to stimulate the follicular development at 1:00 pm. Animals were sacrifice 46 hr later and oocytes were got through puncture the antral follicles with fine needle. The cumuli were removed physically using capillary pipet. Denuded germinal vesicle intact oocytes were pooled and washed three times in fresh medium and used. These processes were performed within 10 minutes. Oocytes were randomly allotted to treatment groups of about 100 per group. Groups were distinguished like followings: from GV intact group (fGV), from GVB group (fGVB), and from MII group (fMII). In fGV, chemicals were treated from GV intact stage and cultured for 18 hr. In GVB, chemicals were treated from GVB stage for 9 hr. In fMII, chemicals were treated in MII oocytes for 2 hr (from 15 hr after maturation induction). After culture, the MII stage oocytes were washed in fresh medium at least 5 times. Groups of 10 washed denuded oocytes were matured in 10 μL drops of equilibrated BWW medium containing 0.4% BAS covered with mineral oil at 37°C, 5% CO2 in air. To evaluate the possible roles of adenosine and its synthetic inhibitors, 750 μM adenosine (A), 20 μM azacerine (Aza), 20 μM hadacidine (Had), and 10 μM mycophenolic acid (MA) were used.
The effect of adenosine and its synthetic inhibitors on oocyte developmental competence was assessed by examining the capacity of the oocyte to support preimplantation embryo development following in vitro maturation. All the media used were BWW medium containing 0.4% BSA. Data presented percentage of 10 replicate experiments. About 100 oocytes were used per treatment group. IVF was performed as described in the Manipulating the Mouse Embryo (Hogan et al., 1994). Fertilization was carried out for 4 hr and then the eggs were washed with medium to remove sperms. 10 zygotes were place in 10 μL drops of equilibrated BWW medium containing 0.4% BAS covered with mineral oil at 37°C, 5% CO2 in air. Fertilization was assessed by the presence of second polar body and the development of 2-cell stage embryos the next day.
RESULTS
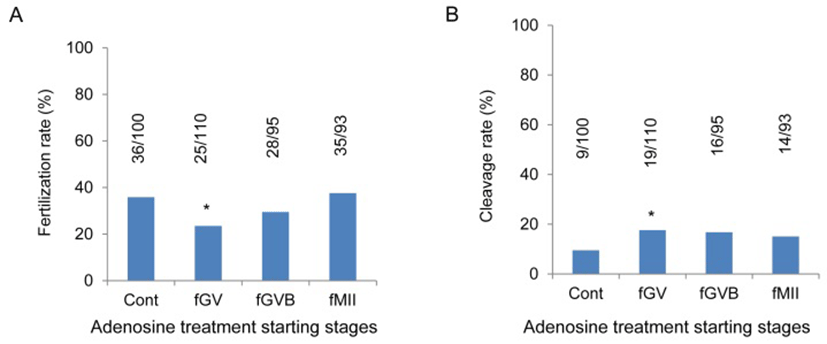
Most of the immature oocytes which were cultured with 750 μM adenosine were failed to become MII stage oocytes but after GVB stage adenosine did not showed specific effects as mentioned in previous report (Cheon et al., 1997). In the fGV groups, adenosine was treated from GV intact stage for 18 hr to induce spontaneous maturation. In the MII stage oocytes getting at 18 hr after incubation (fGV) the fertilization was significantly low compared with other groups. However, the cleavage rate was significantly high compared with the other groups (Fig. 1A, B). Adenosine did not increase or decrease the fertilization rate or cleavage rate in the other groups (Fig. 1A, B). It showed that the effects of adenosine on oocyte competence are existing at GV stage.
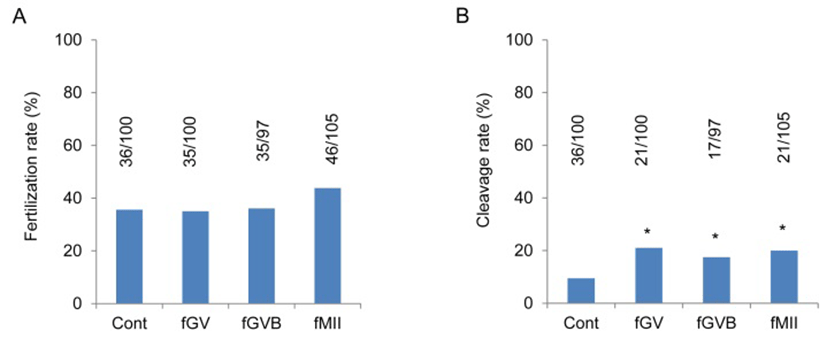
To suppress the endogenous adenosine in oocyte, hadacidin was employed. Hadacidin can suppress synthesis of adenosine from inosine monophosphate (IMP). Hadacidin did not increase or decrease the fertilization rates by the treatment starting stages (Fig. 2A). However, the cleavage rate was significantly higher in all three groups, fGV, fGVB and fMII compared with control (Fig. 2B). It clearly showed the inhibition of adenosine synthesis has positive effects on the improve oocyte quality.
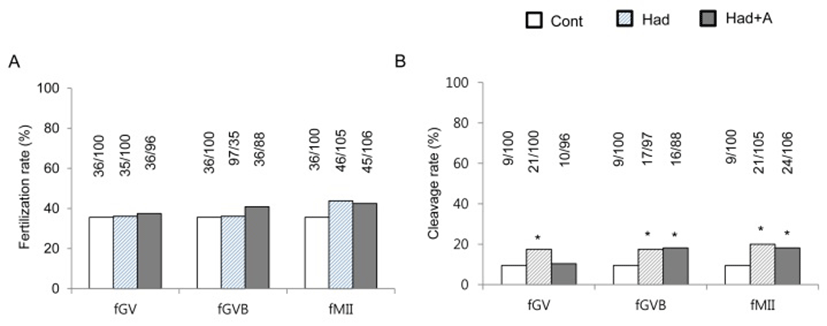
To know whether exogenous adenosine can block the effects of hadacidin on the oocyte developmental competence, hadacidin and adenosine were co-treated as mentioned at Materials and Methods. Adenosine did not block the effects of hadacidin in fGVB and fMII groups. However, it blocked the effects of hadacidin at fGV. It means that adenosine does not work as quantity compensation manners at GV stage at least.
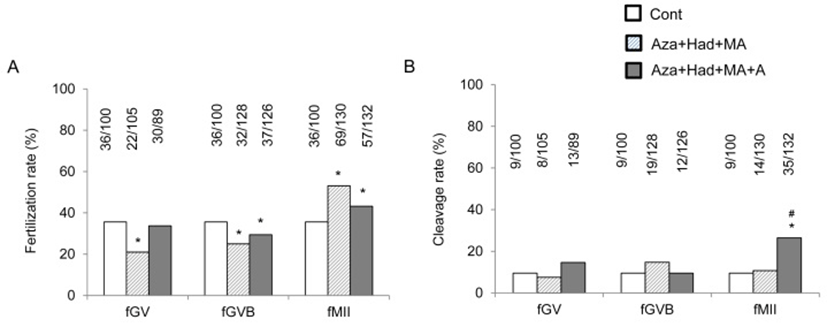
To evaluate the possible role adenosine in the oocyte maturation, synthetic pathways were blocked with azacerine, hadacidin, and mycophenolic acid at one time with or without adenosine. Fertilization rates were decreased in fGV and fGVB groups by the blocking of synthesis but increased in fMII group. Adenosine had opposite effect for the synthesis blocking at fGVB and fMII groups (Fig. 4A). Fertilization rate were improved by the adenosine compared with control and synthesis blocking groups only in fMII group (Fig. 4B).
DISCUSSION
Metabolism of purine nucleotides in oocyte or cumulus is suggested as one of the factors for meiotic arresting. Purine de novo or salvage syntheses are involved in regulation mechanism of oocyte maturation. Purine salvage synthesis supplies adenine or guanine from intermediates in the degradative pathway for nucleotides. Salvage pathways are used to recover bases and nucleosides. De novo synthesis begins with simple molecules such as amino acids and bicarbonate (Berg et al., 2002). Salvage synthesis is much less costly than the de novo synthesis. Adenylosuccinate synthetase catalyzes the first committed step in the biosynthesis of AMP form IMP. Adenine phosphoribosyl transferase catalyzes the formation of adenylate, whereas hypoxanthine-guanine phosphoribosyl transferase catalyzes the formation of inosinate and guanylate. It has been evaluated that these purine metabolism is important in meiotic arrest.
Previous data showed that adenosine treatment from GV intact stage significantly disturbed the maturation processes after GVB (Cheon et al., 1997). Interestingly, however, in the MII oocytes which were matured in the media containing adenosine, the cleavage rate was significantly higher than the other groups. Exogenous adenosine did not inhibit or accelerate of fertilization rate and cleavage rate of MII stage oocytes which were got from fGVB and fMII eggs. It can be supported by the previously evaluated results; meiotic suppression by cAMP-upregulation agents improves subsequent oocyte developmental competence (Luciano et al., 2011; Nogueira et al., 2003a, b). It means that adenosine work mediator of meiotic arrest as well as oocyte competence.
It has been suggested that adenosine blocks meiotic induction via its pronounced inhibition of the purine and pentose phosphate metabolic pathways (Downs, 2000). On the other hand, it also has been suggested that salvage synthesis of purine is not required for meiotic induction (Downs, 1997a, b). Interestingly, present study showed that the blocking of endogenous adenosine synthesis causes of increase of the cleavage rate. Besides, cotreatment of hadacidin and adenosine did not support quantitative compensation. Those suggest that exogenous adenosine work as messenger not as metabolic substance. It also can be supported by Fig. 4.
Previously we suggested that adenosine receptors in oocyte may regulate the maturation (Hwang & Cheon, 2013). It is supported by the present study. In GV intact oocytes adenosine improved fertilization rates without inhibit the effects of hadacidin. It suggests that the adenosine mediate signaling is involved in cytoplasmic maturation and improve developmental competence.
On summary, adenosine improved the cleavage rate when it treated from GV intact stage but not form GVB or MII. By the blocking the endogenous adenosine synthesis, the fertilization rates were increased in all groups, fGV, fGVB, and fMII. Besides, adenosine did not compensation the hadacidine in fGVG and fMII group except in fGV group. Those suggest that adenosine work with paracrine fashion during GV intact stage for oocyte development competence. Blocking de novo synthesis had the treatment stage specific effects on fertilization rate and adenosine only improved the cleavage rate. Put together those results suggest that adenosine works with paracrine fashion and adenosine metabolism has stage specific effects on oocyte developmental competence, although further studies in the molecular level are needed.

